Abstract
Understanding flow distributions in human lungs has attracted significant attention since the last few decades. However, there are still large discrepancies between different studies in the distribution of air flow into alveoli at different generations of bifurcation. In this study, a new method has been developed to calculate expansion ratio of alveoli and ratio of alveolar to ductal flow rate at different generations for air- and saline-filled lungs. The effects of alveolar number, breathing period, lung tidal volume, and surface tension are examined. It is found that the expansion ratio of alveoli varies significantly at different generations in the saline-filled lungs. For the air-filled lung, the expansion ratio of individual alveolus remains constant for different generations. The current study provides new data on the flow rate ratios which is critical for understanding flow distributions and flow behaviors in alveoli. Surface tension in alveoli and alveolar number has obvious effects on the value of flow ratio. The current study sheds new light into the flow behavior in lungs and lays the foundation for detailed study on flow and particle transport characteristics in human lungs.






Similar content being viewed by others
References
Anderson JO, Thundiyil JG, Stolbach A (2012) Clearing the air: a review of the effects of particulate matter air pollution on human health. J Med Toxicol 8:166–175. https://doi.org/10.1007/s13181-011-0203-1
Bachofen H, Schurch S (2001) Alveolar surface forces and lung architecture. Comp Biochem Physiol A Mol Integr Physiol 129:183–193. https://doi.org/10.1016/s1095-6433(01)00315-4
Bachofen H, Schurch S, Urbinelli M, Weibel ER (1987) Relations among alveolar surface-tension, surface-area, volume, and recoil pressure. J Appl Physiol 62:1878–1887
Barrett KE, Boitano S, Barman SM, Brooks HL (2016) Ganong’s review of medical physiology, 25th edn. McGraw-Hill Education, New York
Berg EJ, Weisman JL, Oldham MJ, Robinson RJ (2010) Flow field analysis in a compliant acinus replica model using particle image velocimetry (PIV). J Biomech 43:1039–1047. https://doi.org/10.1016/j.jbiomech.2009.12.019
Chang S et al (2013) Tracking X-ray microscopy for alveolar dynamics in live intact mice. Sci Rep 3:5. https://doi.org/10.1038/srep01304
Chen R, Hu B, Liu Y, Xu JX, Yang GS, Xu DD, Chen CY (2016) Beyond PM2.5: the role of ultrafine particles on adverse health effects of air pollution. Biochim Biophys Acta-Gen Subj 1860:2844–2855. https://doi.org/10.1016/j.bbagen.2016.03.019
Chhabra S, Prasad AK (2011) Flow and particle dispersion in lung acini: effect of geometric and dynamic parameters during synchronous ventilation. J Fluids Eng-Trans ASME 133:10. https://doi.org/10.1115/1.4004362
Dailey HL, Ghadiali SN (2007) Fluid-structure analysis of microparticle transport in deformable pulmonary alveoli. J Aerosol Sci 38:269–288. https://doi.org/10.1016/j.jaerosci.2007.01.001
Ferkol T, Schraufnagel D (2014) The global burden of respiratory disease. Ann Am Thorac Soc 11:404–406. https://doi.org/10.1513/AnnalsATS.201311-405PS
Fishler R, Mulligan MK, Sznitman J (2013) Acinus-on-a-chip: a microfluidic platform for pulmonary acinar flows. J Biomech 46:2817–2823. https://doi.org/10.1016/j.jbiomech.2013.08.020
Fishler R, Hofemeier P, Etzion Y, Dubowski Y, Sznitman J (2015) Particle dynamics and deposition in true-scale pulmonary acinar models. Sci Rep 5:11. https://doi.org/10.1038/srep14071
Fishler R, Ostrovski Y, Lu CY, Sznitman J (2017) Streamline crossing: an essential mechanism for aerosol dispersion in the pulmonary acinus. J Biomech 50:222–227. https://doi.org/10.1016/j.jbiomech.2016.11.043
