Abstract
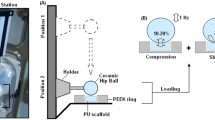
The development of new technologies to produce three-dimensional and biocompatible scaffolds associated with high-end cell culture techniques have shown to be promising for the regeneration of tissues and organs. Some biomedical devices, as meniscus prosthesis, require high flexibility and tenacity and such features are found in polyurethanes which represent a promising alternative. The Poly(PCL–TMC)urethane here presented, combines the mechanical properties of PCL with the elasticity attributed by TMC and presents great potential as a cellular carrier in cartilage repair. Scanning electron microscopy showed the presence of interconnected pores in the three-dimensional structure of the material. The scaffolds were submitted to proliferation and cell differentiation assays by culturing mesenchymal stem cells in bioreactor. The tests were performed in dynamic flow mode at the rate of 0.4 mL/min. Laser scanning confocal microscopy analysis showed that the flow rate promoted cell growth and cartilage ECM synthesis of aggrecan and type II collagen within the Poly(PCL–TMC)urethane scaffolds. This study demonstrated the applicability of the polymer as a cellular carrier in tissue engineering, as well as the ECM was incremented only when under oriented flow rate stimuli. Therefore, our results may also provide data on how oriented flow rate in dynamic bioreactors culture can influence cell activity towards cartilage ECM synthesis even when specific molecular stimuli are not present. This work addresses new perspectives for future clinical applications in cartilage tissue engineering when the molecular factors resources could be scarce for assorted reasons.










Similar content being viewed by others

References
Anderson DE, Johnstone B (2017) Dynamic mechanical compression of chondrocytes for tissue engineering: a critical review. Front Bioeng Biotechnol 5:76. https://doi.org/10.3389/fbioe.2017.00076
Birmingham E, Grogan JA, Niebur GL, Mcnamara LM, Mchugh PE (2013) Computational modelling of the mechanics of trabecular bone and marrow using fluid structure interaction techniques. Ann Biomed Eng 41(4):814–826
Brunelli M, Perrault C, Lacroix D (2019) A review of bioreactors and mechanical stimuli. In: Multiscale mechanobiology in tissue engineering, pp. 1–22
Bulgheroni E, Grassi A, Campagnolo M, Bulgheroni P, Mudhigere A, Gobbi A (2016) Comparative study of collagen versus synthetic-based meniscal scaffolds in treating meniscal deficiency in young active population. Cartilage 7(1):29–38. https://doi.org/10.1177/1947603515600219
Campagnoli C, Roberts IA, Kumar S, Bennett PR, Bellantuono I, Fisk NM (2001) Identification of mesenchymal stem/progenitor cells in human first-trimester fetal blood, liver, and bone marrow. Blood 98(8):2396–2402. https://doi.org/10.1182/blood.v98.8.2396
Caro F, Perdisa F, Dhollander A, Verdonk R, Verdonk P (2020) Meniscus scaffolds for partial meniscus defects. Clin Sports Med 39(1):83–92. https://doi.org/10.1016/j.csm.2019.08.011
Cartmell SH, Porter BD, García AJ, Guldberg RE (2003) Effects of medium perfusion rate on cell-seeded three-dimensional bone constructs in vitro. Tissue Eng 9(6):1197–1203. https://doi.org/10.1089/10763270360728107
Chen G, Ushida T, Tateishi T (2002) Scaffold design for tissue engineering. Macromol Biosci 2(2):67–77. https://doi.org/10.1002/1616-5195(20020201)2:2<67:AID-MABI67>3.0.CO;2-F
Chia SL, Gorna K, Gogolewski S, Alini M (2006) Biodegradable elastomeric polyurethane membranes as chondrocyte carriers for cartilage repair. Tissue Eng 12(7):1945–1953. https://doi.org/10.1089/ten.2006.12.1945
Dos Santos DJ, Tavares LB, Batalha GF (2012) Mechanical and physical properties investigation of polyurethane material obtained from renewable natural source. J Achiev Mater Manuf Eng 54(2):211–217
Elder BD, Athanasiou KA (2009) Hydrostatic pressure in articular cartilage tissue engineering: from chondrocytes to tissue regeneration. Tissue Eng Part B 15(1):43–53. https://doi.org/10.1089/ten.teb.2008.0435
Ge J, Guo L, Wang S, Zhang Y, Cai T, Zhao RCH, Wu Y (2014) The size of mesenchymal stem cells is a significant cause of vascular obstructions and stroke. Stem Cell Rev Rep 10(2):295–303. https://doi.org/10.1007/s12015-013-9492-x
Gomes ME, Sikavitsas VI, Behravesh E, Reis RL, Mikos AG (2003) Effect of flow perfusion on the osteogenic differentiation of bone marrow stromal cells cultured on starch-based three-dimensional scaffolds. J Biomed Mater Res A 67(1):87–95. https://doi.org/10.1002/jbm.a.10075
Gomes RC (2017) Síntese e Caracterização da Poli(PCL-TMC)uretana para Utilização como Biomaterial. Dissertação, Universidade Federal de São Carlos. https://repositorio.ufscar.br/handle/ufscar/9491.
