Abstract
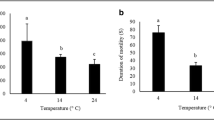
The importance of reactive oxygen species and the antioxidant system in sperm biology has been recognized for different bony fishes but nothing is known in this regard for chondrichthyans. For the first time for cartilaginous fishes, the enzymatic antioxidant system was shown herein to be present in both fractions of sperm (spermatozoa and seminal fluid) collected from two different places (seminal vesicle and cloaca). In internally fertilizing freshwater ocellate river stingray, Potamotrygon motoro, the activity of superoxide dismutase and glutathione peroxidase was not changed upon sperm transition from the seminal vesicle to the cloaca. The activity of catalase was significantly increased for both sperm fractions at transition from the seminal vesicle to the cloaca (1.6 times for spermatozoa and 1.9 times for seminal fluid). The role of the sperm antioxidant system for different aspects of internal fertilization is discussed. The presented results are the initiatory step in uncovering the biochemical events of internal reproduction in Chondrichthyes.


Similar content being viewed by others
Data availability
The original data of the study are available on request.
References
Aitken RJ, Bennetts L (2006) Reactive oxygen species: friend or foe. In: De Jonge C, Barratt C (eds) The sperm cell: production, maturation, fertilization, regeneration, 1st edn. Cambridge University Press, New York, pp 170–193
Alves LMF, Nunes M, Marchand P, Le Bizec B, Mendes S, Correia JPS, Lemos MFL, Novais SC (2016) Blue sharks (Prionace glauca) as bioindicators of pollution and health in the Atlantic Ocean: contamination levels and biochemical stress responses. Sci Total Environ 563-564:282–292
Asakawa T, Matsushita S (1980) Coloring conditions of thiobarbituric acid test for detecting lipid hydroperoxides. Lipids 15:137–140
Baker MA, Netherton J, Aitken RJ (2018) From past to present: an historical overview of the concept of spermatozoa, reactive oxygen species, and male-factor infertility. In: Henkel R, Samanta L, Agarwal A (eds) Oxidants, antioxidants and impact of the oxidative status in male reproduction, 1st edn. Academic Press, London, pp 17–26
Barrera-García A, O'Hara T, Galván-Magaña F, Méndez-Rodríguez LC, Castellini JM, Zenteno-Savin T (2013) Trace elements and oxidative stress indicators in liver and kidney of the blue shark (Prionace glauca). Comp Biochem Phys A 165:483–490
Bize I, Santander G, Cabello P, Driscoll D, Sharpe C (1991) Hydrogen peroxide is involved in hamster sperm capacitation in vitro. Biol Reprod 44:398–403
Dadras H, Dzyuba V, Cosson J, Golpour A, Dzyuba B (2016) The in vitro effect of temperature on motility and antioxidant response of common carp Cyprinus carpio spermatozoa. J Therm Biol 59:64–68
Dadras H, Dzyuba V, Golpour A, Xin M, Dzyuba B (2019) In vitro antioxidant enzyme activity and sperm motility at different temperatures in sterlet Acipenser ruthenus and rainbow trout Oncorhynchus mykiss. Fish Physiol Biochem 45:1791–1800
de Lamirande E, Gagnon C (1993a) Human sperm hyperactivation and capacitation as parts of an oxidative process. Free Radic Biol Med 14:157–166
de Lamirande E, Gagnon C (1993b) Human sperm hyperactivation in whole semen and its association with low superoxide scavenging capacity in seminal plasma. Fertil Steril 59:1291–1295
Durairajanayagam D (2018) Physiological role of reactive oxygen species in male reproduction. In: Henkel R, Samanta L, Agarwal A (eds) Oxidants, antioxidants and impact of the oxidative status in male reproduction, 1st edn. Academic Press, London, pp 65–78
Dzyuba V, Dzyuba B, Cosson J, Boryshpolets S, Yamaner G, Kholodniy V, Rodina M (2014) The antioxidant system of sterlet seminal fluid in testes and Wolffian ducts. Fish Physiol Biochem 40:1731–1739
Dzyuba V, Cosson J, Dzyuba B, Yamaner G, Rodina M, Linhart O (2016) The antioxidant system of seminal fluid during in vitro storage of sterlet Acipenser ruthenus sperm. Fish Physiol Biochem 42:563–568
Dzyuba V, Ninhaus-Silveira A, Kahanec M, Veríssimo-Silveira R, Rodina M, Holt WV, Dzyuba B (2019) Sperm motility in ocellate river stingrays: evidence for post-testicular sperm maturation and capacitation in Chondrichthyes. J Zool 307:9–16
Gorbi S, Pellegrini D, Tedesco S, Regoli F (2004) Antioxidant efficiency and detoxification enzymes in spotted dogfish Scyliorhinus canicula. Mar Environ Res 58:293–297
Hagedorn M, McCarthy M, Carter VL, Meyers SA (2012) Oxidative stress in zebrafish (Danio rerio) sperm. PLoS One 7:e39397
Lahnsteiner F, Mansour N (2010) A comparative study on antioxidant systems in semen of species of the Percidae, Salmonidae, Cyprinidae, and Lotidae for improving semen storage techniques. Aquaculture 307:130–140