Fonseca MD, Andrade CM, de Mello MJE, Crispi CP (2011) Effect of temperature on fluidity of irrigation fluids. Br J Anaesth 106:51–56. https://doi.org/10.1093/bja/aeq303
Fung YC (1975) Does surface-tension make lung inherently unstable. CircRes 37:497–502. https://doi.org/10.1161/01.Res.37.4.497
Gefen A, Elad D, Shiner RJ (1999) Analysis of stress distribution in the alveolar septa of normal and simulated emphysematic lungs. J Biomech 32:891–897. https://doi.org/10.1016/s0021-9290(99)00092-5
Grippi MA, Elias JA, Fishman JA, Kotloff RM, Pack AI, Senior RM, Siegel MD (2015) Fishman’s pulmonary diseases and disorders, 4th edn. McGraw-Hill Education, New York
Haefelibleuer B, Weibel ER (1988) Morphometry of the human pulmonary acinus. Anat Rec 220:401–414. https://doi.org/10.1002/ar.1092200410
Hajari AJ, Yablonskiy DA, Sukstanskii AL, Quirk JD, Conradi MS, Woods JC (2012) Morphometric changes in the human pulmonary acinus during inflation. J Appl Physiol 112:937–943. https://doi.org/10.1152/japplphysiol.00768.2011
Harding EM, Robinson RJ (2010) Flow in a terminal alveolar sac model with expanding walls using computational fluid dynamics. Inhal Toxicol 22:669–678. https://doi.org/10.3109/08958371003749939
Harrington L, Prisk GK, Darquenne C (2006) Importance of the bifurcation zone and branch orientation in simulated aerosol deposition in the alveolar zone of the human lung. J Aerosol Sci 37:37–62. https://doi.org/10.1016/j.jaerosci.2005.03.005
Henry FS, Butler JP, Tsuda A (2002) Kinematically irreversible acinar flow: a departure from classical dispersive aerosol transport theories. J Appl Physiol 92:835–845. https://doi.org/10.1152/japplphysiol.00385.2001
Hofemeier P, Sznitman J (2014) Role of alveolar topology on acinar flows and convective mixing. J Biomech Eng-Trans ASME 136:10. https://doi.org/10.1115/1.4027328
Hofemeier P, Sznitman J (2015) Revisiting pulmonary acinar particle transport: convection, sedimentation, diffusion, and their interplay. J Appl Physiol 118:1375–1385. https://doi.org/10.1152/japplphysiol.01117.2014
Hofemeier P, Fishler R, Sznitman J (2014) The role of respiratory flow asynchrony on convective mixing in the pulmonary acinus. Fluid Dyn Res 46:16. https://doi.org/10.1088/0169-5983/46/4/041407
Hogan B, Tata PR (2019) Cellular organization and biology of the respiratory system. Nat Cell Biol. https://doi.org/10.1038/s41556-019-0357-7
Huh D, Matthews BD, Mammoto A, Montoya-Zavala M, Hsin HY, Ingber DE (2010) Reconstituting organ-level lung functions on a chip. Science 328:1662–1668. https://doi.org/10.1126/science.1188302
Kim KH, Kabir E, Kabir S (2015) A review on the human health impact of airborne particulate matter. Environ Int 74:136–143. https://doi.org/10.1016/j.envint.2014.10.005
Kleinstreuer C, Zhang Z, Donohue JF (2008) Targeted drug-aerosol delivery in the human respiratory system. Annu Rev Biomed Eng 10:195–220. https://doi.org/10.1146/annurev.bioeng.10.061807.160544
Kumar H, Tawhai MH, Hoffman EA, Lin C-L (2009) The effects of geometry on airflow in the acinar region of the human lung. J Biomech 42:1635–1642. https://doi.org/10.1016/j.jbiomech.2009.04.046
Lemmon EW, Huber ML, McLinden MO (2010) NIST standard reference database 23: reference fluid thermodynamic and transport properties—REFPROP, 9th.0 edn. NIST, Gaithersburg
Levitzky MG (2017) A LANGE medical book: pulmonary physiology. LANGE physiology series, 9th edn. McGraw-Hill Medical, New York
