Abstract
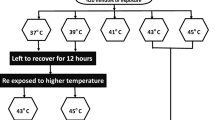
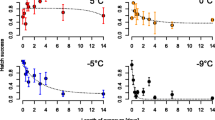
Temperature tolerance is an important factor limiting the distribution of arthropod vectors, with limited information on the ability of mosquitoes to evolve different temperature levels. Under laboratorial conditions effect of thermal stress in late third instar larvae of Aedes aegypti determined by subjecting them to temperature regimes such as 37 °C, 39 °C, 41 °C, 43 °C and 45 °C, and the lethal time required to inflict mortality of 50% (LT50) and 90% (LT90) were also computed. To understand the adaptive thermotolerance in immature stages of Ae.aegypti, late third instar larvae were exposed to 37 °C and 39 °C, and these larvae were further re-exposed to temperatures 43 °C and 45 °C respectively. The median mortality of the larvae exposed to sub-lethal temperatures at 43 °C and 45 °C was 140 and 30 min respectively. While the median mortality for the larvae adapted at 39 °C and exposed to 43 °C and 45 °C was 230 and 50 min. All the larvae exposed at 39 °C were survived up to 420 min whereas at 45 °C within 60 min of exposure time 100% mortality was observed. Larvae pre-adapted at 37 °C and 39 °C were re-exposed to 43 °C and 45 °C and it was observed that larvae pre-adapted at 39 °C were more thermotolerant than larvae pre-adapted at 37 °C. LT50 values were 82.9 and 213.7 min as compared to the non-adapted larvae. Similarly, the LT50 values were 30.5 min and in larvae pre-adapted at 39 °C when re-exposed to 45 °C showed 2 fold increases in thermotolerancein comparison to the same stage larvae kept at room temperature. Larvae pre-adapted from higher temperature showed more thermotolerance than the larvae pre-adapted at lower temperature.



Similar content being viewed by others
References
Alachiotis SN, Stephanou G (1982) Temperature adaptation of Drosophila populations. The heat shock protein system. Comp Biochem Physiol Mol Integr Physiol 73:529–533
Alto BW, Bettinardi D (2013) Temperature and dengue virus infection in mosquitoes: independent effects on the immature and adult stages. Am J Hyg 88:497–505
Andrewartha HG (1971) Introduction to the study of animal populations. The University of Chicago Press, Chicago, IL
Bar-Zeev M (1958) The effect of temperature on the growth rate and survival of the immature stages of Aedes aegypti (L). Bull Entomol Res 49:157–163
Bayoh MN, Lindsay SW (2004) Temperature-related duration of aquatic stages of the afro tropical malaria vector mosquito Anopheles gambiae in the laboratory. Med Vet Entomol 18:174–179
Benedict MQ, Cockburn AF, Seawright JA (1991) Heat shock mortality and induced thermotolerance in larvae of the mosquito. J Am Mosq Control Assoc 7:547–550
Berger E, Vitek M, Morgenelli CM (1984) Genetic reprogramming of Drosophila cells in culture by ecdysterone. In: Kurstak E, Oberlander H, Dubendorfer LA (eds) Invertebrate systems in vitro 11. Elsevier Press, Amsterdam
Briegel H, Timmermann SE (2001a) Aedes albopictus (Diptera: Culicidae): physiological aspects of development and reproduction. J Med Entomol 38:566–571
Briegel H, Timmermann SE (2001b) Aedes aegypti: size, reserves, survival, and flight potential. J Vector Ecol 26:210–231
Briegel H, Waltert A, Kuhn R (2001) Reproductive physiology of Aedes (Aedimorphus) vexans (Diptera: Culicidae). In: relation to flight potential. J Med Entomol 38:557–565
Busvine JR (1971) A critical review of the techniques for testing insecticides. The Commonwealth Agricultural Bureaux, Farnham Royal, Slough
