Abstract
Key Message
Psoralen synthase and angelicin synthase responsible for the formation of psoralen and angelicin in Peucedanum praeruptorum Dunn were identified and functionally characterized, respectively.
Abstract
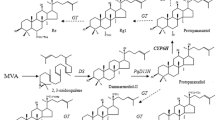
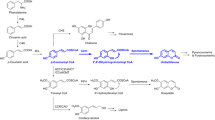
Furanocoumarins were reported to possess several activities such as anticancer, anti-inflammatory and neuroprotective, and function as phytotoxin and allelochemical in plants. Furanocoumarins are the main bioactive ingredient in P. praeruptorum which is a commonly used traditional Chinese medicine. Phenylalanine ammonia lyase (PAL), 4-coumarate: CoA ligase (4CL), p-coumaroyl CoA 2’-hyfroxylase (C2’H) were cloned previously to elucidate the biosynthetic mechanism of coumarin lactone ring. However, the genes involved in complex coumarins in P. praeruptorum have not been explored. Herein, putative psoralen synthase CYP71AJ49 and angelicin synthase CYP71AJ51 were cloned from P. praeruptorum. In vivo and in vitro yeast assays were conducted to confirm their activities. Furthermore, the results of High Performance Liquid Chromatography–Electrospray Ionization Mass Spectrometry (HPLC–ESI–MS) verified that CYP71AJ49 catalyzed the conversion of marmesin to psoralen, and CYP71AJ51 catalyzed columbianetin to angelicin. Subsequently, the expression profile showed that CYP71AJ49 and CYP71AJ51 were easily affected by environmental conditions, especially UV and temperature. The genes tissue-specific expression and compounds tissue-specific distribution pattern indicated the existence of substance transport in P. praeruptorum. Phylogenetic analysis was conducted with 27 CYP71AJs, CYP71AJ49 and CYP71AJ51 were classified in I-4 and I-2, respectively. These results provide further insight to understand the biosynthetic mechanism of complex coumarins.






Similar content being viewed by others
References
Bouimeja B, El Hidan MA, Touloun O, Ait Laaradia M, Ait Dra L, El Khoudri N, Chait A, Boumezzough A (2018) Anti-scorpion venom activity of Thapsia garganica methanolic extract: Histopathological and biochemical evidences. J Ethnopharmacol 211:340–347. https://doi.org/10.1016/j.jep.2017.10.003
Bourgaud F, Hehn A, Larbat R, Doerper S, Gontier E, Kellner S, Matern U (2006) Biosynthesis of coumarins in plants: a major pathway still to be unravelled for cytochrome P450 enzymes. Phytochem Rev 5:293–308. https://doi.org/10.1007/s11101-006-9040-2
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Dueholm B, Krieger C, Drew D, Olry A, Kamo T, Taboureau O, Weitzel C, Bourgaud F, Hehn A, Simonsen HT (2015) Evolution of substrate recognition sites (SRSs) in cytochromes P450 from Apiaceae exemplified by the CYP71AJ subfamily. BMC Evol Biol 15:122. https://doi.org/10.1186/s12862-015-0396-z
Edgar RC (2004) MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 32:1792–1797. https://doi.org/10.1093/nar/gkh340
Hall BG (2013) Building phylogenetic trees from molecular data with MEGA. Mol Biol Evol 30:1229–1235. https://doi.org/10.1093/molbev/mst012
Hamberger B, Bak S (2013) Plant P450s as versatile drivers for evolution of species-specific chemical diversity. Philos Trans R Soc B 368:20120426. https://doi.org/10.1098/rstb.2012.0426
Hamerski D, Matern U (1988) Biosynthesis of psoralens Psoralen 5-monooxygenase activity from elicitor-treated Ammi majus cells. FEBS Lett 239:263–265. https://doi.org/10.1016/0014-5793(88)80930-X