Ma BS, Darquenne C (2011) Aerosol deposition characteristics in distal acinar airways under cyclic breathing conditions. J Appl Physiol 110:1271–1282. https://doi.org/10.1152/japplphysiol.00735.2010
Ma BS, Darquenne C (2012) Aerosol bolus dispersion in acinar airways-influence of gravity and airway asymmetry. J Appl Physiol 113:442–450. https://doi.org/10.1152/japplphysiol.01549.2011
Ma BS, Ruwet V, Corieri P, Theunissen R, Riethmuller M, Darquenne C (2009) CFD simulation and experimental validation of fluid flow and particle transport in a model of alveolated airways. J Aerosol Sci 40:403–414. https://doi.org/10.1016/j.jaerosci.2009.01.002
Oakes JM, Hofemeier P, Vignon-Clementel IE, Sznitman J (2016) Aerosols in healthy and emphysematous in silico pulmonary acinar rat models. J Biomech 49:2213–2220. https://doi.org/10.1016/j.jbiomech.2015.11.026
Ochs M et al (2004) The number of alveoli in the human lung. Am J Respir Crit Care Med 169:120–124. https://doi.org/10.1164/rccm.200308-11070C
Ostrovski Y, Hofemeier P, Sznitman J (2016) Augmenting regional and targeted delivery in the pulmonary acinus using magnetic particles. Int J Nanomed 11:3385–3395. https://doi.org/10.2147/ijn.s102138
Park SE, Georgescu A, Huh D (2019) Organoids-on-a-chip. Science 364:960–965. https://doi.org/10.1126/science.aaw7894
Srinath LS (2009) Advanced mechanics of solids, 3rd edn. Tata McGraw-Hill, New Delhi
Swan AJ, Clark AR, Tawhai MH (2012) A computational model of the topographic distribution of ventilation in healthy human lungs. J Theor Biol 300:222–231. https://doi.org/10.1016/j.jtbi.2012.01.042
Sznitman J (2013) Respiratory microflows in the pulmonary acinus. J Biomech 46:284–298. https://doi.org/10.1016/j.jbiomech.2012.10.028
Sznitman J, Heimsch F, Heimsch T, Rusch D, Rosgen T (2007) Three-dimensional convective alveolar flow induced by rhythmic breathing motion of the pulmonary acinus. J Biomech Eng-Trans ASME 129:658–665. https://doi.org/10.1115/1.2768109
Sznitman J, Heimsch T, Wildhaber JH, Tsuda A, Rosgen T (2009) Respiratory flow phenomena and gravitational deposition in a three-dimensional space-filling model of the pulmonary acinar tree. J Biomech Eng-Trans ASME 131:16. https://doi.org/10.1115/1.3049481
Tenenbaum-Katan J, Fishler R, Rothen-Rutishauser B, Sznitman J (2015) Biomimetics of fetal alveolar flow phenomena using microfluidics. Biomicrofluidics 9:13. https://doi.org/10.1063/1.4908269
Tippe A, Tsuda A (2000) Recirculating flow in an expanding alveolar model: experimental evidence of flow-induced mixing of aerosols in the pulmonary acinus. J Aerosol Sci 31:979–986. https://doi.org/10.1016/s0021-8502(99)00572-8
Tortora GJ, Derrickson B (2014) Principles of anatomy and physiology, 14th edn. John Wiley & Sons, Hoboken
Tsuda A, Henry FS, Butler JP (1995) Chaotic mixing of alveolated duct flow in rhythmically expanding pulmonary acinus. J Appl Physiol 79:1055–1063. https://doi.org/10.1152/jappl.1995.79.3.1055
Tsuda A, Henry FS, Butler JP (2008) Gas and aerosol mixing in the acinus. Respir Physiol Neuro 163:139–149. https://doi.org/10.1016/j.resp.2008.02.010
van Ertbruggen C, Corieri P, Theunissen R, Riethmuller ML, Darquenne C (2008) Validation of CFD predictions of flow in a 3D alveolated bend with experimental data. J Biomech 41:399–405. https://doi.org/10.1016/j.jbiomech.2007.08.013
Weibel ER, Sapoval B, Filoche M (2005) Design of peripheral airways for efficient gas exchange. Respir Physiol Neuro 148:3–21. https://doi.org/10.1016/j.resp.2005.03.005
Whitsett JA, Alenghat T (2015) Respiratory epithelial cells orchestrate pulmonary innate immunity. Nat Immunol 16:27–35. https://doi.org/10.1038/ni.3045