Heath CA (2000) Cells for tissue engineering. Trends Biotechnol 18(1):17–19. https://doi.org/10.1016/S0167-7799(99)01396-7
Ikada Y (2006) Challenges in tissue engineering. J R Soc Interface 3(10):589–601. https://doi.org/10.1098/rsif.2006.0124
Jakus AE, Geisendorfer NR, Lewis PL, Shah RN (2018) 3D-printing porosity: a new approach to creating elevated porosity materials and structures. Acta Biomater 72:94–109. https://doi.org/10.1016/j.actbio.2018.03.039
Junqueira LC, Carneiro J (2013) Histologia Básica: Texto e Atlas, 12th edn. Guanabara Koogan, Rio de Janeiro
Kalvelytė AV, Imbrasaitė A, Krestnikova N, Stulpinas, A (2017) Adult stem cells and anticancer therapy. In: Advances in molecular toxicology, pp 123–202.
Langer R, Vacanti J (1993) Tissue engineering. Science 260(5110):920–926. https://doi.org/10.1126/science.8493529
Leroy A, Beaufils P, Faivre B, Steltzlen C, Boisrenoult P, Pujol N (2017) Actifit® polyurethane meniscal scaffold: MRI and functional outcomes after a minimum follow-up of 5 years. Orthop Traumatol Surg Res 103(4):609–614. https://doi.org/10.1016/j.otsr.2017.02.012
Liao J, Guo X, Grande-Allen KJ, Kasper FK, Mikos AG (2010) Bioactive polymer/extracellular matrix scaffolds fabricated with a flow perfusion bioreactor for cartilage tissue engineering. Biomaterials 31(34):8911–8920. https://doi.org/10.1016/j.biomaterials.2010.07.110
Lin S, Mequanint K (2017) Bioreactor-induced mesenchymal progenitor cell differentiation and elastic fiber assembly in engineered vascular tissues. Acta Biomater 59:200–209. https://doi.org/10.1016/j.actbio.2017.07.012
Mbarki M, Sharrock P, Fiallo M, Elfeki H (2017) Hydroxyapatite bioceramic with large porosity. Mater Sci Eng C 76:985–990. https://doi.org/10.1016/j.msec.2017.03.097
McMurray RJ, Gadegaard N, Tsimbouri PM, Burgess KV, Mcnamara LE, Tare R, Murawski K, Kingham E, Oreffo ROC, Dalby MJ (2011) Nanoscale surfaces for the long-term maintenance of mesenchymal stem cell phenotype and multipotency. Nat Mater 10(8):637–644. https://doi.org/10.1038/nmat3058
Njuguna J, Pielichowski K (2004) Recent developments in polyurethane-based conducting composites. J Mater Sci 39:4081–4094. https://doi.org/10.1023/B:JMSC.0000033387.51728.de
Peroglio M, Gaspar D, Zeugolis DI, Alini M (2018) Relevance of bioreactors and whole tissue cultures for the translation of new therapies to humans. J Orthop Res 36(1):10–21. https://doi.org/10.1002/jor.23655
Pittenger MF, Kerr C (2014) Stem cells. In: Tissue engineering, 2nd edn, pp 23–65.
