Abstract
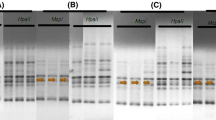
Plants employ DNA methylation to maintain genomic plasticity that allows rapid adaptation to environmental stresses without alteration nucleotide sequence of DNA. The present investigation was carried out to detect DNA methylation changes induced by drought stress in inbred line W22 by using amplified methylation-polymorphism technique. The maize inbred line W22 encountered extensive DNA methylation variations, including hypo- and hypermethylation as a response to drought stress. Differential methylation of many genes involved in plant response to drought stress such as Antagonist of Like Heterochromatin Protein1, peptide methionine sulfoxide reductase (MSRA4), 3-isopropylmalate dehydratase large subunit 2, adenylyl cyclase-associated protein, 3-hydroxyisobutyrate dehydrogenase gene, and others was observed.

Similar content being viewed by others
References
Bassam BJ, Gresshoff PM (2007) Silver staining DNA in polyacrylamide gels. Nat Protoc 2:2649–2654. https://doi.org/10.1038/nprot.2007.330
Bednarek PT, Orłowska R, Niedziela A (2017) A relative quantitative methylation-sensitive amplified polymorphism (MSAP) method for the analysis of abiotic stress. BMC Plant Biol 17:79. https://doi.org/10.1186/s12870-017-1028-0
Buffon G, Blasi ÉAR, Adamski JM et al (2016) Physiological and molecular alterations promoted by Schizotetranychus oryzae mite infestation in rice leaves. J Proteome Res 15:431–446. https://doi.org/10.1021/acs.jproteome.5b00729
Candaele J, Demuynck K, Mosoti D et al (2014) Differential methylation during maize leaf growth targets developmentally regulated genes. Plant Physiol 164:1350–1364. https://doi.org/10.1104/pp.113.233312
Chen Q, Tao S, Bi X et al (2013) Research of total levels on DNA methylation in plant based on HPLC analysis. Am J Mol Biol 03:98–101. https://doi.org/10.4236/ajmb.2013.32013
Chwialkowska K, Korotko U, Kosinska J et al (2017) Methylation sensitive amplification polymorphism sequencing (MSAP-Seq)—a method for high-throughput analysis of differentially methylated CCGG sites in plants with large genomes. Front Plant Sci 8:2056. https://doi.org/10.3389/fpls.2017.02056
Cullings KW (1992) Design and testing of a plant-specific PCR primer for ecological and evolutionary studies. Mol Ecol 1:233–240
Doyle JJ, Doyle JL (1987) A rapid DNA isolation procedure for small quantities of fresh leaf tissue
Eom S, Baek S-A, Kim J, Hyun T (2018) Transcriptome analysis in Chinese cabbage (Brassica rapa ssp. pekinensis) provides the role of glucosinolate metabolism in response to drought stress. Molecules 23:1186. https://doi.org/10.3390/molecules23051186
Erturk FA, Agar G, Arslan E, Nardemir G (2015) Analysis of genetic and epigenetic effects of maize seeds in response to heavy metal (Zn) stress. Environ Sci Pollut Res 22:10291–10297. https://doi.org/10.1007/s11356-014-3886-4
González L, González-Vilar M (2001) Determination of relative water content. In: Roger MJR (ed) Handbook of plant ecophysiology techniques. Springer, Berlin, pp 207–212
He X, Xie Y, Zhao Y et al (2018) Analysis of differentially methylated regions of genomic DNA in maize (Zea mays L.) exposed to salt stress. Caryologia 71:331–340. https://doi.org/10.1080/00087114.2018.1469817
Hildebrandt TM, Nunes Nesi A, Araújo WL, Braun H-P (2015) Amino acid catabolism in plants. Mol Plant 8:1563–1579. https://doi.org/10.1016/j.molp.2015.09.005
Huang T, Jander G (2017) Abscisic acid-regulated protein degradation causes osmotic stress-induced accumulation of branched-chain amino acids in Arabidopsis thaliana. Planta 246:737–747. https://doi.org/10.1007/s00425-017-2727-3
Jennings MP, Beacham IR (1989) MacConkey agar as an alternative to Xgal in the detection of recombinant plasmids. Biotechniques 7:1082
Kapazoglou A, Tsaftaris A (2011) Epigenetic chromatin regulators as mediators of abiotic stress responses in cereals. In: Shanker A (ed) Abiotic stress in plants—mechanisms and adaptations. InTech, Rijeka
Knill T, Reichelt M, Paetz C et al (2009) Arabidopsis thaliana encodes a bacterial-type heterodimeric isopropylmalate isomerase involved in both Leu biosynthesis and the Met chain elongation pathway of glucosinolate formation. Plant Mol Biol 71:227–239. https://doi.org/10.1007/s11103-009-9519-5
Korbie DJ, Mattick JS (2008) Touchdown PCR for increased specificity and sensitivity in PCR amplification. Nat Protoc 3:1452–1456. https://doi.org/10.1038/nprot.2008.133
Labra M, Grassi F, Imazio S et al (2004) Genetic and DNA-methylation changes induced by potassium dichromate in Brassica napus L. Chemosphere 54:1049–1058. https://doi.org/10.1016/j.chemosphere.2003.10.024
