Abstract
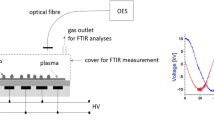
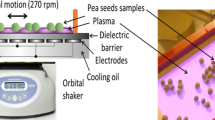
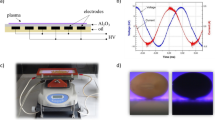
Cold atmospheric pressure plasma treatment is currently being explored as an alternative way to improve the germination and growing parameters of plant seeds. However, it is important to pay attention to the effect of plasma treatment on DNA damage of the seeds as well as detailed characteristics of plasma composition and parameters. The aim of this work was to study the DNA damage of plasma-treated pea seeds (Pisum sativum L.) and plasma parameters such as the chemical composition of plasma gaseous compounds and plasma radiation. Seeds were treated with plasma using the diffuse coplanar surface barrier discharge generated in different working gases (ambient air, nitrogen, oxygen and different mixtures of oxygen and nitrogen) at atmospheric pressure and at 60 s, 180 s and 300 s exposure times. DNA damage was studied using the single cell-gel electrophoresis called the comet assay and the plasma parameters were investigated by Fourier transform infrared spectroscopy and optical emission spectroscopy. Experiments in different ratios of oxygen and nitrogen were realized in order to understand the reaction mechanism between the ambient air plasma and the treated seeds. Based on our results, ambient air plasma appears to be the most advantageous for the plasma treatment due to no significant DNA damage because of the proper combination of plasma composition in combination with water vapor present in ambient air.










Similar content being viewed by others
References
Surowsky B, Schlüter O, Knorr D (2015) Interactions of non-thermal atmospheric pressure plasma with solid and liquid food systems: a review. Food Eng Rev 7:82–108. https://doi.org/10.1007/s12393-014-9088-5
Tendero C, Tixier C, Tristant P et al (2006) Atmospheric pressure plasmas: a review. Spectrochim Acta B 61:2–30. https://doi.org/10.1016/j.sab.2005.10.003
Adamovich I, Baalrud SD, Bogaerts A et al (2017) The 2017 Plasma Roadmap: low temperature plasma science and technology. J Phys D Appl Phys 50:323001. https://doi.org/10.1088/1361-6463/aa76f5
Homola T, Matoušek J, Kormunda M et al (2013) Plasma treatment of glass surfaces using diffuse coplanar surface barrier discharge in ambient air. Plasma Chem Plasma Process 33:881–894. https://doi.org/10.1007/s11090-013-9467-3
Johnsen K, Redford K (1996) Modification of polyolefin surfaces by plasma-induced grafting. J Appl Polym Sci 59:1651–1657. https://doi.org/10.1002/(SICI)1097-4628(19960307)59:10%3c1651:AID-APP17%3e3.0.CO;2-Z
Homola T, Matoušek J, Kormunda M et al (2012) Activation of poly(ethylene terephthalate) surfaces by atmospheric pressure plasma. Polym Degrad Stab 97:2249–2254. https://doi.org/10.1016/j.polymdegradstab.2012.08.001
Prysiazhnyi V, Cernak M (2012) Air plasma treatment of copper sheets using diffuse coplanar surface barrier discharge. Thin Solid Films 520:6561–6565. https://doi.org/10.1016/j.tsf.2012.06.069
Akram M, Jansen KMB, Ernst LJ, Bhowmik S (2011) Atmospheric pressure plasma surface modification of titanium for high temperature adhesive bonding. Int J Adhes Adhes 31:598–604. https://doi.org/10.1016/j.ijadhadh.2011.05.009
Laroussi M (2005) Low temperature plasma-based sterilization: overview and state-of-the-art. Plasma Process Polym 2:391–400. https://doi.org/10.1002/ppap.200400078
Lerouge S, Wertheimer MR, Yahia LH (2001) Plasma sterilization : a review of parameters, mechanisms, and limitations. Plasmas Polym 6:175–188. https://doi.org/10.1023/A:1013196629791
Moisan M, Barbeau J, Crevier M et al (2002) Plasma sterilization. Methods and mechanisms. Pure Appl Chem 74:349–358. https://doi.org/10.1351/pac200274030349
Fridman G, Friedman G, Gutsol A et al (2008) Applied plasma medicine. Plasma Process Polym 5:503–533. https://doi.org/10.1002/ppap.200700154
Morfill GE, Kong MG, Zimmermann JL (2009) Focus on plasma medicine. New J Phys 11:115011. https://doi.org/10.1088/1367-2630/11/11/115011
