Abstract
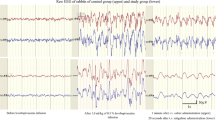
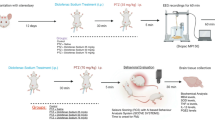
Ibuprofen is a non-steroidal anti-inflammatory drug (NSAID) that is commonly used as an anti-inflammatory, anti-pyretic, and analgesic. Although some studies have focused on the anti-inflammatory and anti-pyretic properties of ibuprofen during febrile convulsions, only one has investigated its antiepileptic effects. In the present study, we aimed to investigate the effects of ibuprofen in rats exposed to pentylenetetrazol (PTZ)-induced seizures. In total, 48 rats were randomly divided in two groups: Group A for electroencephalography (EEG) recordings and Group B for behavioral assessment. All EEG recordings and behavioral assessment protocols were performed. In addition, groups were compared in terms of prostaglandin F2 alfa (PGF2α) levels in the brain. We demonstrated the beneficial effects of the administration of ibuprofen in PTZ-induced seizures in rats via the following findings: spike percentages and Racine convulsion scale values were significantly lower and first myoclonic jerk (FMJ) onset times were significantly higher in the ibuprofen-administered groups. Moreover, PGF2α levels in the brain were significantly higher in the saline and PTZ 70 mg/kg group than in the control and PTZ 70 mg/kg and 400 mg/kg ibuprofen groups. Our study is the first to demonstrate the beneficial effects of ibuprofen on seizures through behavioral, EEG, and PGF2α brain assessments. Ibuprofen can be used for epilepsy and febrile seizures safely and without inducing seizures. However, further experimental and clinical studies are needed to confirm our results.


Similar content being viewed by others
References
French JA, Pedley TA (2008) Clinical practice. Initial management of epilepsy. N Engl J Med 359:166–176. https://doi.org/10.1056/NEJMcp0801738
Chen H, He H, Xiao Y et al (2019) Losigamone add-on therapy for focal epilepsy. Cochrane Database Syst. Rev. 2019
Gibson AP, Patel NC (2008) Mechanisms of action of antiepileptic drugs. In: Antiepileptic Drugs to Treat Psychiatric Disorders. Taylor and Francis, pp 1–15
Lehmann JM, Lenhard JM, Oliver BB et al (1997) Peroxisome proliferator-activated receptors α and γ are activated by indomethacin and other non-steroidal anti-inflammatory drugs. J Biol Chem 272:3406–3410. https://doi.org/10.1074/jbc.272.6.3406
Boje KMK, Jaworowicz D, Raybon JJ (2003) Neuroinflammatory role of prostaglandins during experimental meningitis: Evidence suggestive of an in vivo relationship between nitric oxide and prostaglandins. J Pharmacol Exp Ther 304:319–325. https://doi.org/10.1124/jpet.102.041533
McElrath TF, Allred EN, Van Marter L et al (2013) Perinatal systemic inflammatory responses of growth-restricted preterm newborns. Acta Paediatr Int J Paediatr 102:439–442. https://doi.org/10.1111/apa.12339
Parepally JMR, Mandula H, Smith QR (2006) Brain uptake of nonsteroidal anti-inflammatory drugs: Ibuprofen, flurbiprofen, and indomethacin. Pharm Res 23:873–881. https://doi.org/10.1007/s11095-006-9905-5
Kokki H, Kumpulainen E, Lehtonen M et al (2007) Cerebrospinal fluid distribution of ibuprofen after intravenous administration in children. Pediatrics 120:1002–1008. https://doi.org/10.1542/peds.2007-0064
Gournay V, Savagner C, Thiriez G et al (2002) Pulmonary hypertension after ibuprofen prophylaxis in very preterm infants. Lancet 359:1486–1488. https://doi.org/10.1016/S0140-6736(02)08424-6
Tatli MM, Kumral A, Duman N et al (2004) Spontaneous intestinal perforation after oral ibuprofen treatment of patent ductus arteriosus in two very-low-birthweight infants. Acta Paediatr Int J Paediatr 93:999–1001. https://doi.org/10.1080/08035250410031279