Chang LH, Hsu EL, Teng HJ, Ho CM (2007) Differential survival of Aedes aegypti and Aedes albopictus (Diptera: Culicidae) larvae exposed to low temperatures. In: Taiwan. J Med Entomol 44:205–210
Christophers SR (1960) Aedes aegypti (L): the yellow fever mosquito: its life history, bionomics and structure. Cambridge University Press, Cambridge
Delatte H, Gimonneau G, Triboire A, Fontenille D (2009) Influence of temperature on immature development, survival, longevity, fecundity, and gonotrophic cycles of Aedes albopictus, vector of chikungunya and dengue in the Indian Ocean. J Med Entomol 46:33–34
Feder JN, Tsuchihashi Z, Irrinki A, Lee VK, Mapa FA, Morikang E, Prass CE, Starnes SM, Wolff RK, Parkkila S, Sly WS, Schatzman RC (1997) The hemochromatosis founder mutation in HLA-H disrupts beta 2-microglobulin interaction and cell surface expression. J Biol Chem 272:14025–14028
Githeko AK, Lindsay SW, Confalonieri UE, Patz JA (2000) Climate change and vector borne diseases: a regional analysis. Bull World Health Organ 78:1136–1146
Gray KM, Burkett-Cadena ND, Eubanks MD, Unnasch TR (2011) Crepuscular flight activity of Culex erraticus (Diptera: Culicidae). J Med Entomol 48:167–172
Gunathilaka N, Ranathunge T, Udayanga L, Abeyewickreme W (2017) Efficacy of blood sources and artificial blood feeding methods in rearing of Aedes aegypti (Diptera: Culicidae) for sterile insect technique and incompatible insect technique approaches in Sri Lanka. Biomed Res Int 2017:1–7. https://doi.org/10.1155/2017/3196924
Joshi DS (1996) Effect of fluctuating and constant temperatures on development, adult longevity and fecundity in the mosquito Aedes kroembini. J Therm Biol 21:151–154
Kartick C, Bharathi GSJ, Surya P, Anwesh M, Arun S, Muruganandam N, Avijit R, Vijayachari P (2017) Outbreak investigation of fever mimicking dengue in Havelock Island, an important tourist destination in the Andaman & Nicobar Archipelago, 2014. Epidemiol Infect 145:1437–1442
Kramer LD, Hardy JL, Presser SB (1983) Effect of temperature of extrinsic incubation on the vector competence of Culex tarsalis for western equine encephalomyelitis virus. Am J Trop Med Hyg 32:1130–1139
Lactin DJ, Holliday NJ, Johnson DL, Craigen R (1995) Improved rate model of temperature-dependent development by arthropods. Environ Pollut 24:68–75
Mahroof R, Yan Zhu K, Neven L, Subramanyam B, Bai J (2005) Expression patterns of three heat shock protein 70 genes among developmental stages of the red four beetle, Triboliumcastaneum (Coleoptera: Tenebrionidae). Comp Biochem Physiol A Mol Integr Physiol 141:247–256
Manimunda SP, Singh SS, Sugunan AP, Singh O, Roy S, Shriram AN et al (2007) Chikungunya fever, Andaman and Nicobar Islands, India. Emerg Infect Dis 13:1259–1260
Martens MJ, Nissen LW, Rothmans J, Jetten TH, McMichael AJ (1995) Potential impact of global climate change on malaria risk. Environ Health Perspect 103:458–464
Meyer RP, Hardy JL, Reisen WK (1990) Diel changes in adult mosquito microhabitat temperatures and their relationship to the extrinsic incubation of arbovirues in mosquitoes in Kern County, California. J Med Entomol 27:607–614
Mourya DT, Yadav P, Mishra AC (2004) Effect of temperature stress on immature stages and susceptibility of Aedes aegypti mosquitoes to chikungunya virus. Am J Trop Med Hyg 70:346–350
Muturi EJ, Alto BW (2011) Larval environmental temperature and insecticide exposure alters Aedes aegypti competence for arboviruses. Vector Borne Zoonotic Dis 11:1157–1163
