Abstract
Objectives
To compare different approaches for the expression of an anti-PCSK9 biosimilar monoclonal antibody (mAb) in CHO cells using IRES-mediated tricistronic plasmid vectors combining different signal peptides, IRES elements and selection markers.
Results
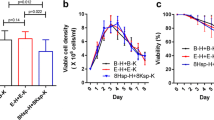
Transient transfection indicated a similar level of secreted mAb 48 h post-transfection for all constructs. However, transfections carried out with circular plasmids showed a higher expression than with linearized plasmids. After two months under selection pressure, only part of the transfected pools recovered. The cultures co-transfected using two antibiotics as selection markers for double selection did not recover. Growth, metabolism and mAb production profiles of the only part of the transfected pools recovered resulting stable pools were compared and the stable pool transfected with circular L1-LC-IRES-H7-HC-IRES-NEO plasmid was chosen for further studies, due to higher cell growth and mAb production. Critical quality attributes of the protein A-purified mAb such as purity, homogeneity, binding affinity to PCSK9, and amino acid sequence were assessed confirming the success of the approach adopted in this study.
Conclusions
The expression platform proposed showed to be efficient to produce a high-quality anti-PCSK9 mAb in stable CHO cell pools and provides benchmarks for fast production of different mAbs for characterization, formulation studies and pre-clinical investigation.







Similar content being viewed by others
References
Ahmadi S, Davami F, Davoudi N, Nematpour F, Ahmadi M, Ebadat S, Azadmanesh K, Barkhordari F, Mahboudi F (2017) Monoclonal antibodies expression improvement in CHO cells by PiggyBac transposition regarding vectors ratios and design. PLoS ONE 12:e0179902
Balasubramanian S, Rajendra Y, Baldi L, Hacker DL, Wurm FM (2016) Comparison of three transposons for the generation of highly productive recombinant CHO cell pools and cell lines. Biotechnol Bioeng 113:1234–1243
Bayat H, Hossienzadeh S, Pourmaleki E, Ahani R, Rahimpour A (2018) Evaluation of different vector design strategies for the expression of recombinant monoclonal antibody in CHO cells. Prep Biochem Biotechnol 48:160–164
Berkowitz SA, Engen JR, Mazzeo JR, Jones GB (2012) Analytical tools for characterizing biopharmaceuticals and the implications for biosimilars. Nat Rev Drug Discov 11:527–540
Catapano AL, Pirillo A, Norata GD (2017) Anti-PCSK9 antibodies for the treatment of heterozygous familial hypercholesterolemia: patient selection and perspectives. Vasc Health Risk Manag 13:343–351
EMA (2015) Committee for medicinal products for human use (CHMP)—Assessment report Repatha. https://www.ema.europa.eu/en/documents/assessment-report/repatha-epar-public-assessment-report_en.pdf. Accessed 10 Jan 2020
Haryadi R, Ho S, Kok YJ, Pu HX, Zheng L, Pereira NA, Li B, Bi X, Goh LT, Yang Y, Song Z (2015) Optimization of heavy chain and light chain signal peptides for high level expression of therapeutic antibodies in CHO cells. PLoS ONE 10:e0116878
Ho SCL, Bardor M, Feng H, Wah Y, Song Z, Yap MGS, Yang Y (2012) IRES-mediated tricistronic vectors for enhancing generation of high monoclonal antibody expressing CHO cell lines. J Biotechnol 57:130–139
Ho SCL, Koh YCE, Van Beers M, Mueller M, Wan C, Teo G, Song Z, Tong YM, Bardor M, Yang Y (2013) Control of IgG LC:HC ratio in stably transfected CHO cells and study of the impact on expression, aggregation, glycosylation and conformational stability. J Biotechnol 165:157–166
Kirchhoff CF, Wang XM, Conlon HD, Anderson S, Ryan AM, Bose A (2017) Biosimilars: key regulatory considerations and similarity assessment tools. Biotechnol Bioeng 114:2696–2705
Kunert R, Reinhart D (2016) Advances in recombinant antibody manufacturing. Appl Microbiol Biot 100:3451–3461
McLenachan S, Sarsero JP, Ioannou PA (2007) Flow-cytometric analysis of mouse embryonic stem cell lipofection using small and large DNA constructs. Genomics 89:708–720
Ng SK, Lin W, Sachdeva R, Wang DI, Yap MG (2010) Vector fragmentation: characterizing vector integrity in transfected clones by Southern blotting. Biotechnol Progr 26:11–20
Noh S, Shin S, Lee G (2018) Comprehensive characterization of glutamine synthetase-mediated selection for the establishment of recombinant CHO cells producing monoclonal antibodies. Sci Rep 8:5361
Rahimpour A, Bayat H, Omidi M, Peyrovan M, Mohammadian O, Naderi F (2016) Stable expression of anti-CD52 monoclonal antibody using a bicistronic vector system. Biol Med (Aligarh) 8:341
Stuchbury G, Münch G (2010) Optimizing the generation of stable neuronal cell lines via pre-transfection restriction enzyme digestion of plasmid DNA. Cytotechnology 62:189–194
Von Groll A, Levin Y, Barbosa MC, Ravazzolo AP (2006) Linear DNA low efficiency transfection by liposome can be improved by the use of cationic lipid as charge neutralizer. Biotechnol Prog 22:1220–1224
WHO (2013) International Nonproprietary Names for Pharmaceutical Substances (INN). WHO Drug Information Vol. 27, No. 3. https://www.who.int/medicines/publications/druginformation/issues/RL_70.pdf?ua=1. Accessed 10 Oct 2020
Wirth M (1997) Isolation of recombinant cell clones exhibiting high-level expression of the introduced gene. In: Hauser H, Wager R (eds) Mammalian cell biotechnology in protein production. Walter de Gruyter, Berlin, p 128
Yang Y, You M, Chen F, Jia T, Chen Y, Zhou B, Mi Q, An Z, Luo W, Xia N (2018) Efficient development of a stable cell pool for antibody production using a single plasmid. J Biochem 163:391–398
Yeo JH, Ho SC, Mariati M, Koh E, Tay SJ, Woen S, Zhang P, Yang Y (2017) Optimized selection marker and CHO host cell combinations for generating high monoclonal antibody producing cell lines. Biotechnol J 12:1700175
Zagari F, Jordan M, Stettler M, Broly H, Wurm F (2013) Lactate metabolism shift in CHO cell culture: the role of mitochondrial oxidative activity. New Biotechnol 30:238–245
Acknowledgements
The authors would like to acknowledge the Brazilian research funding agencies CAPES, CNPq, and FAPERJ for financial support. The authors also thank Augusto Vieira (UEMP/UFRJ) and Eduardo Matos (CEMBIO/UFRJ) for support in the peptide mapping assay.
Supporting information
Supplementary Table 1. List of the forward (F) and reverse (R) primer pairs used for Sanger sequencing and PCR analysis carried out to validate correct subcloning into pCI-neo vector.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cruz, T.A., Pinho, M.B. & Castilho, L.R. Evaluation of different IRES-mediated tricistronic plasmid designs for expression of an anti-PCSK9 biosimilar monoclonal antibody in CHO cells. Biotechnol Lett 42, 2511–2522 (2020). https://doi.org/10.1007/s10529-020-02952-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-020-02952-8