Zhang TZ, Orton NP, Mackie TR, Paliwal BR (2004) Technical note: a novel boundary condition using contact elements for finite element based deformable image registration. Med Phys 31:2412–2415. https://doi.org/10.1118/1.1774131
Acknowledgements
This research was supported by National Natural Science Foundation of China under Grant number of 11772112, and Science, Technology and Innovation Commission of Shenzhen Municipality under Grant numbers of KQJSCX20170329111827540 and JCYJ20170413105329648. Y. Zhu also acknowledges the financial support from HIT via the special start-up program.
Author information
Authors and Affiliations
Contributions
JD: investigation, software, formal analysis, data curation, and writing—original draft. YY: formal analysis, writing—review and editing. YZ: conceptualization, methodology, resources, writing—review and editing, and supervision.
Corresponding author
Ethics declarations
Conflict of interest
No conflicts of interest, financial or otherwise, are declared by the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Appendices
Appendix 1 Volume of intersection between a cylinder and a sphere
Monte Carlo method
We create a cube whose center locates in the coordinate origin in the cartesian coordinate system. There is an inscribed sphere inside the cube and this spherical diameter is same with alveolar diameter. To derive the intersection volume Va∩D, we randomly generate a large number of coordinate points that are only inside the cube first. Then Va∩D can be calculated by counting the percentage of points which locate in the intersection body. To simplify the process, Va∩D is only calculated once at the FRC condition. The domain of the intersection volume can be expressed by the equations below:
where Ra is the alveolar radius; Rd is the ductal radius; t is the thickness of the ductal wall; h is the distance between the spherical center and the outer surface of the alveolar duct along the line connecting spherical center and ductal center. If there are totally X and Xin coordinate points inside the cube and the intersection volume respectively, then the volume Va∩D is given by
Double integral method
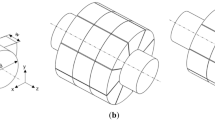
As shown in Fig.
Schematic diagram. a A section along the axis of the duct through the center of the spherical alveolus. b Cross section of the duct through the center of the spherical alveolus. c Section of an inscribed sphere inside a cube. Va∩D is the intersection volume between the alveolar duct and the alveolus at a typical generation under the FRC condition; Ra is the alveolar radius; Rd is the ductal radius; t is the thickness of the ductal wall; h is the distance between the spherical center and the outer surface of the alveolar duct along the line connecting spherical center and ductal center
7b, the alveolus and duct in the cartesian coordinate system are defined by
where m = Rd + h + t. The intersection points in yz plane between alveolus and duct is
The intersection volume is derived by double integral as
where f(z) is derived by integration by part,
Then a numerical integration, named composite Simpson’s rule, is applied to approximate integral for f(z),
where n is an even number of subintervals between z1 and z2; h = (z2 − z1)/n is the spacing of one subinterval.
Rights and permissions
About this article
Cite this article
Dong, J., Yang, Y. & Zhu, Y. New insight into air flow distribution in alveoli based on air- and saline-filled lungs. Microfluid Nanofluid 24, 71 (2020). https://doi.org/10.1007/s10404-020-02377-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10404-020-02377-9