Randeniya LK, De Groot GJJB (2015) Non-thermal plasma treatment of agricultural seeds for stimulation of germination, removal of surface contamination and other benefits: a review. Plasma Process Polym 12:608–623. https://doi.org/10.1002/ppap.201500042
Hertwig C, Reineke K, Ehlbeck J et al (2015) Decontamination of whole black pepper using different cold atmospheric pressure plasma applications. Food Control 55:221–229. https://doi.org/10.1016/j.foodcont.2015.03.003
Hertwig C, Leslie A, Meneses N et al (2016) Inactivation of Salmonella Enteritidis PT30 on the surface of unpeeled almonds by cold plasma. Innov Food Sci Emerg Technol 44:242–248. https://doi.org/10.1016/j.ifset.2017.02.007
Măgureanu M, Sîrbu R, Dobrin D, Gîdea M (2018) Stimulation of the germination and early growth of tomato seeds by non-thermal plasma. Plasma Chem Plasma Process 38:989–1001. https://doi.org/10.1007/s11090-018-9916-0
Stolárik T, Henselová M, Martinka M et al (2015) Effect of low-temperature plasma on the structure of seeds, growth and metabolism of endogenous phytohormones in pea (Pisum sativum L.). Plasma Chem Plasma Process 35:659–676. https://doi.org/10.1007/s11090-015-9627-8
Zahoranová A, Hoppanová L, Šimončicová J et al (2018) Effect of cold atmospheric pressure plasma on maize seeds: enhancement of seedlings growth and surface microorganisms inactivation. Plasma Chem Plasma Process 38:969–988. https://doi.org/10.1007/s11090-018-9913-3
Zahoranová A, Henselová M, Hudecová D et al (2016) Effect of cold atmospheric pressure plasma on the wheat seedlings vigor and on the inactivation of microorganisms on the seeds surface. Plasma Chem Plasma Process 36:397–414. https://doi.org/10.1007/s11090-015-9684-z
Lotfy K (2017) Effects of cold atmospheric plasma jet treatment on the seed germination and enhancement growth of watermelon. Open J Appl Sci 07:705–719. https://doi.org/10.4236/ojapps.2017.712050
Misra NN, Pankaj SK, Segat A, Ishikawa K (2016) Cold plasma interactions with enzymes in foods and model systems. Trends Food Sci Technol 55:39–47. https://doi.org/10.1016/j.tifs.2016.07.001
Dobrynin D, Fridman G, Friedman G, Fridman A (2009) Physical and biological mechanisms of direct plasma interaction with living tissue. New J Phys 11:115020. https://doi.org/10.1088/1367-2630/11/11/115020
Kyzek S, Holubová Ľ, Medvecká V et al (2018) Cold atmospheric pressure plasma can induce adaptive response in pea seeds. Plasma Chem Plasma Process 39:475–486. https://doi.org/10.1007/s11090-018-9951-x
Bradley JW, Bryant PM (2007) The diagnosis of plasmas used in the processing of textiles and other materials. Plasma Technol Text. https://doi.org/10.1533/9781845692575.1.25
Förster S, Mohr C, Viöl W (2005) Investigations of an atmospheric pressure plasma jet by optical emission spectroscopy. Surf Coatings Technol 200:827–830. https://doi.org/10.1016/j.surfcoat.2005.02.217
Machala Z, Janda M, Hensel K et al (2007) Emission spectroscopy of atmospheric pressure plasmas for bio-medical and environmental applications. J Mol Spectrosc 243:194–201. https://doi.org/10.1016/j.jms.2007.03.001
Rodero A, García MC (2017) Gas temperature determination of non-thermal atmospheric plasmas from the collisional broadening of argon atomic emission lines. J Quant Spectrosc Radiat Transf 198:93–103. https://doi.org/10.1016/j.jqsrt.2017.05.004
Pacheco M, Pacheco J, Moreno H, Mercado A, Valdivia R, Santana A (2008) OES analysis in a nonthermal plasma used for toxic gas removal: rotational and excitation temperature estimation. Laser Phys 18:303–307. https://doi.org/10.1134/S1054660X08030183
Ono R (2016) Optical diagnostics of reactive species in atmospheric-pressure nonthermal plasma. J Phys D Appl Phys 49:83001. https://doi.org/10.1088/0022-3727/49/8/083001
Sivachandiran L, Khacef A (2017) Enhanced seed germination and plant growth by atmospheric pressure cold air plasma: combined effect of seed and water treatment. RSC Adv 7:1822. https://doi.org/10.1039/c6ra24762h
