Abstract
Main conclusion
In Brachiaria brizantha BbrizSERK1, BbrizSERK2 and BbrizSERK3 were identified. SERK expression marks somatic embryogenesis, sexual MMC, and sexual and apomictic PMC. BbrizSERK3 might have a regulatory role in reproductive development.
Abstract
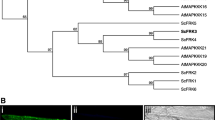
Somatic embryogenesis receptor-like kinase (SERK) consists of plasma membrane receptor genes that have been characterized in various species, associated with several aspects of plant development, including reproduction. SERK genes are involved in anther development and in early embryo development in sexual and asexual seed formation. To comprehend the complexity of the SERK genes and their function in Brachiaria reproduction, we performed a homology-based search in a genomic database of a sexual B. brizantha and identified sequences of three SERK genes, BbrizSERK1, BbrizSERK2, and BbrizSERK3. RNASeq data showed equivalent abundance of BbrizSERK1 and BbrizSERK2 transcripts in ovaries at early megasporogenesis of sexuals and apomicts, while BbrizSERK3 transcripts were more abundant in ovaries of sexuals than in apomicts. BbrizSERK3 results in three coding sequences due to alternative splicing, among them Variant 1 results in a protein with all the predicted domains of a SERK. BbrizSERK transcripts were detected in male reproductive tissues of both sexual and apomictic plants, suggesting a role in controlling anther development. BbrizSERK transcripts were detected early in ovule development, in the integuments, and in the megaspore mother cell of the sexual plant, but not in the cells that give rise to apomictic embryo sacs, suggesting a role in female reproductive development of sexuals. This paper provides evidences that SERK genes plays a role in the onset and establishment of somatic embryogenesis and in the reproductive development of B. brizantha and suggests a distinct role of BbrizSERK in apomixis initiation.







Similar content being viewed by others
Abbreviations
- AI:
-
Aposporic initials
- MMC:
-
Megaspore mother cell
- PMC:
-
Pollen mother cell
- SERK:
-
Somatic embryogenesis receptor-like kinase
References
aan den Toorn M, Albrecht C, de Vries S (2015) On the origin of SERKs: bioinformatics analysis of the somatic embryogenesis receptor kinases. Mol Plant 8(5):762–782. https://doi.org/10.1016/j.molp.2015.03.015
Albertini E, Marconi G, Reale L, Barcaccia G, Porceddu A, Ferranti F, Falcinelli M (2005) SERK and APOSTART. Candidate genes for apomixis in Poa pratensis. Plant Physiol 138:2185–2199. https://doi.org/10.1104/pp.105.062059
Albrecht C, Russinova E, Hecht V, Baaijens E, De Vries S (2005) The Arabidopsis thaliana SOMATIC EMBRYOGENESIS RECEPTOR-LIKE KINASES 1 and 2 control male sporogenesis. Plant Cell 17:3337–3349. https://doi.org/10.1105/tpc.105.036814
Alves ER, Carneiro VTC, Dusi DMA (2007) In situ localization of three cDNA sequences associated with the later stages of aposporic embryo sac development of Brachiaria brizantha. Protoplasma 231(3–4):161–171
Araujo ACG, Mukhambetzhanov S, Pozzobon MT, Santana EF, Carneiro VTC (2000) Female gametophyte development in apomictic and sexual Brachiaria brizantha (Poaceae). Rev Cytol Biol Vég Botaniste Tome 23(1–2):13–28
Baudino S, Hansen S, Brettschneider R, Hecht VRG, Dresselhaus T, Lörz H, Dumas C, Rogowsky PM (2001) Molecular characterisation of two novel maize LRR receptor-like kinases, which belong to the SERK gene family. Planta 213:1–10. https://doi.org/10.1007/s004250000471
Boddey RM, Macedo R, Tarré RM, Ferreira E, de Oliveira OC, Rezende CP, Cantarutti RB, Pereira JM, Alves BJR, Urquiaga S (2004) Nitrogen cycling in Brachiaria pastures: the key to understanding the process of pasture decline. Agric Ecosyst Environ 103:389–403. https://doi.org/10.1016/j.agee.2003.12.010