Desfrere L, Zohar S, Morville P et al (2005) Dose-finding study of ibuprofen in patent ductus arteriosus using the continual reassessment method. J Clin Pharm Ther 30:121–132. https://doi.org/10.1111/j.1365-2710.2005.00630.x
Ambat MTC, Ostrea EM, Aranda JV (2008) Effect of ibuprofen L-lysinate on bilirubin binding to albumin as measured by saturation index and horseradish peroxidase assays. J Perinatol 28:287–290. https://doi.org/10.1038/sj.jp.7211925
Ohlsson A, Shah PS (2018) Paracetamol (acetaminophen) for patent ductus arteriosus in preterm or low birth weight infants. Cochrane Database Syst. Rev. 2018
Wixey JA, Chand KK, Pham L et al (2018) Therapeutic potential to reduce brain injury in growth restricted newborns. J Physiol 596:5675–5686
A NP,G, M. B, et al (2015) Brain Growth Gains and Losses in Extremely Preterm Infants at Term. Cereb Cortex 25:1897–1905
Peng J, Wu S, Guo C et al (2019) Effect of Ibuprofen on Autophagy of Astrocytes During Pentylenetetrazol-Induced Epilepsy and its Significance: An Experimental Study. Neurochem Res 44:2566–2576. https://doi.org/10.1007/s11064-019-02875-5
Erbas O, Yilmaz M, Korkmaz HA et al (2013) Oxytocin inhibits pentylentetrazol-induced seizures in the rat. Peptides 40:141–144. https://doi.org/10.1016/j.peptides.2012.12.003
Erbaş O, Solmaz V, Aksoy D (2015) Inhibitor effect of dexketoprofen in rat model of pentylenetetrazol-induced seizures. Neurol Res 37:1096–1101. https://doi.org/10.1179/1743132814Y.0000000391
Danbolt NC (2001) Glutamate uptake (review, online-version). Prog Neurobiol 65:1–105
Kalani M, Shariat M, Khalesi N et al (2016) A comparison of early ibuprofen and indomethacin administration to prevent intraventricular hemorrhage among preterm infants. Acta Med Iran 54:788–792
Liu S, Pan G, Liu G et al (2017) Electrospun fibrous membranes featuring sustained release of ibuprofen reduce adhesion and improve neurological function following lumbar laminectomy. J Control Release 264:1–13. https://doi.org/10.1016/j.jconrel.2017.08.011
Sekiyama K, Fujita M, Sekigawa A et al (2012) Ibuprofen ameliorates protein aggregation and astrocytic gliosis, but not cognitive dysfunction, in a transgenic mouse expressing dementia with Lewy bodies-linked P123H β-synuclein. Neurosci Lett 515:97–101. https://doi.org/10.1016/j.neulet.2012.03.037
Yoshikawa K, Kita Y, Kishimoto K, Shimizu T (2006) Profiling of eicosanoid production in the rat hippocampus during kainic acid-induced seizure: Dual phase regulation and differential involvement of COX-1 AND COX-2. J Biol Chem 281:14663–14669. https://doi.org/10.1074/jbc.M511089200
Claycomb RJ, Hewett SJ, Hewett JA (2012) Neuromodulatory role of endogenous interleukin-1β in acute seizures: Possible contribution of cyclooxygenase-2. Neurobiol Dis 45:234–242. https://doi.org/10.1016/j.nbd.2011.08.007
Kim HJ, Chung JI, Lee SH et al (2008) Involvement of endogenous prostaglandin F2α on kainic acid-induced seizure activity through FP receptor: The mechanism of proconvulsant effects of COX-2 inhibitors. Brain Res 1193:153–161. https://doi.org/10.1016/j.brainres.2007.12.017
Salvadori MGSS, Banderõ CRR, Jesse AC et al (2012) Prostaglandin E 2 potentiates methylmalonate-induced seizures. Epilepsia 53:189–198. https://doi.org/10.1111/j.1528-1167.2011.03326.x
Régnier A, Vicaut E, Mraovitch S (2010) Aggravation of seizure-associated microvascular injuries by ibuprofen may involve multiple pathways. Epilepsia 51:2412–2422. https://doi.org/10.1111/j.1528-1167.2009.02480.x
