Abstract
Purpose
Intracranial atherosclerosis disease (ICAD) is an essential cause of stroke. The characteristics of effective treatment include low periprocedural risk and a sustained treatment effect. Angioplasty with a conventional balloon for ICAD is safe but has a dauntingly high restenosis rate. Drug-coated balloon (DCB) angioplasty might reduce the risk of restenosis while maintaining the overall safety of the procedure.
Methods
This study included symptomatic ICAD patients with more than 70% stenosis. Intermediate catheters were placed distally, and the lesions were predilated with a conventional balloon, followed by a DCB (SeQuent Please, B Braun, Melsungen, Germany). The primary endpoint was any stroke or death within 30 days or ipsilateral ischemic stroke thereafter. The secondary endpoint was arterial restenosis of more than 50% during follow-up.
Results
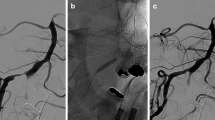
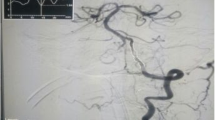
A total of 39 sessions of DCB angioplasty were performed for 39 lesions in 35 patients between October 2015 and April 2018 in a single center. All of the DCBs could be navigated to the lesions. Major periprocedural complications were noted in two patients (5.7%, 2/35), and minor periprocedural complications were also noted in two patients (5.7%, 2/35). The average percentages of stenosis of the lesions were 76.6% ± 7% before treatment, 32.4% ± 11.2% after DCB angioplasty, and 25% ± 16% at follow-up. Stenosis over 50% was present in 3 lesions during the follow-up period (8.3%, 3/36).
Conclusion
In this study, the application of DCBs to treat ICAD patients was feasible and safe. A larger scale clinical trial is warranted to further evaluate the safety and efficacy of this treatment.



Similar content being viewed by others
References
Jung JM, Kang DW, Yu KH, Koo JS, Lee JH, Park JM, et al. Predictors of recurrent stroke in patients with symptomatic intracranial arterial stenosis. Stroke. 2012;43:2785–7.
Derdeyn CP, Chimowitz MI, Lynn MJ, Fiorella D, Turan TN, Janis LS, et al. Aggressive medical treatment with or without stenting in high-risk patients with intracranial artery stenosis (SAMMPRIS): the final results of a randomised trial. Lancet. 2014;383:333–41.
Zaidat OO, Fitzsimmons BF, Woodward BK, Wang Z, Killer-Oberpfalzer M, Wakhloo A, et al. Effect of a balloon-expandable intracranial stent vs medical therapy on risk of stroke in patients with symptomatic intracranial stenosis: the VISSIT randomized clinical trial. JAMA. 2015;313:1240–8.
Miao Z, Zhang Y, Shuai J, Jiang C, Zhu Q, Chen K, et al. Thirty-day outcome of a multicenter registry study of stenting for symptomatic intracranial artery stenosis in China. Stroke. 2015;46:2822–9.
Alexander MJ, Zauner A, Chaloupka JC, Baxter B, Callison RC, Gupta R, et al. WEAVE Trial: Final Results in 152 On-Label Patients. Stroke. 2019;50:889–94.
Derdeyn CP, Fiorella D, Lynn MJ, Turan TN, Cotsonis GA, Lane BF, et al. Nonprocedural symptomatic infarction and in-stent restenosis after intracranial angioplasty and stenting in the SAMMPRIS trial (stenting and aggressive medical management for the prevention of recurrent stroke in intracranial stenosis). Stroke. 2017;48:1501–6.
Li TX, Gao BL, Cai DY, Wang ZL, Zhu LF, Xue JY, et al. Wingspan stenting for severe symptomatic intracranial atherosclerotic stenosis in 433 patients treated at a single medical center. PLoS ONE. 2015;10:e0139377.
Unverdorben M, Vallbracht C, Cremers B, Heuer H, Hengstenberg C, Maikowski C, et al. Paclitaxel-coated balloon catheter versus paclitaxel-coated stent for the treatment of coronary in-stent restenosis. Circulation. 2009;119:2986–94.
Megaly M, Rofael M, Saad M, Rezq A, Kohl LP, Kalra A, et al. Outcomes with drug-coated balloons in small-vessel coronary artery disease. Catheter Cardiovasc Interv. 2019;93:E277–86.
Samuels OB, Joseph GJ, Lynn MJ, Smith HA, Chimowitz MI. A standardized method for measuring intracranial arterial stenosis. AJNR Am J Neuroradiol. 2000;21:643–6.
