Abstract
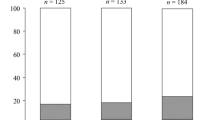
Females of the hymenopteran parasitoid Habrobracon hebetor show two types of photoperiodic response: development under short day conditions results in an increase of the adult size (quantitative response) and in the induction of reproductive diapause (qualitative response). In a study conducted with individuals from a laboratory population, the progeny of each female was divided into two groups, which developed under the short (10 h) and long (18 h) day conditions. Females significantly differed both in the mean body size of the progeny and in the proportion of diapausing individuals. The qualitative and quantitative responses interact: short day caused an increase in size only in diapausing individuals. In addition, family analysis revealed significant positive correlations between the proportion of diapausing individuals (a manifestation of the qualitative response) and the mean size of the progeny (a manifestation of the quantitative response). The results of the study support the hypothesis on the unity of the mechanism of the qualitative and quantitative photoperiodic responses.





Similar content being viewed by others
REFERENCES
Ahmed, U.A., Shi, Z.H., Guo, Y.L., Zou, X.F., Hao, Z.P., and Pang, S.T., Maternal photoperiod effect on and geographic variation of diapause incidence in Cotesia plutellae (Hymenoptera: Braconidae) from China, Appl. Entomol. Zool., 2007, vol. 42, no. 3, p. 383. https://doi.org/10.1303/aez.2007.383
Amadou, L., Ba, M.N., Baoua, I., and Muniappan, R., Timing of releases of the parasitoid Habrobracon hebetor and numbers needed in augmentative biological control against the millet head miner Heliocheilus albipunctella, BioControl, 2019, vol. 64, no. 5, p. 573. https://doi.org/10.1007/s10526-019-09960-2
Askari Seyahooei, M., Bagheri, A., Bavaghar, M., Dousti, A.F., and Parichehreh, S., Mating and carbohydrate feeding impacts on life-history traits of Habrobracon hebetor (Hymenoptera: Braconidae), J. Econom. Entomol., 2018, vol. 111, no. 6, p. 2605. https://doi.org/10.1093/jee/toy253
Bradshaw, W.E. and Lounibos, L.P., Photoperiodic control of development in the pitcher-plant mosquito, Wyeomyia smithii,Canad. J. Zool., 1972, vol. 50, no. 6, p. 713.
Chen, H., Zhang, H., Zhu, K.Y., and Throne, J.E., Induction of reproductive diapause in Habrobracon hebetor (Hymenoptera: Braconidae) when reared at different photoperiods at low temperatures, Environ. Entomol., 2012, vol. 41, no. 3, p. 697. https://doi.org/10.1603/EN11311
Colinet, H., Muratori, F., and Hance, T., Cold-induced expression of diapause in Praon volucre: fitness cost and morpho-physiological characterization, Physiol. Entomol., 2010, vol. 35, no. 4, p. 301. https://doi.org/10.1111/j.1365-3032.2010.00743.x
Danilevsky, A.S., Photoperiodism i sezonnoe razvitie nasekomykh (Photoperiodism and seasonal development of insects), Leningrad: Nauka, 1961.
Danks, H.V., The elements of seasonal adaptations in insects, Can. Entomol., 2007, vol. 139, no. 1, p. 1.
Denlinger, D.L., Induction and termination of pupal diapause in Sarcophaga (Diptera: Sarcophagidae), Biol. Bull., 1972, vol. 142, no. 1, p. 11.
Denlinger, D.L., Regulation of diapause, Ann. Rev. Entomol., 2002, vol. 47, p. 93. https://doi.org/10.1146/annurev.ento.47.091201.145137
Geispitz, K.F., Glinjanaja, E.I., Dubynina, T.S., Kvitko, N.V., Pidzhakova, T.V., Sapozhnikova, F.D., Simonenko, N.P., and Taranets, M.N., Annual endogenous rhythm of changes in photoperiodic reaction of arthropods and its connection with exogenous factors, Entomol. Obozr., 1978, vol. 57, no. 4, p. 731.
Goto, S.G., Roles of circadian clock genes in insect photoperiodism, Entomol. Sci., 2013, vol. 16, no. 1, p. 1. https://doi.org/10.1111/ens.12000
Hao, Z.P., Zhao, Y.Q., Yuan, Z.Q., and Shi, Z.H., Effects of photoperiod on body size and hormone titres relating to diapause regulation in Cotesia vestalis (Hymenoptera: Braconidae), Can. Entomol., 2013, vol. 145, no. 4, p. 369. https://doi.org/10.4039/tce.2013.6
Honĕk, A., Regulation of diapause, number of instars, and body growth in the moth species Amathes c-nigrum (Lepidoptera: Noctuidae), Entomol. Generalis, 1979, vol. 5, no. 3, p. 221.