Cabral GB, Carneiro VTC, Lacerda AL, do Valle CB, Martinelli AP, Dusi DMA (2011) Somatic embryogenesis and organogenesis in apomictic and sexual Brachiaria brizantha. Plant Cell Tissue Organ Cult 107(2):271–282. https://doi.org/10.1007/s11240-011-9978-7
Cabral GB, Carneiro VTC, Rossi ML, da Silva JP, Martinelli AP, Dusi DMA (2015) Plant regeneration from embryogenic callus and cell suspensions of Brachiaria brizantha. Vitro Cell Dev Biol Plant 51(3):369–377. https://doi.org/10.1007/s11627-015-9690-0
Cheung AY, Wu H-M (2015) Stomatal patterning: SERKs put the mouths in their right place. Curr Biol 25(19):R838–R840. https://doi.org/10.1016/j.cub.2015.08.049
Colcombet J, Boisson-Dernier A, Ros-Palau R, Vera CE, Schroeder JI (2005) Arabidopsis SOMATIC EMBRYOGENESIS RECEPTOR KINASES 1 and 2 are essential for tapetum development and microspore maturation. Plant Cell 17(12):3350–3361. https://doi.org/10.1105/tpc.105.036731
Drechsel G, Kahles A, Kesarwani AK, Stauffer E, Behr J, Drewe P, Rätsch G, Wachter A (2013) Nonsense-mediated decay of alternative precursor mRNA splicing variants is a major determinant of the Arabidopsis steady state transcriptome. Plant Cell 25(10):3726–3742. https://doi.org/10.1105/tpc.113.115485
Du J, Yin H, Zhang S, Wei Z, Zhao B, Zhang J, Gou X, Lin H, Li J (2012) Somatic embryogenesis receptor kinases control root development mainly via brassinosteroid-independent actions in Arabidopsis thaliana. J Integr Plant Biol 54(6):388–399. https://doi.org/10.1111/j.1744-7909.2012.01124.x
Dusi DMA (2001) Apomixis in Brachiaria decumbens Stapf. PhD thesis, University of Wageningen. https://edepot.wur.nl/192704. Accessed 07 July 2020
Dusi DMA (2015) Hibridização in situ para detecção da expressão de genes em tecidos vegetais. In: Brasileiro ACM, Carneiro VTC (eds) Manual de transformação genética de plantas, 2nd edn. Embrapa, Brasilia, pp 303–327
Dusi DMA, Willemse MTM (1999) Apomixis in Brachiaria decumbens Stapf.: gametophytic development and reproductive calendar. Acta Biol Cracov Bot 41:151–162
Dusi DMA, Alves ER, Willemse MTM, Falcão R, Valle CB, Carneiro VTC (2010) Toward in vitro fertilization in Brachiaria spp. Sex Plant Rep 23:187–197
Felsenstein J (1985) Phylogenies and the comparative method. Am Nat 125(1):1–15
Gómez JF, Talle B, Wilson ZA (2015) Anther and pollen development: a conserved developmental pathway. J Integr Plant Biol 57(11):876–891. https://doi.org/10.1111/jipb.12425
Gruszka D (2013) The brassinosteroid signaling pathway—new key players and interconnections with other signaling networks crucial for plant development and stress tolerance. Int J Mol Sci 14:8740–8774. https://doi.org/10.3390/ijms14058740
Hecht V, Vielle-Calzada JP, Hartog MV, Schmidt EDL, Boutilier K, Grossniklaus U, de Vries SC (2001) The Arabidopsis SOMATIC EMBRYOGENESIS RECEPTOR KINASE 1 gene is expressed in developing ovules and embryos and enhances embryogenic competence in culture. Plant Physiol 127:803–816. https://doi.org/10.1104/pp.010324
Heese A, Hann DR, Gimenez-Ibanez S, Jones AME, He K, Li J, Schroeder JI, Peck SC, Rathjen JP (2007) The receptor-like kinase SERK3/BAK1 is a central regulator of innate immunity in plants. Proc Natl Acad Sci USA 104(29):12217–12222. https://doi.org/10.1073/pnas.0705306104
Hu H, Xiong L, Yang Y (2005) Rice SERK1 gene positively regulates somatic embryogenesis of cultured cell and host defense response against fungal infection. Planta 222:107–117. https://doi.org/10.1007/s00425-005-1534-4
Hu B, Jin J, Guo A-Y, Zhang H, Luo J, Gao G (2015) GSDS 2.0: an upgraded gene feature visualization server. Bioinformatics 31(8):1296–1297. https://doi.org/10.1093/bioinformatics/btu817
Kumar S, Stecher G, Tamura K (2016) MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33(7):1870–1874. https://doi.org/10.1093/molbev/msw054
Kwaaitaal MACJ, de Vries SC (2007) The SERK1 gene is expressed in procambium and immature vascular cells. J Exp Bot 58(11):2887–2896. https://doi.org/10.1093/jxb/erm103
