Abstract
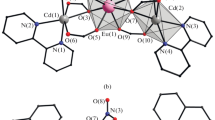
The reactions of cadmium(II) and europium(III) pentafluorobenzoates ([Cd(Pfbz)\(\left( {{{{\text{H}}}_{{\text{2}}}}{\text{O}}} \right)_{{\text{4}}}^{ + }\)}n ∙ n(Pfbz)–] (I) and [Eu2(Pfbz)6(H2O)8] ∙ 2H2O (II), respectively; Pfbz is pentafluorobenzoic acid anion) with the N-donor ligands (pyridine (Py), 2-phenylpyridine (Phpy), and 3-ethynylpyridine (Etypy)) afford a series of new Cd–Ln complexes with Pfbz– anions and Py or its substituted analogues: [Cd2Eu2(Pfbz)10(Py)4] (III), [Cd2Eu2(Pfbz)10(Phpy)2(MeCN)2] · 5MeCN (IV), and [CdEu2(Pfbz)8(Etypy)(H2O)2]n ∙ 3nMeCN ∙ n(Etypy) (VI). Similar reactions of compounds I and II with 2,4-lutidine (Lut), isoquinoline (Iquin), 2,2'-biquinoline (Biquin), and 7,8-benzoquinoline (Bquin) give the following cadmium complexes as the major products of crystallization from the reaction solutions: [Cd(Pfbz)2(Lut)]n (V), [Cd(H2O)(Pfbz)2(Iquin)2]n (VII), [Cd(Рfbz)2(Biquin)] (VIII), and \([{\text{Cd}}{{({\text{Pfbz}})}_{{\text{3}}}}]_{n}^{-}\) ∙ n(HBquin)+ (IX), respectively. The X-ray diffraction data (CIF files CCDC nos. 1987805 (I), 1987808 (III), 1987829 (IV), 1987817 (V), 1987822 (VI), 1987823 (VII), 1987824 (VIII), and 1987826 (IX)) are examples of the formation of new unusual coordination polymers.







Similar content being viewed by others
REFERENCES
Niekerk, J.N., van Schoening, F.R.L., and Talbot, J.H., Acta Crystallogr., 1953, vol. 6, p. 720.
Niekerk, J.N., van Schoening, F.R.L., Acta Crystallogr., 1953, vol. 6, p. 609.
Downie, T.C., Harrison, W., Raper, E.S., and Hepworth, M.A., Acta Crystallogr., Sect. B: Struct. Crystallogr. Cryst. Chem., 1971, vol. 27, p. 706.
Harrison, W. and Trotter, J., J. Chem. Soc., Dalton Trans., 1972, p. 956.
Amma, E.L., Griffith, E.A.H., Charles, N.G., and Rodesiler, P.F., Abstr. Papers Am. Chem. Soc. (Summer), 1983, p. 39.
Rodesiler, P.F., Griffith, E.A.H., Charles, N.G., and Amma, E.L., Acta Crystallogr., Sect. C: Cryst. Struct. Commun., 1985, vol. 41, p. 673.
Xiang-Hu Huang, Peng-Zhi Hong, and Wen-Dong Song, Acta Crystallogr.,Sect. E: Cryst. Struct. Online, 2008, vol. 64, p. m175.
Golubichnaya, M.A., Sidorov, A.A., Fomina, I.G., et al., Russ. J. Inorg. Chem., 1999, vol. 44, p. 1479.
Burley, J.C. and Prior, T.J., Acta Crystallogr.,Sect. E: Cryst. Struct. Online, 2005, vol. 61, p. m1422.
Karmakar, A. and Baruah, J.B., Polyhedron, 2008, vol. 27, p. 3409.
Dey, D., Roy, S., Purkayastha, R.N.D., et al., J. Coord. Chem., 2011, vol. 64, p. 1165.
Karmakar, A., Sarma, R.J., and Baruah, J.B., Eur. J. Inorg. Chem., 2006, p. 4673.
Eremenko, I.L., Nefedov, S.E., and Sidorov, A.A., Ino-rg. Chem., 1999, vol. 38, p. 3764.
Saxena, P. and Thirupathi, N., Polyhedron, 2015, vol. 98, p. 238.
Gao Zhu-Qing, Li Hong-Jin, and Gu Jin-Zh, Wuji Huaxue Xuebao (Chin.) (Chin. J. Inorg. Chem.), 2004, vol. 30, p. 2803.
Stoll, I., Brodbeck, R., Neumann, B., et al., CrystEngComm, 2009, vol. 11, p. 309.
Collings, J.C., Roscoe, K.P., Robins, E.G., et al., New J. Chem., 2002, vol. 26, p. 1740.
Imai, Y., Kawaguchi, K., Sato, T., et al., Mol. Cryst. Liq. Cryst., 2008, vol. 487, p. 153.
Williams, J.H., Cockcroft, J.K., and Fitch, A.N., Angew. Chem Int. Ed., 1992, vol. 31, p. 1655.
Cockcroft, J.K., Ghosh, R.E., Shephard, J.J., et al., CrystEngComm, 2017, vol. 19, p. 1019.
Kong, Y.-J., Li, P., Han, L.J., et al., Acta Crystallogr.,Sect. C: Cryst. Chem., 2017, vol. 73, p. 424.
Maas, H., Currao, A., and Calzaferri, G., Angew. Chem., Int. Ed. Engl., 2002, vol. 41, p. 2495.