Ismail, S. and Fuzeau-Braesch, S., Programmation de la diapause chez Gryllus campestris, J. Insect Physiol., 1976, vol. 22, no. 1, p. 133.
Kabore, A., Ba, N.M., Dabire-Binso, C., and Sanon, A., Towards development of a parasitoid cottage industry of the parasitoid wasp Habrobracon hebetor (Say): optimum rearing and releases conditions for successful biological control of the millet head miner Heliocheilus albipunctella (De Joannis) in the Sahel, Int. J. Trop. Insect Sci., 2019, vol. 39, no. 1, p. 25. https://doi.org/10.1007/s42690-019-00005-w
Kamm, J.A., Photoperiodic regulation of growth in an insect: response to progressive changes in daylength, J. Insect Physiol., 1972, vol. 18, no. 9, p. 1745.
Koštál, V., Insect photoperiodic calendar and circadian clock: independence, cooperation, or unity?, J. Insect Physiol., 2011, vol. 57, no. 5, p. 538. https://doi.org/10.1016/j.jinsphys.2010.10.006
Mbata, G.N. and Warsi, S., Habrobracon hebetor and Pteromalus cerealellae as tools in post-harvest integrated pest management, Insects, 2019, vol. 10, p. 85. https://doi.org/10.3390/insects10040085
Mousseau, T.A. and Dingle, H., Maternal effects in insect life histories, Annu. Rev. Entomol., 1991, vol. 36, p. 511. https://www.annualreviews.org/doi/pdf/10.1146/annurev.en.36.010191.002455
Nakamura, K., Effect of photoperiod on the size–temperature relationship in a pentatomid bug, Dolycoris baccarum,J. Therm. Biol., 2002, vol. 27, no. 6, p. 541. https://doi.org/10.1016/S0306-4565(02)00028-1
Nealis, V.G., Oliver, D., and Tchir, D., The diapause response to photoperiod in Ontario populations of Cotesia melanoscela (Ratzeburg) (Hymenoptera: Braconidae), Can. Entomol., 1996, vol. 128, no. 1, p. 41.
Noor-ul-Ane, M., Mirhosseini, M.A., Crickmore, N., Saeed, S., Noor, I., and Zalucki, M.P., Temperature-dependent development of Helicoverpa armigera (Hübner) (Lepidoptera: Noctuidae) and its larval parasitoid, Habrobracon hebetor (Say) (Hymenoptera: Braconidae): implications for species interactions, Bull. Entomol. Res., 2018, vol. 108, no. 3, p. 295. https://doi.org/10.1017/S0007485317000724
Nylin, S. and Gotthard, K., Plasticity in life-history traits, Annu. Rev. Entomol., 1998, vol. 43, p. 63.
Nylin, S., Wickman, P.E.R.O., and Wiklund, C., Life-cycle regulation and life history plasticity in the speckled wood butterfly: are reaction norms predictable?, Biol. J. Linn. Soc., 1995, vol. 55, no. 2, p. 143.
Reznik, S.Ya., Vaghina, N.P., and Voinovich, N.D., Variations in the tendency to diapause among successive generations of laboratory strains of Trichogramma species (Hymenoptera, Trichogrammatidae): Endogenous or exogenous?, Zool. Zhurn., 2015a, vol. 94, no. 4, p. 446.
Reznik, S.Ya., Ovchinnikov, A.N., Dolgovskaya, M.Yu., and Belyakova, N.A., Intraspecific variation in photoperiodic effect on the rate of preimaginal development in Harmonia axyridis (Pallas) (Coleoptera, Coccinellidae), Entomol. Obozr., 2015b, vol. 94, no. 1, p. 35.
Salminen, T.S., Vesala, L., and Hoikkala, A., Photoperiodic regulation of life-history traits before and after eclosion: Egg-to-adult development time, juvenile body mass and reproductive diapause in Drosophila montana,J. Insect Physiol., 2012, vol. 58, no. 12, p. 1541. https://doi.org/10.1016/j.jinsphys.2012.09.007
Saulich, A.Kh. and Musolin, D.L., Diapause in the seasonal cycle of stink bugs (Heteroptera, Pentatomidae) from the temperate zone, Entomol. Obozr., 2011, vol. 90, no. 4, p. 740.
Saulich, A.Kh. and Musolin, D.L., Biologiya i ecologiya pereponchatokrylykh parazitoidov. Uchebno-metodicheskoe posobie k kursu “Sezonnye tsikly nasekomykh” dlya studentov magistratury na kafedre entomologii (Biology and ecology of hymenopteran parasitiods (Hymenoptera: Apocrita: Parasitica): Teaching aid to the lecture course “Seasonal cycles of insects” for master students of the Entomology chair), St. Petersburg: Izd. SPbGU, 2013.