Le DT, Tarrago L, Watanabe Y et al (2013) Diversity of plant methionine sulfoxide reductases B and evolution of a form specific for free methionine sulfoxide. PLoS ONE 8:e65637. https://doi.org/10.1371/journal.pone.0065637
Lei M, Zhang H, Julian R et al (2015) Regulatory link between DNA methylation and active demethylation in Arabidopsis. Proc Natl Acad Sci 112:3553–3557. https://doi.org/10.1073/pnas.1502279112
Li Q, Gent JI, Zynda G et al (2015) RNA-directed DNA methylation enforces boundaries between heterochromatin and euchromatin in the maize genome. Proc Natl Acad Sci 112:14728–14733. https://doi.org/10.1073/pnas.1514680112
Liang SC, Hartwig B, Perera P et al (2015) Kicking against the PRCs—a domesticated transposase antagonises silencing mediated by polycomb group proteins and is an accessory component of polycomb repressive complex 2. PLoS Genet 11:e1005660. https://doi.org/10.1371/journal.pgen.1005660
Liu S, Lv Z, Liu Y et al (2018) Network analysis of ABA-dependent and ABA-independent drought responsive genes in Arabidopsis thaliana. Genet Mol Biol 41:624–637. https://doi.org/10.1590/1678-4685-gmb-2017-0229
Phutikanit N, Suwimonteerabutr J, Harrison D et al (2010) Different DNA methylation patterns detected by the amplified methylation polymorphism polymerase chain reaction (AMP PCR) technique among various cell types of bulls. Acta Vet Scand 52:18. https://doi.org/10.1186/1751-0147-52-18
Sanguinetti CJ, Dias EN, Simpson AJ (1994) Rapid silver staining and recovery of PCR products separated on polyacrylamide gels. Biotechniques 17:914–921
Saraswat S, Yadav AK, Sirohi P, Singh NK (2017) Role of epigenetics in crop improvement: water and heat stress. J Plant Biol 60:231–240. https://doi.org/10.1007/s12374-017-0053-8
Schertl P, Danne L, Braun H-P (2017) 3-Hydroxyisobutyrate dehydrogenase is involved in both, valine and isoleucine degradation in Arabidopsis thaliana. Plant Physiol 175:51–61. https://doi.org/10.1104/pp.17.00649
Schreiner M, Beyene B, Krumbein A, Stützel H (2009) Ontogenetic changes of 2-propenyl and 3-indolylmethyl glucosinolates in Brassica carinata leaves as affected by water supply. J Agric Food Chem 57:7259–7263. https://doi.org/10.1021/jf901076h
Schulz B, Eckstein RL, Durka W (2013) Scoring and analysis of methylation-sensitive amplification polymorphisms for epigenetic population studies. Mol Ecol Resour 13:642–653. https://doi.org/10.1111/1755-0998.12100
Shan X, Wang X, Yang G et al (2013) Analysis of the DNA methylation of maize (Zea mays L.) in response to cold stress based on methylation-sensitive amplified polymorphisms. J Plant Biol 56:32–38. https://doi.org/10.1007/s12374-012-0251-3
Tan M (2010) Analysis of DNA methylation of maize in response to osmotic and salt stress based on methylation-sensitive amplified polymorphism. Plant Physiol Biochem 48:21–26. https://doi.org/10.1016/j.plaphy.2009.10.005
Thomas L, Marondedze C, Ederli L et al (2013) Proteomic signatures implicate cAMP in light and temperature responses in Arabidopsis thaliana. J Proteomics 83:47–59. https://doi.org/10.1016/j.jprot.2013.02.032
Wang X, Cai X, Xu C et al (2016) Drought-responsive mechanisms in plant leaves revealed by proteomics. Int J Mol Sci 17:1706. https://doi.org/10.3390/ijms17101706
Yang C-M, Fan M-J, Hsiang W-M (1993) Growth and yield response of maize (Zea mays L.) to soil water deficits ii effects of water deficit timing and strength. J Agric Res China 42:173–186
Zhang X, Li Q, Kong L, Yu H (2017) DNA methylation changes detected by methylation-sensitive amplified polymorphism in the Pacific oyster (Crassostrea gigas) in response to salinity stress. Genes Genomics 39:1173–1181. https://doi.org/10.1007/s13258-017-0583-y
Zhao Y, Yu S, Ye W et al (2010) Study on DNA cytosine methylation of cotton (Gossypium hirsutum L.) genome and its implication for salt tolerance. Agric Sci China 9:783–791. https://doi.org/10.1016/S1671-2927(09)60155-3
Zhu J, Ding P, Li Q et al (2015) Molecular characterization and expression profile of methionine sulfoxide reductase gene family in maize (Zea mays) under abiotic stresses. Gene 562:159–168. https://doi.org/10.1016/j.gene.2015.02.066
Acknowledgements
The authors of this manuscript are grateful to the United States Department of Agriculture (USDA) for providing maize seeds used in this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Communicated by Nehal Sallam.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Table S1
Sequences of the primers used for Amp PCR (DOCX 19 kb)
Rights and permissions
About this article
Cite this article
Sallam, N., Moussa, M., Yacout, M. et al. Analysis of methylated genomic cytosines of maize inbred line W22 in response to drought stress. CEREAL RESEARCH COMMUNICATIONS 48, 459–465 (2020). https://doi.org/10.1007/s42976-020-00066-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42976-020-00066-5