Abstract
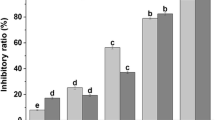
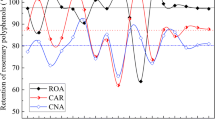
Rosemary extract (RE) has significant antioxidant and antibacterial properties; however, the application of RE to areas with an aqueous solution is limited due to its poor solubility. There is a need for research focused on finding a method to improve water solubility for incorporating RE into aqueous systems, such as food and cosmetic. Therefore, in this study, the micellar solubilization of RE is conducted using four types of surfactants (Tween 20, polyglyceryl-10-laurate, polyglyceryl-10-myristate, and polyglyceryl-10-monooleate) to increase the water solubility of RE and the effects of various surfactant types and concentration on solubility were investigated. Antibacterial activities of the mixture solutions containing RE and surfactants were also examined. The water solubility of RE significantly improved when surfactants were added into the RE solution and especially in polyglyceryl-10-monooleate, with the longest tail, was the most effective for increasing solubility. In terms of the antibacterial effect on Bacillus subtilis, it was observed that a relatively lower concentration of surfactants was effective. The results of this study provide useful information for the development of a new RE-loaded delivery system for food and cosmetic application.






Similar content being viewed by others
References
Adamczak M. Surfactants, polyelectrolytes and nanoparticles as building blocks for nanocarriers. PhD thesis, AGH University of Science and Technology, Krakow, Poland (2013)
Aguilar F, Autrup H, Barlow S, Castle L, Crebelli R, Engel K, Gontard N, Gott D, Grilli S. Use of rosemary extracts as a food additive-Scientific opinion of the panel on food additives, flavourings, processing aids and materials in contact with food. Eur. Food Safe. Auth. 6 (6): 1-29 (2008)
Ambriz-Pérez DL, Leyva-López N, Gutierrez-Grijalva EP, Heredia JB. Phenolic compounds: Natural alternative in inflammation treatment. A Review. Cogent Food Agric. 2 (1): 1131412 (2016)
Aschemann-Witzel J, Varela P, Peschel AO. Consumers’ categorization of food ingredients: Do consumers perceive them as ‘clean label’ producers expect? An exploration with projective mapping. Food Qual. Prefer. 71: 117-128 (2019)
Baeurle SA, Kroener J. Modeling effective interactions of micellar aggregates of ionic surfactants with the Gauss-core potential. J. Math. Chem. 36: 409-421 (2004)
Bazaka K, Jacob M, Chrzanowski W, Ostrikov K. Anti-bacterial surfaces: natural agents, mechanisms of action, and plasma surface modification. Int. J. Food Microbiol. 5: 48739-48759 (2015)
Bohren CF, Huffman DR. Absorption and scattering of light by small particles. Wiley, Hoboken, NJ, USA. pp. 130-157 (2008)
Chaiyasit W, Silvestre M, McClements DJ, Decker EA. Ability of surfactant hydrophobic tail group size to alter lipid oxidation in oil-in-water emulsions. J. Agr. Food Chem. 48: 3077-3080 (2000)
Gelardi G, Mantellato S, Marchon D, Palacios M, Eberhardt AB, Flatt RJ. 9 - Chemistry of chemical admixtures. Woodhead Publishing, pp. 149-218 (2016)
Holmberg K, JoÈnsson B, Kronberg B, Lindman B. Surfactant and Polymers in Aqueous Solution. 2nd ed. Wiley-Blackwell, Hoboken, NJ, USA. pp. 39-66 (2002)
Kumar S, Singh P. Various techniques for solubility enhancement: An overview. Pharm. Innov. J. 5: 23-28 (2016)