Hamilton R, Watanabe C, de Boer H (1987) Compilation and comparison of the sequence around the ATG start codons in Saccharomyces cerevisiae mRNAs. Nucleic Acids Res 15:3581–3593. https://doi.org/10.1093/nar/15.8.3581
Hung W-L, Suh JH, Wang Y (2017) Chemistry and health effects of furanocoumarins in grapefruit. J Food Drug Anal 25:71–83. https://doi.org/10.1016/j.jfda.2016.11.008
Karamat F, Olry A, Munakata R, Koeduka T, Sugiyama A, Paris C, Hehn A, Bourgaud F, Yazaki K (2014) A coumarin-specific prenyltransferase catalyzes the crucial biosynthetic reaction for furanocoumarin formation in parsley. Plant J 77:627–638. https://doi.org/10.1111/tpj.12409
Kviesis J, Klimenkovs I, Arbidans L, Podjava A, Klavins M, Liepins E (2019) Evaluation of furanocoumarins from seeds of the wild parsnip (Pastinaca sativa L. s.l.). J Chromatogr B 1105:54–66. https://doi.org/10.1016/j.jchromb.2018.12.012
Larbat R, Kellner S, Specker S, Hehn A, Gontier E, Hans J, Bourgaud F, Matern U (2007) Molecular cloning and functional characterization of psoralen synthase, the first committed monooxygenase of furanocoumarin biosynthesis. J Biol Chem 282:542–554. https://doi.org/10.1074/jbc.M604762200
Larbat R, Hehn A, Hans J, Schneider S, Jugde H, Schneider B, Matern U, Bourgaud F (2009) Isolation and functional characterization of CYP71AJ4 encoding for the first P450 monooxygenase of angular furanocoumarin biosynthesis. J Biol Chem 284:4776–4785. https://doi.org/10.1074/jbc.M807351200
Li J, Chen X, Zhong L, Wang X, Zhou X, Tang Y, Liu Y, Zheng H, Zhan R, Chen L (2019) Comparative iTRAQ-based proteomic analysis provides insight into a complex regulatory network of Pogostemon cablin in response to exogenous MeJA and Ethrel. Ind Crop Prod 140:111661. https://doi.org/10.1016/j.indcrop.2019.111661
Ling-Yi K, Yi L, Zhi-Da M, Xian L, Ting-Ru Z (1996) Coumarins from Peucedanum praeruptorum. Phytochemistry 41:1423–1426. https://doi.org/10.1016/0031-9422(95)00783-0
Liu T, Yao R, Zhao Y, Xu S, Huang C, Luo J, Kong L (2017) Cloning, functional characterization and site-directed mutagenesis of 4-coumarate: coenzyme A Ligase (4CL) involved in Coumarin biosynthesis in Peucedanum praeruptorum. Dunn Front Plant Sci 8:4. https://doi.org/10.3389/fpls.2017.00004
Masuda T, Takasugi M, Anetai M (1998) Psoralen and other linear furanocoumarins as phytoalexins in Glehnia littoralis. Phytochemistry 47:13–16. https://doi.org/10.1016/S0031-9422(97)00528-1
Munakata R, Olry A, Karamat F, Courdavault V, Sugiyama A, Date Y, Krieger C, Silie P, Foureau E, Papon N, Grosjean J, Yazaki K, Bourgaud F, Hehn A (2016) Molecular evolution of parsnip (Pastinaca sativa) membrane-bound prenyltransferases for linear and/or angular furanocoumarin biosynthesis. New Phytol 211:332–344. https://doi.org/10.1111/nph.13899
Omura T, Sato JoBC (1964) The carbon monoxide-binding pigment of liver microsomes I. Evidence for its hemoprotein nature. J Biol Chem 239:2370–2378
Parast BM, Chetri SK, Sharma K, Agrawal V (2011) In vitro isolation, elicitation of psoralen in callus cultures of Psoralea corylifolia and cloning of psoralen synthase gene. Plant Physiol Biochem 49:1138–1146. https://doi.org/10.1016/j.plaphy.2011.03.017
Pathak MA, Fitzpatrick TB (1992) The evolution of photochemotherapy with psoralens and UVA (PUVA): 2000 BC to 1992 AD. J Photochem Photobiol B 14:3–22. https://doi.org/10.1016/1011-1344(92)85080-E
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29:e45–e45. https://doi.org/10.1093/nar/29.9.e45