Saulich, A.Kh. and Volkovich, T.A., Ekologiya fotoperiodizma nasekomykh (Ecology of photoperiodism in insects), St. Petersburg: Izd. SPbGU, 2004.
Saunders, D.S., Unity and diversity in the insect photoperiodic mechanism, Entomol. Sci., 2011, vol. 14, no. 3, p. 235. https://doi.org/10.1111/j.1479-8298.2011.00463.x
Saunders, D.S., Dormancy, diapause, and the role of the circadian system in insect photoperiodism, Annu. Rev. Entomol., 2020, vol. 65, p. 373. https://doi.org/10.1146/annurev-ento-011019-025116
Saunders, D.S., Steel, C.G.H., Vafopoulou, X., and Lewis, R.D., Insect Clocks, Amsterdam: Elsevier, 2002.
Solà, M., Castañé, C., Lucas, E., and Riudavets, J., Optimization of a banker box system to rear and release the parasitoid Habrobracon hebetor (Hymenoptera: Braconidae) for the control of stored-product moths, J. Econom. Entomol., 2018, vol. 111, no. 5, p. 2461. https://doi.org/10.1093/jee/toy219
Tauber, M.J., Tauber, C.A., and Masaki, S., Seasonal adaptations of insects, New York: Oxford University Press, 1986.
Tougeron, K., Diapause research in insects: historical review and recent work perspectives, Entomol. Exp. Appl., 2019, vol. 167, no. 1, p. 27. https://doi.org/10.1111/eea.12753
Tyshchenko, V.P., A bioscillatory model of the physiological mechanism of insect photoperiodic reaction, Zh. Obshch. Biol., 1966, vol. 27, no. 2, p. 209.
Tyshchenko, V.P., Fiziologiya fotoperiodizma nasekomykh (Physiology of insect photoperiodism), Lenidgrad: Nauka, 1984.
Tyshchenko, V.P. and Gasanov, O.G., Comparative investigation of the photoperiodic regulation of diapause and pupal weight in several moth species (Lepidoptera), Zool. Zh., 1983, vol. 62, no. 1, p. 63.
Tyshchenko, V.P., Lanevich, V.P., and Gasanov, O.G., Correlations of quantitative and qualitative manifestations of photoperiodism in the cabbage moth (Barathra brassicae), Zh. Obshch. Biol., 1977, vol. 38, no. 2, p. 264.
Vinogradova, E.B. and Bogdanova, T.P., Endogenous cyclic changes in the tendency to diapause in a continuous strain of flesh flies developing at constant conditions, Entomol. Obozr., 1980, vol. 59, no. 1, p. 26.
Voinovich, N.D., Reznik, S.Ya., and Vaghina, N.P., Variations in the “spontaneous” dynamics of the tendency to diapause among generations of Trichogramma telengai Sor. (Hymenoptera, Trichogrammatidae), Entomol. Obozr., 2013, vol. 92, no. 3, p. 465.
Weseloh, R.M., Termination and induction of diapause in the gypsy moth larval parasitoid, Apanteles melanoscelus,J. Insect Physiol., 1973, vol. 19, no. 10, p. 2025.
Zaslavski, V.A., Fotoperiodicheskii i temperaturnyi kontrol’ razvitiya nasekomykh (Photoperiodic and temperature control of insect development), Leningrad: Nauka, 1984.
Zaslavski, V.A., The diversity of environmental factors controlling insect seasonal development and the possible unity of underlying physiological mechanism, Entomol. Obozr., 1996, vol. 75, no. 2, p. 233.
Zaslavski, V.A. and Umarova, T.Ya., Environmental and endogenous control of diapause in Trichogramma species, Entomophaga, 1990, vol. 35, no. 1, p. 23.
Zinovyeva, K.B., The influence of host hemolymph feeding on the photoperiodic response of Bracon hebetor (Hymenoptera, Braconidae), Zool. Zh., 1992, vol. 71, no. 9, p. 153.
ACKNOWLEDGMENTS
We are deeply grateful to O.I. Spirina (Zoological Institute RAS) for assistance in conducting experiments.
Funding
The study was performed within the frame of the state research project AAAA-A19-119020690082-8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest. All applicable international, national, and institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Rights and permissions
About this article
Cite this article
Reznik, S.Y., Samartsev, K.G. & Dolgovskaya, M.Y. Intrapopulation Variability of the Qualitative and Quantitative Photoperiodic Responses in Habrobracon hebetor (Say) (Hymenoptera, Braconidae). Entmol. Rev. 100, 277–286 (2020). https://doi.org/10.1134/S001387382003001X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S001387382003001X