Serrano GE, Lelutiu N, Rojas A et al (2011) Ablation of cyclooxygenase-2 in forebrain neurons is neuroprotective and dampens brain inflammation after status epilepticus. J Neurosci 31:14850–14860. https://doi.org/10.1523/JNEUROSCI.3922-11.2011
Baik EJ, Kim EJ, Lee SH, Moon CH (1999) Cyclooxygenase-2 selective inhibitors aggravate kainic acid induced seizure and neuronal cell death in the hippocampus. Brain Res 843:118–129. https://doi.org/10.1016/S0006-8993(99)01797-7
Holtman L, Van Vliet EA, Edelbroek PM et al (2010) Cox-2 inhibition can lead to adverse effects in a rat model for temporal lobe epilepsy. Epilepsy Res 91:49–56. https://doi.org/10.1016/j.eplepsyres.2010.06.011
Egg D, Herold M, Rumpl E, Günther R (1980) Prostaglandin F2α levels in human cerebrospinal fluid in normal and pathological conditions. J Neurol 222:239–248. https://doi.org/10.1007/BF00313153
Candelario-Jalil E, Slawik H, Ridelis I et al (2005) Regional distribution of the prostaglandin E2 receptor EP1 in the rat brain: Accumulation in Purkinje cells of the cerebellum. J Mol Neurosci 27:303–310. https://doi.org/10.1385/JMN:27:3:303
Baran H, Heldt R, Hertting G (1987) Increased prostaglandin formation in rat brain following systemic application of kainic acid. Brain Res 404:107–112. https://doi.org/10.1016/0006-8993(87)91360-6
Naffah-Mazzacoratti MG, Bellíssimo MI, Cavalheiro EA (1995) Profile of prostaglandin levels in the rat hippocampus in pilocarpine model of epilepsy. Neurochem Int 27:461–466. https://doi.org/10.1016/0197-0186(95)80003-4
Haba R, Shintani N, Onaka Y et al (2014) Central CRTH2, a second prostaglandin D2 receptor, mediates emotional impairment in the lipopolysaccharide and tumor-induced sickness behavior model. J Neurosci. https://doi.org/10.1523/JNEUROSCI.1407-13.2014
Watanabe K (2002) Prostaglandin F synthase. Prostaglandins Other Lipid Mediat 68–69:401–407. https://doi.org/10.1016/S0090-6980(02)00044-8
Urade Y, Kitahama K, Ohishi H et al (1993) Dominant expression of mRNA for prostaglandin D synthase in leptomeninges, choroid plexus, and oligodendrocytes of the adult rat brain. Proc Natl Acad Sci 90:9070–9074. https://doi.org/10.1073/pnas.90.19.9070
Nelson KB, Ellenberg JH (1976) Predictors of epilepsy in children who have experienced febrile seizures. N Engl J Med 295:1029–1033. https://doi.org/10.1056/NEJM197611042951901
Förstermann U, Neufang B, Hertting G (1982) Rapid and effective conversion of 6-keto-prostaglandin F1α to 6,15-diketo-13,14-dihydro-prostaglandin F1α-immunoreactive material in vivo. Prostaglandins Leukot Med 9:277–284. https://doi.org/10.1016/S0262-1746(82)80014-0
Tamai I, Takei T, Maekawa K, Ohta H (1983) Prostaglandin F2α concentrations in the cerebrospinal fluid of children with febrile convulsions, epilepsy and meningitis. Brain Dev 5:357–362. https://doi.org/10.1016/S0387-7604(83)80039-4
Lyneham RC, Mcleod JG, Smith ID et al (1973) Convulsions and electroencephalogram abnormalities after intra-amniotic prostaglandin F2a. Lancet 302:1003–1005. https://doi.org/10.1016/S0140-6736(73)91094-5
Acknowledgements
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical Approval
The experimental procedures employed in the present study were approved by the Animal Ethics Committee (51879863-59a). Present manuscript was approved by Local Institutional Animal Care and Ethics Committee.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Durankuş, F., Şenkal, E., Sünnetçi, E. et al. Beneficial Effects of Ibuprofen on Pentylenetetrazol-induced Convulsion. Neurochem Res 45, 2409–2416 (2020). https://doi.org/10.1007/s11064-020-03101-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-020-03101-3