Li Z, Wang Y, Huang J, Ahsan N, Biener G, Paprocki J, Thelen JJ, Raicu V, Zhao D (2017) Two SERK receptor-like kinases interact with EMS1 to control anther cell fate determination. Plant Physiol 173(1):326–337. https://doi.org/10.1104/pp.16.01219
Ma J, He Y, Wu C, Liu H, Hu Z, Sun G (2012) Cloning and molecular characterization of a SERK gene transcriptionally induced during somatic embryogenesis in Ananas comosus cv. Shenwan. Plant Mol Biol Rep 30:195–203. https://doi.org/10.1007/s11105-011-0330-5
Meng X, Chen X, Mang H, Liu C, Yu X, Gao X, Torii Keiko U, He P, Shan L (2015) Differential function of Arabidopsis SERK family receptor-like kinases in stomatal patterning. Curr Biol 25(18):2361–2372. https://doi.org/10.1016/j.cub.2015.07.068
Meng X, Zhou J, Tang J, Li B, de Oliveira Marcos VV, Chai J, He P, Shan L (2016) Ligand-induced receptor-like kinase complex regulates floral organ abscission in Arabidopsis. Cell Rep 14(6):1330–1338. https://doi.org/10.1016/j.celrep.2016.01.023
Nielsen H (2017) Predicting secretory proteins with signal P. Protein function prediction. Methods Mol Biol 1611:59–73. https://doi.org/10.1007/978-1-4939-7015-5_6
Nolan KE, Kurdyukov S, Rose RJ (2009) Expression of the SOMATIC EMBRYOGENESIS RECEPTOR-LIKE KINASE1 (SERK1) gene is associated with developmental change in the life cycle of the model legume Medicago truncatula. J Exp Bot 60(6):1759–1771. https://doi.org/10.1093/jxb/erp046
Nolan KE, Kurdyukov S, Rose RJ (2011) Characterisation of the legume SERK-NIK gene superfamily including splice variants: implications for development and defence. BMC Plant Biol 11(1):44. https://doi.org/10.1186/1471-2229-11-44
Oliveira EJ, Koehler AD, Rocha DI, Vieira LM, Pinheiro MVM, Matos EM, Cruz ACF, Silva TCR, Tanaka FAO, Nogueira FTS, Otoni WC (2017) Morpho-histological, histochemical, and molecular evidences related to cellular reprogramming during somatic embryogenesis of the model grass Brachypodium distachyon. Protoplasma 254(5):2017–2034. https://doi.org/10.1007/s00709-017-1089-9
Petersen TN, Brunak S, von Heijne G, Nielsen H (2011) Signal P 4.0: discriminating signal peptides from transmembrane regions. Nat Methods 8:785–786. https://doi.org/10.1038/nmeth.1701
Podio M, Felitti SA, Siena LA, Delgado L, Mancini M, Seijo JG, González AM, Pessino SC, Ortiz JP (2014) Characterization and expression analysis of SOMATIC EMBRYOGENESIS RECEPTOR KINASE (SERK) genes in sexual and apomictic Paspalum notatum. Plant Mol Biol 84:479–495. https://doi.org/10.1007/s11103-013-0146-9
Robinson JT, Thorvaldsdóttir H, Winckler W, Guttman M, Lander ES, Getz G, Mesirov JP (2011) Integrative genomics viewer. Nat Biotechnol 29:24–26. https://doi.org/10.1038/nbt.1754
Rocha DI, Monte-Bello CC, Aizza LCB, Dornelas MC (2016a) A passion fruit putative ortholog of the SOMATIC EMBRYOGENESIS RECEPTOR KINASE1 gene is expressed throughout the in vitro de novo shoot organogenesis developmental program. Plant Cell Tiss Organ Cult 125:107–117. https://doi.org/10.1007/s11240-015-0933-x
Rocha DI, Pinto DLP, Vieira LM, Tanaka FAO, Dornelas MC, Otoni WC (2016b) Cellular and molecular changes associated with competence acquisition during passion fruit somatic embryogenesis: ultrastructural characterization and analysis of SERK gene expression. Protoplasma 253:595–609. https://doi.org/10.1007/s00709-015-0837-y
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4(4):406–425. https://doi.org/10.1093/oxfordjournals.molbev.a040454
Santa-Catarina C, Hanai LR, Dornelas MC, Viana AM, Floh EIS (2004) SERK gene homolog expression, polyamines and amino acids associated with somatic embryogenetic competence of Ocotea catharinensis Mez. (Lauraceae). Plant Cell Tissue Organ Cult 79:53–61. https://doi.org/10.1023/B:TICU.0000049450.56271.f0
Savona M, Mattioli R, Nigro S, Falasca G, Della Rovere F, Costantino P, De Vries S, Ruffoni B, Trovato M, Altamura MM (2012) Two SERK genes are markers of pluripotency in Cyclamen persicum Mill. J Exp Bot 63(1):471–488. https://doi.org/10.1093/jxb/err295