NVBDCP (2020) Distribution of dengue/DHF in India. https://nvbdcp.gov.in/index4.php?lang=1&level=0&linkid=431&lid=3715
Paaijmans KP, Heusinkveld BG, Jacobs AF (2008) A simplified model to predict diurnal water temperature dynamics in a shallow tropical water pool. Int J Biometeorol 52:797–803
Richardson K, Hoffmann AA, Johnson P, Ritchie SA, Kearney MR (2011) Thermal sensitivity of Aedes aegypti from Australia: empirical data and prediction of effects on distribution. J Med Entomol 48:914–924
Rinehart JP, Hayward SA, Elnitsky MA, Sandro LH, Lee RE Jr, Denlinger DL (2006) Continuous up-regulation of heat shock proteins in larvae, but not adults, of a polar insect. Proc Natl Acad Sci U S A 103:14223–14227
Rinehart JP, Robich RM, Denlinger DL (2006) Enhanced cold and desiccation tolerance in diapausing adults of Culex pipiens, and a role for Hsp70 in response to cold shock but not as a component of the diapauses program. J Med Entomol 43:713–722
Robich RM, Rinehart JP, Kitchen LJ, Denlinger DL (2007) Diapause-specific gene expression in the northern house mosquito, Culex pipiens L, identified by suppressive subtractive hybridization. J Insect Physiol 53:235–245
Rowley WA, Graham CL (1968) The effect of temperature and relative humidity on the flight performance of female Aedes aegypti. J Insect Physiol 14:1251–1257
Rueda LM, Patel KJ, Axtell RC, Stinner RE (1990) Temperature-dependent development and survival rates of Culex quinquefasciatus and Aedes aegypti (Diptera: Culicidae). J Med Entomol 27:892–898
Shriram AN, Sehgal SC (1999) Aedes aegypti (L) in Port Blair, Andaman and Nicobar Islands-distribution and larval ecology. J Commun Dis 31:185–192
Shriram AN, Sivan A, Sugunan AP (2018) Spatial distribution of Aedes aegypti and Aedes albopictus in relation to geo-ecological features in South Andaman, Andaman and Nicobar Islands, India. Bull Entomol Res 108:166–174
Shriram AN, Sugunan AP, Manimunda SP, Vijayachari P (2009) Community-centred approach for the control of AedesSpp in a peri-urban zone in the Andaman and Nicobar Islands using Temephos. Natl Med J India 22:116–120
Shriram AN, Sugunan AP, Vijayachari P (2008) Infiltration of Aedes aegypti in to peri-urban areas in south Andaman. Indian J Med Res127: 618–620
Sivan A, Shriram AN, Muruganandam N, Thamizhmani R (2017) Expression of heat shock proteins (HSPs) in Aedes aegypti(L) and Aedes albopictus(Skuse) (Diptera: Culicidae) larvae in response to thermal stress. Acta Trop 167:121–127
Sivan A, Shriram AN, Sugunan AP, Anwesh M, Muruganandam N, Kartik C, Vijayachari P (2016) Natural transmission of dengue virus serotype 3 by Aedes albopictus(Skuse) during an outbreak in Havelock Island: entomological characteristics. Acta Trop 156:122–129. https://doi.org/10.1016/j.actatropica.2016.01.015
Sivan A, Shriram AN, Vanamail P, Sugunan AP (2019) Impact of temperature variant on survival of Aedes albopictus Skuse (Diptera: Culicidae): implications on Thermotolerance and acclimation. Neotrop Entomol 48:561–571. https://doi.org/10.1007/s13744-019-00680-x Epub 2019 Apr 11
Sorensen JG, Dahlgaard J, Loeschcke V (2001) Genetic variation in thermal tolerance among natural populations of Drosophila buzzatii: down regulation of Hsp70 expression and variation in heat stress resistance traits. Funct Ecol 15:289–296
Stephanou G, Alahiotis SN, Christodoulou C, Marmaras VJ (1983) Adaptation of Drosophila to temperature: heat shock proteins and survival in Drosophila melanogaster. Dev Genet 3:299–308