Goldberg, A., Kiskin, M., Shalygina, O., et al., Chem. Asian J., 2016, vol. 11, p. 604.
SMART (control) and SAINT (integration). Software. Version 5.0, Madison: Bruker AXS Inc., 1997.
Sheldrick, G.M., SADABS, Madison: Bruker AXS Inc., 1997
Sheldrick, G.M., Acta Crystallogr.,Sect. C: Cryst. Chem., 2015, vol. 71, p. 3.
Dolomanov, O.V., Bourhis, L.J., Gildea, R.J., et al., J. Appl. Crystallogr., 2009, vol. 42, p. 339.
Kabsch, W., Acta Crystallogr., Sect. D: Biol. Crystallogr., 2010, vol. 66, p. 125.
Alvarez, S. and Llunell, M., Dalton Trans., 2000, vol. 19, p. 3288.
Casanova, D., Llunell, M., Alemany, P., and Alvarez, S., Chem.-Eur. J., 2005, vol. 11, p. 1479.
Chen Sheng-Chun, Zhang Zhi-Hui, Huang Kun-Lin, et al., CrystEngComm, 2013, vol. 15, p. 9613.
Nadzhafov, G.N., Shnulin, A.N., Mamedov, Kh.S., and Shil’nikov, V.I., Koord. Khim., 1982, vol. 8, p. 1276.
Glinskaya, L.A., Leonova, T.G., Klevtsova, R.F., and Larionov, S.V., J. Struct. Chem., 2010, vol. 51, p. 610.
Kalyakina, A.S., Utochnikova, V.V., Bushmarinov, I.S., et al., Chem.-Eur. J., 2015, vol. 21, p. 17921.
Chi Yu-Xian, Niu Shu-Yun, Jing Jin, et al., Dalton Trans, 2009, no. 37, p. 7653.
Wu Bin and Guo Yong-Sheng, Acta Crystallogr., Sect. E: Struct. Rep. Online, 2004, vol. 60, p. m1261.
Kiraev, S.R., Nikolaevskii, S.A., Kiskin, M.A., et al., Inorg. Chim. Acta, 2018, vol. 477, p. 15.
Boyle, T.J., Raymond, R., Boye, D.M., et al., Dalton Trans., 2010, vol. 39, p. 8050.
Gogoleva, N.V., Shmelev, M.A., Kiskin, M.A., et al., Russ.Chem. Bull., 2016, vol. 65, p. 1198.
Gogoleva, N.V., Shmelev, M.A., Evstifeev, I.S., et al., Izv. Akad. Nauk, Ser. Khim., 2016, no. 1, p. 181.
Sato, Y., Ouchi, A., Yukawa, Y., and Takeuchi, T., Chem. Lett., 1982, p. 1495.
Miklovic, J., Packova, A., Segla, P., et al., Inorg. Chim. Acta, 2015, vol. 429, p. 73.
Ivanikova, R., Boca, R., Dlhan, L., et al., Polyhedron, 2006, vol. 25, p. 3261.
Clegg, W. and Straughan, B.P., Acta Crystallogr., Sect. C: Cryst. Struct. Commun., 1992, vol. 45, p. 1989.
Hudak, J., Boca, R., Moncol, J., and Titis, J., Inorg. Chim. Acta, 2013, vol. 394, p. 401.
Niedzielska, D., Pazderski, L., Wojtczak, A., et al., Polyhedron, 2018, vol. 139, p. 155.
Robertson, K.N., Bakshi, P.K., Lantos, S.D., et al., Can. J. Chem., 1998, vol. 76, p. 583.
Ha Kwang-Ho, Z. Kristallogr., 2012, vol. 227, p. 33.
Esteruelas, M.A., Fernandez-Alvarez, F.J., Olivan, M., and Onate, E., Organometallics, 2009, vol. 28, p. 2276.
ACKNOWLEDGMENTS
The XRD analyses (complexes I and II), IR spectroscopy, and elemental analyses (of all complexes) were carried out on the equipment of the Center for Collective Use “Physical Methods of Investigation” at the Kurnakov Institute of General and Inorganic Chemistry (Russian Academy of Sciences) in terms of the state assignment of the Kurnakov Institute of General and Inorganic Chemistry (Russian Academy of Sciences) in the area of basic research. The data on the structure of compound V were obtained on the X-ray beam of the Belok station of the Kurchatov Synchrotron Radiation Source at the Kurchatov Institute Russian Research Center.
Funding
Complexes I, III, and IV were synthesized and studied in terms of the Russian Science Foundation (project no. 16-13-10537), complexes V and VII–IX were synthesized and studied in terms of the state assignment of the Kurnakov Institute of General and Inorganic Chemistry (Russian Academy of Sciences) in the area of basic research, and the synthesis and study of complex VI were supported by the Russian Foundation for Basic Research (project no. 18-29-04043).
Author information
Authors and Affiliations
Corresponding author
Additional information
The coauthors congratulate Academician I.L. Eremenko on his 70th jubilee
Translated by E. Yablonskaya
Rights and permissions
About this article
Cite this article
Shmelev, M.A., Gogoleva, N.V., Kuznetsova, G.N. et al. Cd(II) and Cd(II)–Eu(III) Complexes with Pentafluorobenzoic Acid Anions and N-Donor Ligands: Synthesis and Structures. Russ J Coord Chem 46, 557–572 (2020). https://doi.org/10.1134/S1070328420080060
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070328420080060