Schmidt A (2020) Controlling apomixis: shared features and distinct characteristics of gene regulation. Genes 11(3):329. https://doi.org/10.3390/genes11030329
Schmidt EDL, Guzzo F, Toonen MAJ, De Vries SC (1997) A leucine-rich repeat containing receptor-like kinase marks somatic plant cells competent to form embryos. Development 124:2049–2062
Sharma SK, Millam S, Hein I, Bryan GJ (2008) Cloning and molecular characterization of a potato SERK gene transcriptionally induced during initiation of somatic embryogenesis. Planta 228:319. https://doi.org/10.1007/s00425-008-0739-8
Silveira ED, Alves-Ferreira M, Guimarães L, Silva FR, Carneiro VTC (2009) Selection of reference genes for quantitative real-time PCR expression studies in the apomictic and sexual grass Brachiaria brizantha. BMC Plant Biol 9:84. https://doi.org/10.1186/1471-2229-9-84
Singh A, Khurana P (2017) Ectopic expression of Triticum aestivum SERK genes (TaSERKs) control plant growth and development in Arabidopsis. Sci Rep 7:12368. https://doi.org/10.1038/s41598-017-10038-1
Singla B, Khurana JP, Khurana P (2009) Structural characterization and expression analysis of the SERK/SERL gene family in rice (Oryza sativa). Int J Plant Genom. https://doi.org/10.1155/2009/539402
Somleva MN, Schimdt EDL, de Vries SC (2000) Embryogenic cells in Dactylis glomerata L. (Poaceae) explants identified by cell tracking and by SERK expression. Plant Cell Rep 19:718–726
Steiner N, Santa-Catarina C, Guerra MP, Cutri L, Dornelas MC, Floh EIS (2012) A gymnosperm homolog of SOMATIC EMBRYOGENESIS RECEPTOR-LIKE KINASE-1 (SERK1) is expressed during somatic embryogenesis. Plant Cell Tissue Organ Cult 109:41–50. https://doi.org/10.1007/s11240-011-0071-z
Szakonyi D, Duque P (2018) Alternative splicing as a regulator of early plant development. Front Plant Sci 9:1174. https://doi.org/10.3389/fpls.2018.01174
Tucker MR, Araujo ACG, Paech N, Hecht V, Schmidt EDL, Rossel J-B, de Vries SC, Koltunow AMG (2003) Sexual and apomictic reproduction in Hieracium subgenus Pilosella are closely interrelated developmental pathways. Plant Cell 15:1524–1537
Yang C, Vizcay-Barrena G, Conner K, Wilson ZA (2007) MALE STERILITY1 is required for tapetal development and pollen wall biosynthesis. Plant Cell 19:3530–3548. https://doi.org/10.1105/tpc.107.054981
Zhang S, Liu X, Lin Y, Xie G, Fu F, Liu H, Wang J, Gao S, Lan H, Rong T (2011) Characterization of a ZmSERK gene and its relationship to somatic embryogenesis in a maize culture. Plant Cell Tissue Organ Cult 105:29–37. https://doi.org/10.1007/s11240-010-9834-1
Zhang H, Lin X, Han Z, Wang J, Qu L-J, Chai J (2016) SERK family receptor-like kinases function as co-receptors with PXY for plant vascular development. Mol Plant 9(10):1406–1414. https://doi.org/10.1016/j.molp.2016.07.004
Acknowledgements
The authors acknowledge the Conselho Nacional de Desenvolvimento Científico e Tecnológico, (CNPq) for the doctorate scholarship (141959/2006-1) for ADK and Research Fellowship for APM (305785/2008-7), and financial support—project 476332/2013-3; Empresa Brasileira de Pesquisa Agropecuária (Embrapa) (project 12.14.01.014.00.00) and Fundação de Apoio à Pesquisa do Distrito Federal, (FAPDF)—Project 00193.0000039-99 for financial support. Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001. ADK thanks Larissa Arrais Guimarães for helpful advice on laboratory technical issues.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Dorothea Bartels.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Koehler, A.D., Irsigler, A.S.T., Carneiro, V.T.C. et al. SERK genes identification and expression analysis during somatic embryogenesis and sporogenesis of sexual and apomictic Brachiaria brizantha (Syn. Urochloa brizantha). Planta 252, 39 (2020). https://doi.org/10.1007/s00425-020-03443-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00425-020-03443-w