Lin CM, Wu YM, Chang CH, Chen CC, Wang AY. The ANTRACK technique: employing a compliant balloon or stent retriever to advance a large-bore catheter to an occlusion during thrombectomy procedures in acute stroke patients. Oper Neurosurg (Hagerstown). 2019;16:692–9.
Grüntzig AR, Senning A, Siegenthaler WE. Nonoperative dilatation of coronary-artery stenosis: percutaneous transluminal coronary angioplasty. N Engl J Med. 1979;301:61–8.
Smits PC, Vlachojannis GJ, McFadden EP, Royaards K‑J, Wassing J, Joesoef KS, et al. Final 5‑year follow-up of a randomized controlled trial of everolimus- and paclitaxel-eluting stents for coronary revascularization in daily practice: the COMPARE trial (a trial of everolimus-eluting stents and paclitaxel stents for coronary revascularization in daily practice). JACC Cardiovasc Interv. 2015;8:1157–65.
Scheller B, Speck U, Romeike B, Schmitt A, Sovak M, Böhm M, Stoll HP. Contrast media as carriers for local drug delivery. Successful inhibition of neointimal proliferation in the porcine coronary stent model. Eur Heart J. 2003;24(15):1462–7.
Windecker S, Kolh P, Alfonso F, Collet JP, Cremer J, Falk V, et al. ESC/EACTS guidelines on myocardial revascularization: the task force on myocardial revascularization of the european society of cardiology (ESC) and the European Association For Cardio-Thoracic Surgery (EACTS) Developed with the special contribution of the European Association of Percutaneous Cardiovascular Interventions (EAPCI). Eur Heart J. 2014;35:2541–619.
Unverdorben M, Kleber FX, Heuer H, Figulla HR, Vallbracht C, Leschke M, et al. Treatment of small coronary arteries with a paclitaxel-coated balloon catheter in the PEPCAD I study: are lesions clinically stable from 12 to 36 months? EuroIntervention. 2013;9:620–8.
Connors JJ, Wojak JC. Percutaneous transluminal angioplasty for intracranial atherosclerotic lesions: evolution of technique and short-term results. J Neurosurg. 1999;91:415–23.
Nguyen TN, Zaidat OO, Gupta R, Nogueira RG, Tariq N, Kalia JS, et al. Balloon angioplasty for intracranial atherosclerotic disease: periprocedural risks and short-term outcomes in a multicenter study. Stroke. 2011;42:107–11.
Okada H, Terada T, Tanaka Y, Tomura N, Kono K, Yoshimura R, et al. Reappraisal of primary balloon angioplasty without stenting for patients with symptomatic middle cerebral artery stenosis. Neurol Med Chir (Tokyo). 2015;55:133–40.
Jiang WJ, Cheng-Ching E, Abou-Chebl A, Zaidat OO, Jovin TG, Kalia J, et al. Multicenter analysis of stenting in symptomatic intracranial atherosclerosis. Neurosurgery. 2012;70:25–30; discussion 31.
Kurre W, Aguilar-Pérez M, Fischer S, Arnold G, Schmid E, Bäzner H, et al. Solving the issue of restenosis after stenting of intracranial stenoses: experience with two thin-strut drug-eluting stents (DES)-taxus element™ and resolute integrity™. Cardiovasc Intervent Radiol. 2015;38:583–91.
Vajda Z, Aguilar M, Göhringer T, Horváth-Rizea D, Bäzner H, Henkes H. Treatment of intracranial atherosclerotic disease with a balloon-expandable paclitaxel eluting stent: procedural safety, efficacy and mid-term patency. Clin Neuroradiol. 2012;22:227–33.
Vajda Z, Güthe T, Perez MA, Heuschmid A, Schmid E, Bäzner H, et al. Neurovascular in-stent stenoses: treatment with conventional and drug-eluting balloons. AJNR Am J Neuroradiol. 2011;32:1942–7.
Vajda Z, Güthe T, Perez MA, Kurre W, Schmid E, Bäzner H, et al. Prevention of intracranial in-stent restenoses: predilatation with a drug eluting balloon, followed by the deployment of a self-expanding stent. Cardiovasc Intervent Radiol. 2013;36:346–52.