Stephanou G, Alahiotis SN, Marmaras VJ, Christodoulou C (1983) Heat shock response in Ceratitiscapitata. Comp Biochem Physiol 74:425–432
Tun-Lin W, Burkot TR, Kay BH (2000) Effects of temperature and larval diet on development rates and survival of the dengue vector Aedes aegypti in North Queensland, Australia. Med Vet Entomol 14:31–37
Van der Have TM (2008), Slaves to the Eyring equation? Temperature dependence of life-history characters in developing ectotherms. Ph.D. dissertation, Wageningen University, Wageningen
Vijayachari P, Singh SS, Sugunan AP, Shriram AN, Manimunda SP, Bharadwaj AP et al (2011) Emergence of dengue in Andaman and Nicobar archipelago: eco-epidemiological perspective. Indian J Med Res 134:235–237
WHO (1996) In: AJ MM et al (eds) Climate change and human health: an assessment/prepared by a Task Group on behalf of the World Health Organization, the World Meteorological Association and the United Nations Environment Programme. World Health Organisation, Geneva Web: https://apps.who.int/iris/handle/10665/62989. Accessed July 2019
Winokur OC, Main BJ, Nicholson J (2020 Mar 18) Barker CM (2020) impact of temperature on the extrinsic incubation period of Zika virus in Aedes aegypti. PLoS Negl Trop Dis 14(3):e0008047. https://doi.org/10.1371/journal.pntd.0008047
Wright RE, Knight KL (1966) Effect of environmental factors on biting activity of Aedes vexans (Meigen) and Aede strivittatus (Coquillett). Mosq News 26:565–578
Wu X, Lu Y, Zhou S, Chen L, Xu B (2016) Impact of climate change on human infectious diseases: empirical evidence and human adaptation. Environ Int 86:14–23
Yadav P, Barde PV, Gokhale MD, Vipat V, Mishra AC, Pal JK et al (2005) Effect of temperature and insecticide stresses on Aedes aegypti larvae and their influence on the susceptibility of mosquitoes to dengue-2 virus. Southeast Asian J Trop Med Public Health 36:1139–1144
Zhao L, Becnel JJ, Clark GG, Linthicum KJ (2010) Expression of AeaHsp26 and AeaHsp83 in Aedes aegypti (Diptera: Culicidae) larvae and pupae in response to heat shock stress. J Med Entomol 47:367–375
Zhao L, Pridgeon JW, Becnel JJ, Clark GG, Linthicum KJ (2009) Identification of genes differentially expressed during heat shock treatment in Aedes aegypti. J Med Entomol 46:490–4965
Acknowledgements
The authors are grateful to Dr. P. Vijayachari, The Director, ICMR-Regional Medical Research Centre, Port Blair for extending all the facilities for the conduct of the study. Authors acknowledge the suggestions given by Dr. K. Raghavendra, Scientist “G” at the ICMR-National Institute of Malaria Research, New Delhi. Technical Assistance rendered by the staff of the Division of Medical Entomology and Vector Borne Diseases is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
We do not have any conflict of interest whatsoever.
Ethical approval
The study did not involve human or animals; therefore, ethical approval was not sought.
Ethical approval retrospective studies
Authors assure that we had checked with our respective institutions to make sure we are complying with the specific requirements of our country.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sivan, A., Shriram, A.N., Vanamail, P. et al. Thermotolerance and acclimation in the immature stages of Aedes aegypti (L) (Diptera: Culicidae) to simulated thermal stress. Int J Trop Insect Sci 41, 333–344 (2021). https://doi.org/10.1007/s42690-020-00211-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42690-020-00211-x