Lahnsteiner F, Mansour N, Plaetzer K (2010) Antioxidant systems of brown trout (Salmo trutta f. fario) semen. Anim Reprod Sci 119:314–321
Lawrence RA, Burk RF (1976) Glutathione peroxidase activity in selenium-deficient rat liver. Biochem Biophys Res Commun 71:952–958
López-Cruz RI, Zenteno-Savin T, Galván-Magaña F (2010) Superoxide production, oxidative damage and enzymatic antioxidant defenses in shark skeletal muscle. Comp Biochem Phys A 156:50–56
MacLeod J (1943) The role of oxygen in the metabolism and motility of human spermatozoa. Am J Phys 138:512–518
Marklund S, Marklund G (1974) Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur J Biochem 47:469–474
Marklund S, Nordensson I, Back O (1981) Normal CuZn superoxide dismutase, Mn superoxide dismutase, catalase and glutathione peroxidase in Werner’s syndrome. J Gerontol 36:405–409
Martínez-Páramo S, Diogo P, Dinis MT, Herráez MP, Sarasquete C, Cabrita E (2012) Incorporation of ascorbic acid and α-tocopherol to the extender media to enhance antioxidant system of cryopreserved sea bass sperm. Theriogenology 77:1129–1136
McCarthy MJ, Baumber J, Kass PH, Meyers SA (2010) Osmotic stress induces oxidative cell damage to rhesus macaque spermatozoa. Biol Reprod 82:644–651
Rosa R, Paula JR, Sampaio E, Pimentel M, Lopes AR, Baptista M, Guerreiro M, Santos C, Campos D, Almeida-Val VMF, Calado R, Diniz M, Repolho T (2016) Neuro-oxidative damage and aerobic potential loss of sharks under elevated CO2 and warming. Mar Biol 163:119
Shalgi R, Smith TT, Yanagimachi R (1992) A quantitative comparison of the passage of capacitated and uncapacitated hamster spermatozoa through the uterotubal junction. Biol Reprod 46:419–424
Shaliutina-Kolešová A, Gazo J, Cosson O, Linhart O (2013) Comparison of oxidant and antioxidant status of seminal plasma and spermatozoa of several fish species. Czech J Anim Sci 58:313–320
Smith HW (1931) The absorption and excretion of water and salts by the elasmobranch fishes. I. Fresh water elasmobranchs. Am J Phys 98:279–295
Vélez-Alavez M, Labrada-Martagón V, Méndez-Rodriguez LC, Galván-Magaña F, Zenteno-Savín T (2013) Oxidative stress indicators and trace element concentrations in tissues of mako shark (Isurus oxyrinchus). Comp Biochem Phys A 165:508–514
Wang X, Shi X, Liu Y, Yu D, Guan S, Liu Q, Li J (2016) Effects of chilled storage and cryopreservation on sperm characteristics, antioxidant enzyme activities, and lipid peroxidation in Pacific cod Gadus microcephalus. Chin J Oceanol Limn 34:763–771
Zini A, de Lamirande E, Gagnon C (1993) Reactive oxygen species in semen of infertile patients: levels of superoxide dismutase- and catalase-like activities in seminal plasma and spermatozoa. Int J Androl 16:183–188
Acknowledgments
The authors are grateful to the staff of LINEO (Laboratory of Neotropical Ichthyology), Department of Biology and Zootechny, São Paulo State University, Ilha Solteira, SP, Brazil, for their technical support and provision of facilities.
Funding
Funding was provided by the Czech Science Foundation (No. 16-03754S), by the Ministry of Education, Youth and Sports of the Czech Republic—project “CENAKVA” (LM2018099), by project Biodiversity (CZ.02.1.01./0.0/0.0/16_025/0007370 Reproductive and genetic procedures for preserving fish biodiversity and aquaculture), and by the Grant Agency of the University of South Bohemia in Ceske Budejovice (125/2016/Z).
Author information
Authors and Affiliations
Contributions
Viktoriya Dzyuba, Borys Dzyuba, and Marek Rodina designed the experiments; Alexandre Ninhaus-Silveira and Rosicleire Veríssimo-Silveira provided the facilities for fish maintenance and participated in sample collection; Viktoriya Dzyuba and Borys Dzyuba performed the experiments and analyzed the experimental data; Viktoriya Dzyuba wrote the manuscript with input from all the authors; Borys Dzyuba, Marek Rodina, Alexandre Ninhaus-Silveira, and Rosicleire Veríssimo-Silveira revised the manuscript. All the authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
All manipulations with animals were performed in accordance with the authorization for the use of experimental animals (Reference number: 2293/2015-MZE-17214 (16OZ22302/2014-17214), Czech Republic) and approved by the local Ethics Commission on Animal Use (CEUA-FEIS/UNESP 14/2018).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Dzyuba, V., Ninhaus-Silveira, A., Veríssimo-Silveira, R. et al. Sperm antioxidant system in ocellate river stingray Potamotrygon motoro at transition from seminal vesicle to cloaca. Fish Physiol Biochem 46, 1975–1980 (2020). https://doi.org/10.1007/s10695-020-00848-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-020-00848-y