Zhang J, Zhang X, Zhang JP, Han J. Endovascular recanalisation with drug coated balloon for chronic symptomatic middle cerebral artery total occlusion. BMJ Case Rep. 2018. https://doi.org/10.1136/bcr-2017-013693
Gruber P, Garcia-Esperon C, Berberat J, Kahles T, Hlavica M, Anon J, et al. Neuro elutax SV drug-eluting balloon versus wingspan stent system in symptomatic intracranial high-grade stenosis: a single-center experience. J Neurointerv Surg. 2018;10:e32.
Wang Y, Ma Y, Gao P, Chen Y, Yang B, Jiao L. First report of drug-coated balloon angioplasty for vertebral artery origin stenosis. JACC Cardiovasc Interv. 2018;11:500–2.
Kleber FX, Schulz A, Waliszewski M, Hauschild T, Böhm M, Dietz U, et al. Local paclitaxel induces late lumen enlargement in coronary arteries after balloon angioplasty. Clin Res Cardiol. 2015;104:217–25.
Yu SCH, Lau TWW, Wong SSM, Lee KT, Wong LKS, Leung TWH. Long-term evolutionary change in the lumen of intracranial atherosclerotic stenosis following angioplasty and stenting. Oper Neurosurg (Hagerstown). 2017;14:128–38.
Cortese B, Silva Orrego P, Agostoni P, Buccheri D, Piraino D, Andolina G, et al. Effect of drug-coated balloons in native coronary artery disease left with a dissection. JACC Cardiovasc Interv. 2015;8:2003–9.
Kim S, Choi JH, Kang M, Cha JK, Huh JT. Safety and efficacy of intravenous tirofiban as antiplatelet premedication for stent-assisted coiling in acutely ruptured intracranial aneurysms. AJNR Am J Neuroradiol. 2016;37:508–14.
Wang ZL, Gao BL, Li TX, Cai DY, Zhu LF, Xue JY, et al. Severe symptomatic intracranial internal carotid artery stenosis treated with intracranial stenting: a single center study with 58 patients. Diagn Interv Radiol. 2016;22:178–83.
Wang ZL, Gao BL, Li TX, Cai DY, Zhu LF, Xue JY, et al. Outcomes of middle cerebral artery angioplasty and stenting with Wingspan at a high-volume center. Neuroradiology. 2016;58:161–9.
Wang ZL, Gao BL, Li TX, Cai DY, Zhu LF, Bai WX, Xue JY, Li ZS. Symptomatic intracranial vertebral artery atherosclerotic stenosis (≥70%) with concurrent contralateral vertebral atherosclerotic diseases in 88 patients treated with the intracranial stenting. Eur J Radiol. 2015;84:1801 4.
Bai WX, Gao BL, Li TX, Wang ZL, Cai DY, Zhu LF, et al. Wingspan stenting can effectively prevent long-term strokes for patients with severe symptomatic atherosclerotic basilar stenosis. Interv Neuroradiol. 2016;22:318–24.
Ramakrishna CD, Dave BA, Kothavade PS, Joshi KJ, Thakkar AS. Basic concepts and clinical outcomes of drug-eluting balloons for treatment of coronary artery disease: an overview. J Clin Diagn Res. 2017;11:OE01–4.
Belkacemi A, Agostoni P, Nathoe HM, Voskuil M, Shao C, Van Belle E, et al. First results of the DEB-AMI (drug eluting balloon in acute ST-segment elevation myocardial infarction) trial: a multicenter randomized comparison of drug-eluting balloon plus bare-metal stent versus bare-metal stent versus drug-eluting stent in primary percutaneous coronary intervention with 6-month angiographic, intravascular, functional, and clinical outcomes. J Am Coll Cardiol. 2012;59:2327–37.
Author information
Authors and Affiliations
Contributions
AY Wang and CH Chang wrote the original draft, with equal contributions and both as first authors. All other authors reviewed and provided meaningful feedback in the creation of the final draft. All authors approved the final draft.
Corresponding author
Ethics declarations
Conflict of interest
A.Y.-C. Wang, C.-H. Chang, C.-C. Chen, Y.‑M. Wu, C.-M. Lin, C.-T. Chen and P.-C. Hsieh declare that they have no competing interests.
Additional information
Alvin Yi-Chou Wang and Chien-Hung Chang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Wang, A.YC., Chang, CH., Chen, CC. et al. Leave Nothing Behind: Treatment of Intracranial Atherosclerotic Disease with Drug-Coated Balloon Angioplasty. Clin Neuroradiol 31, 35–44 (2021). https://doi.org/10.1007/s00062-020-00935-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00062-020-00935-w