Schiorlin M, Paradisi C, Brandenburg R et al (2015) Pollutant degradation in gas streams by means of non-thermal plasmas. In: Nejadkoorki F (ed) Current air quality issues. IntechOpen. https://doi.org/10.5772/60049
Reuter S, Sousa JS, Stancu GD, Hubertus Van Helden JP (2015) Review on VUV to MIR absorption spectroscopy of atmospheric pressure plasma jets. Plasma Sources Sci Technol 24:054001. https://doi.org/10.1088/0963-0252/24/5/054001
Černák M, Černáková L, Hudec I et al (2009) Diffuse coplanar surface barrier discharge and its applications for in-line processing of low-added-value materials. Eur Phys J Appl Phys 47:22806. https://doi.org/10.1051/epjap/2009131
Hartmann A, Agurell E, Beevers C et al (2003) Recommendations for conducting the in vivo alkaline comet assay. Mutagenesis 18:45–51. https://doi.org/10.1093/mutage/18.1.45
Collins AR (2004) The comet assay for DNA damage and repair: principles, applications, and limitations. Mol Biotechnol 26:249–261. https://doi.org/10.1385/MB:26:3:249
Gichner T, Patková Z, Száková J et al (2008) DNA damage in potato plants induced by cadmium, ethyl methanesulphonate and γ-rays. Environ Exp Bot 62:113–119. https://doi.org/10.1016/j.envexpbot.2007.07.013
Gordon IE, Rothman LS, Hill C et al (2017) The HITRAN2016 molecular spectroscopic database. J Quant Spectrosc Radiat Transf 203:3–69. https://doi.org/10.1016/j.jqsrt.2017.06.038
Abdelaziz AA, Ishijima T, Osawa N, Seto T (2019) Quantitative analysis of ozone and nitrogen oxides produced by a low power miniaturized surface dielectric barrier discharge: effect of oxygen content and humidity level. Plasma Chem Plasma Process 39:165–185. https://doi.org/10.1007/s11090-018-9942-y
Wang H, Zhuang Z, Sun C et al (2016) Numerical evaluation of the effectiveness of NO2 and N2O5 generation during the NO ozonation process. J Environ Sci (China) 41:51–58. https://doi.org/10.1016/j.jes.2015.05.015
Kuwahata H, Mikami I (2014) Generation of nitric acid and nitrous acid in distilled water irradiated with atmospheric-pressure plasma jet. e-J Surf Sci Nanotechnol 12:410–413. https://doi.org/10.1380/ejssnt.2014.410
Tomeková J, Medvecká V, Hergelová B et al (2018) Spectroscopic study of diffuse coplanar surface barrier discharge plasma used for treatment of pea seeds. In: Šafránková J, Pavlů J (eds) WDS’18 Proceedings of contributed papers—physics. Matfyzpress, Prague, pp 124–129
Gill SS, Anjum NA, Gill R et al (2015) DNA damage and repair in plants under ultraviolet and ionizing radiations. Sci World J 2015:5–7. https://doi.org/10.1155/2015/250158
Pournavab RF, Mejía EB, Mendoza AB et al (2019) Ultraviolet radiation effect on seed germination and seedling growth of common species from northeastern Mexico. Agronomy 9:269. https://doi.org/10.3390/agronomy9060269
Švubová R, Kyzek S, Medvecká V et al (2020) Novel insight at the effect of cold atmospheric pressure plasma on the activity of enzymes essential for the germination of pea (Pisum sativum L. cv. Prophet) seeds. Plasma Chem Plasma Process. https://doi.org/10.1007/s11090-020-10089-9
Acknowledgements
This work was supported by the Slovak Research and Development Agency under the Contract No. APVV-16-0216, Project VEGA 1/0410/18 and the University Grant UK/366/2019.
Author information
Authors and Affiliations
Contributions
JT: diagnostics of plasma—Fourier transform infrared spectroscopy, optical emission spectroscopy: data measurements and analysis, writing—first draft preparation; SK: DNA damage analysis, writing—first draft preparation; VM: plasma treatment of seeds, writing—review and editing; EG: DNA damage analysis, writing—review and editing; AZ: plasma diagnostics, writing—first draft preparation, review and editing.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tomeková, J., Kyzek, S., Medvecká, V. et al. Influence of Cold Atmospheric Pressure Plasma on Pea Seeds: DNA Damage of Seedlings and Optical Diagnostics of Plasma. Plasma Chem Plasma Process 40, 1571–1584 (2020). https://doi.org/10.1007/s11090-020-10109-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11090-020-10109-8