Lalas S, Dourtoglou V. Use of rosemary extract in preventing oxidation during deep-fat frying of potato chips. J. Am. Oil. Chem. Soc. 80: 579-583 (2003)
Lu GW, Gao P. Emulsions and microemulsions for topical and transdermal drug delivery. Elsevier, Amsterdam. pp. 59-94 (2010)
Mahmood ME, Al-Koofee DA. Effect of temperature changes on critical micelle concentration for tween series surfactant. Global J. Sci. Front. Res. 13: 1-8 (2013)
Matsumoto K, Igarashi Y, Shirai D, Hayashi K. Investigation of the influence of surfactant on the degree of supercooling (coexisting system of solid–liquid and gas–liquid interfaces). Int. J. Refrig. 36 (4): 1302-1309 (2013)
McClements DJ. Food emulsions: principles, practices, and techniques. CRC press, Boca Raton. (2015)
Mirgorodskaya AB, Mamedov VA, Zakharova LY, Valeeva FG, Mamedova VL, Galimullina VR, Kushnasarova RA, Sinyashin OG. Surfactant solutions for enhancing solubility of new arylquinolin-2-ones. J. Mol. Liq. 242: 732-738 (2017)
Pérez-Fons L, GarzÓn MaT, Micol V. Relationship between the antioxidant capacity and effect of rosemary (Rosmarinus officinalis L.) polyphenols on membrane phospholipid order. J. Agr. Food Chem. 58 (1): 161-171 (2009)
Rangel-Yagui CO, Hsu HWL, Pessoa-Jr A, Tavares LC. Micellar solubilization of ibuprofen: influence of surfactant head groups on the extent of solubilization. Rev. Bras. Cienc. Farm. 41: 237-246 (2005)
Robbins K, Sewalt V. Extending freshness with rosemary extract. Food Tech. 16: 8 (2005)
Savoia D. Plant-derived antimicrobial compounds: alternatives to antibiotics. Future Microbiol. 7: 979-990 (2012)
Silva DGd, Sarruf FD, Oliveira LCDd, Arêas EPG, Kaneko TM, Consiglieri VO, Velasco MVR, Baby AR. Influence of particle size on appearance and in vitro efficacy of sunscreens. Braz. J. Pharm. Sci. 49: 251-261 (2013)
Smith MC, Crist RM, Clogston JD, McNeil SE. Zeta potential: a case study of cationic, anionic, and neutral liposomes. Anal. Bioanal. Chem. 409: 5779-5787 (2017)
Stoyanova K, Vinarov Z, Tcholakova S. Improving Ibuprofen solubility by surfactant-facilitated self-assembly into mixed micelles. J. Drug Deliv. Sci. Tec. 36: 208-215 (2016)
Tehrani-Bagha AR, Holmberg K. Solubilization of hydrophobic dyes in surfactant solutions. Mater. 6: 580-608 (2013)
Urzúa A, Rezende MC, Mascayano C, Vásquez L. A structure-activity study of antibacterial diterpenoids. Molecules. 13: 882-891 (2008)
Vinarov Z, Dobreva P, Tcholakova S. Effect of surfactant molecular structure on progesterone solubilization. J. Drug Deliv. Sci. Tec. 43: 44-49 (2018)
Yoon S, Kim J, Jo B, Kim J, Lee S, Ahn B, Cho Y. Isolation and identification of antimicrobial compounds against Helicobacter pylori from Rosemary (Rosmarinus officinalis L.) extracts. J. Appl. Biol. Chem. 54 (3): 159-165 (2011)
Zhang Y, Smuts JP, Dodbiba E, Rangarajan R, Lang JC, Armstrong DW. Degradation study of carnosic acid, carnosol, rosmarinic acid, and rosemary extract (Rosmarinus officinalis L.) assessed using HPLC. J. Agr. Food Chem. 60: 9305-9314 (2012)
Acknowledgements
This research was supported by the National Research Foundation of Korea (NRF) grant funded by Korea government (MSIT) (No. 2019R1A2C1010708).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Park, S., Mun, S. & Kim, YR. Influences of added surfactants on the water solubility and antibacterial activity of rosemary extract. Food Sci Biotechnol 29, 1373–1380 (2020). https://doi.org/10.1007/s10068-020-00792-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10068-020-00792-w