Piao XL, Park IH, Baek SH, Kim HY, Park MK, Park JH (2004) Antioxidative activity of furanocoumarins isolated from Angelicae dahuricae. J Ethnopharmacol 93:243–246. https://doi.org/10.1016/j.jep.2004.03.054
Raju R, Singh A, Reddell P, Münch G (2018) Anti-inflammatory activity of prenyl and geranyloxy furanocoumarins from Citrus garrawayi (Rutaceae). Phytochem Lett 27:197–202. https://doi.org/10.1016/j.phytol.2018.07.030
Richard EG (2020) The science and (lost) art of Psoralen plus UVA phototherapy. Dermatol Clin 38:11–23. https://doi.org/10.1016/j.det.2019.08.002
Singh T, Yadav R, Agrawal V (2020) Effective protocol for isolation and marked enhancement of psoralen, daidzein and genistein in the cotyledon callus cultures of Cullen corylifolium (L.) Medik. Ind Crop Prod 143:111905. https://doi.org/10.1016/j.indcrop.2019.111905
Song Y, Jing W, Yan R, Wang Y (2015) Research progress of the studies on the roots of Peucedanum praeruptorum dunn (Peucedani radix). Pak J Pharm Sci 28:71–81. https://doi.org/10.1254/jphs.13237FP
Sui Z, Luo J, Yao R, Huang C, Zhao Y, Kong L (2019) Functional characterization and correlation analysis of phenylalanine ammonia-lyase (PAL) in coumarin biosynthesis from Peucedanum praeruptorum Dunn. Phytochemistry 158:35–45. https://doi.org/10.1016/j.phytochem.2018.11.006
Truan G, Cullin C, Reisdorf P, Urban P, Pompon D (1993) Enhanced in vivo monooxygenase activities of mammalian P450s in engineered yeast cells producing high levels of NADPH-P450 reductase and human cytochrome b5. Gene 125:49–55. https://doi.org/10.1016/0378-1119(93)90744-N
Yang W-Q, Song Y-L, Zhu Z-X, Su C, Zhang X, Wang J, Shi S-P, Tu P-F (2015) Anti-inflammatory dimeric furanocoumarins from the roots of Angelica dahurica. Fitoterapia 105:187–193. https://doi.org/10.1016/j.fitote.2015.07.006
Yao R, Zhao Y, Liu T, Huang C, Xu S, Sui Z, Luo J, Kong L (2017) Identification and functional characterization of a p-coumaroyl CoA 2’-hydroxylase involved in the biosynthesis of coumarin skeleton from Peucedanum praeruptorum Dunn. Plant Mol Biol 95:199–213. https://doi.org/10.1007/s11103-017-0650-4
Zhao Y, Liu T, Zhang Q, Xu S, Han C, Xu J, Chen M, Chen Y, Kong L (2015) Integration of a decrescent transcriptome and metabolomics dataset of Peucedanum praeruptorum to investigate the CYP450 and MDR genes involved in coumarins biosynthesis and transport. Front Plant Sci 6:996. https://doi.org/10.3389/fpls.2015.00996
Zhao Y, Luo J, Xu S, Wang W, Liu T, Han C, Chen Y, Kong L (2016) Selection of reference genes for gene expression normalization in Peucedanum praeruptorum Dunn under abiotic stresses, hormone treatments and different tissues. PLoS ONE 11:e0152356. https://doi.org/10.1371/journal.pone.0152356
Acknowledgements
This project was funded by National Natural Science Foundation of China (81703637) and Natural Science Fund in Jiangsu Province (BK20170736). This research was also supported by the Program for Changjiang Scholars and Innovative Research Team in University (IRT_15R63) and the 111 Project from the Ministry of Education of China and the State Administration of Foreign Export Affairs of China (B18056).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jian, X., Zhao, Y., Wang, Z. et al. Two CYP71AJ enzymes function as psoralen synthase and angelicin synthase in the biosynthesis of furanocoumarins in Peucedanum praeruptorum Dunn. Plant Mol Biol 104, 327–337 (2020). https://doi.org/10.1007/s11103-020-01045-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-020-01045-4