Plunkett NA, Partap S, O’Brien FJ (2010) Osteoblast response to rest periods during bioreactor culture of collagen-glycosaminoglycan scaffolds. Tissue Eng Part A 16(3):943–951. https://doi.org/10.1089/ten.TEA.2009.0345
Puetzer JL, Petitte JN, Loboa EG (2010) Comparative review of growth factors for induction of three-dimensional in vitro chondrogenesis in human mesenchymal stem cells isolated from bone marrow and adipose tissue. Tissue Eng Part B 16(4):435–444. https://doi.org/10.1089/ten.TEB.2009.0705
Richardson SM, Kalamegam G, Pushparaj PN, Matta C, Memic A, Khademhosseini A, Mobasheri R, Poletti FL, Hoyland JA, Mobasheri A (2016) Mesenchymal stem cells in regenerative medicine: Focus on articular cartilage and intervertebral disc regeneration. Methods 99:69–80. https://doi.org/10.1016/j.ymeth.2015.09.015
Rosser J, Thomas DJ (2018) Bioreactor processes for maturation of 3D bioprinted tissue. In: 3D Bioprinting for reconstructive surgery, pp 191–215
Ruan ZB, Zhu L, Yin YG, Chen GC (2014) Karyotype stability of human umbilical cord-derived mesenchymal stem cells during in vitro culture. Exp Ther Med 8(5):1508–1512. https://doi.org/10.3892/etm.2014.1977
Sacchetti B, Funari A, Remoli C, Giannicola G, Kogler G, Liedtke S, Cossu G, Serafini M, Sampaolesi M, Tagliafico E, Tenedini E, Saggio I, Robey PG, Riminucci M, Bianco P (2016) No identical “Mesenchymal Stem Cells” at different times and sites: human committed progenitors of distinct origin and differentiation potential are incorporated as adventitial cells in microvessels. Stem Cell Rep 6(6):897–913. https://doi.org/10.1016/j.stemcr.2016.05.011
Schüller-Ravoo S, Feijen J, Grijpma DW (2012) Flexible, elastic and tear-resistant networks prepared by photo-crosslinking poly(trimethylene carbonate) macromers. Acta Biomater 8(10):3576–3585. https://doi.org/10.1016/j.actbio.2012.06.004
Setayeshmehr M, Esfandiari E, Rafienia M, Hashemibeni B, Taheri-Kafrani A, Samadikuchaksaraei A, Kaplan DL, Moroni L, Joghataei M (2019) Hybrid and composite scaffolds based on extracellular matrices for cartilage tissue engineering. Tissue Eng Part B 25(3):202–224. https://doi.org/10.1089/ten.TEB.2018.0245
Shahin K, Doran PM (2011) Strategies for enhancing the accumulation and retention of extracellular matrix in tissue-engineered cartilage cultured in bioreactors. PLoS ONE 6(8):e23119. https://doi.org/10.1371/journal.pone.0023119
Stiehler M, Bünger C, Baatrup A, Lind M, Kassem M, Mygind T (2008) Effect of dynamic 3-D culture on proliferation, distribution, and osteogenic differentiation of human mesenchymal stem cells. J Biomed Mater Res A 89(1):96–107. https://doi.org/10.1002/jbm.a.31967
Tonelli FMP, De Cássia Oliveira Paiva N, De Medeiros RVB, Pinto MCX, Tonelli FCP, Resende RR (2016) Tissue engineering: the use of stem cells in regenerative medicine. In: Current developments in biotechnology and bioengineering: human and animal health applications, pp 315–324.
Touchet TJ, Cosgriff-Hernandez EM (2016) Hierarchal structure–property relationships of segmented polyurethanes. In: Advances in polyurethane biomaterials, pp 3–22. https://doi.org/10.1016/B978-0-08-100614-6.00001-9.
Tuan RS, Chen AF, Klatt BA (2013) Cartilage regeneration. J Am Acad Orthop Surg 21(5):303–311. https://doi.org/10.5435/JAAOS-21-05-303
Verdonk R, Verdonk P, Huysse W, Forsyth R, Heinrichs EL (2011) Tissue ingrowth after implantation of a novel, biodegradable polyurethane scaffold for treatment of partial meniscal lesions. Am J Sports Med 39(4):774–782. https://doi.org/10.1177/0363546511398040
Vetsch JR, Müller R, Hofmann S (2015) The evolution of simulation techniques for dynamic bone tissue engineering in bioreactors. J Tissue Eng Regen Med 9(8):903–917. https://doi.org/10.1002/term.1733
Wang T, Gardiner BS, Lin Z, Rubenson J, Kirk TB, Wang A, Xu J, Smith DW, Lloyd DG, Zheng MH (2013) Bioreactor design for tendon/ligament engineering. Tissue Eng Part B 19(2):133–146. https://doi.org/10.1089/ten.TEB.2012.0295
Acknowledgements
The authors thanks to UNICAMP/FEM for assistance in scan electron microscopy and UFSCar-Sorocaba/PPGBMA for permission to use the Laser Scanning Confocal Microscope, Granted by Pró-Equipamentos/CAPES: 3420/2013-17, 2610/2014- 90.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that there is no conflict of interest regarding the publication of this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pedrini, F., Hausen, M., Gomes, R. et al. Enhancement of cartilage extracellular matrix synthesis in Poly(PCL–TMC)urethane scaffolds: a study of oriented dynamic flow in bioreactor. Biotechnol Lett 42, 2721–2734 (2020). https://doi.org/10.1007/s10529-020-02983-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-020-02983-1