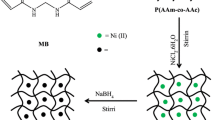
Abstract
In this paper we present the results of the recovery of Ni(II) ions in aqueous media using poly(acrylamide-co-itaconic acid) hydrogels. The copolymer was synthetized via free radical polymerization at 30 °C using the potassium persulfate and N,N,N′,N′-tetramethylethylenediamine redox pair. Several methodologies were studied and compared by varying the following parameters: temperature, pH of the media, Ni(II) ion concentration and aqueous solution / xerogel weight ratio. Finally, the removal efficiency of the applications of the hydrogel and the influence of the parameters was measured. Our main result was found by swelling the hydrogel in an aqueous media with pH 13, then placed at the Ni(II) solution. With this methodology, the ion recovery performance was up to 94%, this because the polymeric matrix is saturated with a NaOH solution and the nickel ions cannot penetrate the hydrogel considerably. Thus instead of absorbing the Ni(II) ion, it reacts with the OH− ions delivered at the surface of the hydrogel, forming Ni(OH)2 layer at the surface, which is partially absorbed.







Similar content being viewed by others
References
Prica M, Adamovic S, Dalmacija B, Rajic L, Trickovic J, Rapajic S, Becelic-Tomin M (2015) The electrocoagulation/flotation study: the removal of heavy metals from the waste fountain solution. Process Saf Environ Prot 94:262–273. https://doi.org/10.1016/j.psep.2014.07.002
Benvenuti T, Krapf RS, Rodrigues MAS, Bernardes AM, Zoppas-Ferreira J (2014) Recovery of nickel and water from nickel electroplating wastewater by electrodialysis. Sep Purif Technol 129:106–112. https://doi.org/10.1016/j.seppur.2014.04.002
Li T, Zhang W, Zhai S, Gao G, Ding J, Zhang W, Liu Y, Zhao X, Pan B, Lv L (2018) Efficient removal of nickel (II) from high salinity wastewater by a novel PAA/ZIF-8/PVDF hybrid ultrafiltration membrane. Water Res 2018(143):87–98. https://doi.org/10.1016/j.watres.2018.06.031
Zhang Q, Gao J, Qiu YR (2019) Removal of Ni (II) and Cr (III) by complexation-ultrafiltration using rotating disk membrane and the selective separation by shear induced dissociation. Chemical Engineering and Processing-Process Intensification 135:236–244. https://doi.org/10.1016/j.cep.2018.12.005
Akbal F, Camcı S (2011) Copper, chromium and nickel removal from metal plating wastewater by electrocoagulation. Desalination 269:214–222. https://doi.org/10.1016/j.desal.2010.11.001
Keränen A, Leiviskä T, Salakka A, Tanskanen J (2015) Removal of nickel and vanadium from ammoniacal industrial wastewater by ion exchange and adsorption on activated carbon. Desalin Water Treat 53:2645–2654. https://doi.org/10.1080/19443994.2013.868832
Ozay O, Ekici S, Baran Y, Aktas N, Sahiner N (2009) Removal of toxic metal ions with magnetic hydrogels. Water Res 43:4403–4411. https://doi.org/10.1016/j.watres.2009.06.058
Madill E, Garcia-Valdez O, Champagne P, Cunningham M (2017) CO2-responsive graft modified chitosan for heavy metal (nickel) recovery. Polymer 9:394. https://doi.org/10.3390/polym9090394
Aguilar-González MÁ, Gorokhovsky AV, Aguilar-Elguezabal A (2010) Removal of lead and nickel from aqueous solutions by SiO2 doped potassium titanate. Mater Sci Eng B 174:105–113. https://doi.org/10.1016/j.mseb.2010.03.057
Malamis S, Katsou E (2013) A review on zinc and nickel adsorption on natural and modified zeolite, bentonite and vermiculite: examination of process parameters, kinetics and isotherms. J Hazard Mater 252:428–461. https://doi.org/10.1016/j.jhazmat.2013.03.024
Peric J, Trgo M, Medvidovic NV (2004) Removal of zinc, copper and lead by natural zeolite—a comparison of adsorption isotherms. Water Res 38:1893–1899. https://doi.org/10.1016/j.watres.2003.12.035
Ouadjenia-Marouf F, Marouf R, Schott J, Yahiaoui A (2013) Removal of cu(II), cd(II) and Cr(III) ions from aqueous solution by dam silt. Arab J Chem 6:401–406. https://doi.org/10.1016/j.arabjc.2010.10.018
Muya FN, Sunday CE, Baker P, Iwuoha E (2015) Environmental remediation of heavy metal ions from aqueous solution through hydrogel adsorption: a critical review. Water Sci Technol 73:983–992. https://doi.org/10.2166/wst.2015.567
Özkahraman B, Acar I, Emik S (2011) Removal of cationic dyes from aqueous solutions with poly (N-isopropylacrylamide-co-itaconic acid) hydrogels. Polym Bull 66:551–570. https://doi.org/10.1007/s00289-010-0371-1
Ma X, Wen G (2020) Development history and synthesis of super-absorbent polymers: a review. J Polym Res 27:1–12. https://doi.org/10.1007/s10965-020-02097-2
Li T, Liu X, Li L, Wang Y, Ma P, Chen M, Dong W (2019) Polydopamine-functionalized graphene oxide compounded with polyvinyl alcohol/chitosan hydrogels on the recyclable adsorption of cu (II), Pb (II) and cd (II) from aqueous solution. J Polym Res 26(12):281. https://doi.org/10.1007/s10965-019-1971-6
El Halah A, Contreras J, Rojas-Rojas L, Rivas M, Romero M, López-Carrasquero F (2015) New superabsorbent hydrogels synthesized by copolymerization of acrylamide and N-2-hydroxyethyl acrylamide with itaconic acid or itaconates containing ethylene oxide units in the side chain. J Polym Res 22:233. https://doi.org/10.1007/s10965-015-0876-2
El-Halah A, Machado D, González N, Contreras J, López-Carrasquero F (2019) Use of super absorbent hydrogels derivative from acrylamide with itaconic acid and itaconates to remove metal ions from aqueous solutions. J Appl Polym Sci 136:46999. https://doi.org/10.1002/app.46999
Li J, Xu Z, Wu W, Jing Y, Dai H, Fang G (2018) Nanocellulose/poly (2-(dimethylamino) ethyl methacrylate) interpenetrating polymer network hydrogels for removal of Pb (II) and cu (II) ions. Colloids Surf A Physicochem Eng Asp 538:474–480. https://doi.org/10.1016/j.colsurfa.2017.11.019
Hong TT, Okabe H, Hidaka Y, Omondi BA, Hara K (2019) Radiation induced modified CMC-based hydrogel with enhanced reusability for heavy metal ions adsorption. Polymer 181:121772. https://doi.org/10.1016/j.polymer.2019.121772
Katime I, Rodríguez E (2001) Absorption of metal ions and swelling properties of poly (acrylic acid-co-itaconic acid) hydrogels. J Macromol Sci A 38:543–558. https://doi.org/10.1081/MA-100103366
Roy P, Swami V, Kumar D, Rajagopal C (2011) Removal of toxic metals using superabsorbent polyelectrolytic hydrogels. J Appl Polym Sci 122:2415–2423. https://doi.org/10.1002/app.34384
Barati A, Asgari M, Miri T, Eskandari Z (2013) Removal and recovery of copper and nickel ions from aqueous solution by poly (methacrylamide-co-acrylic acid)/montmorillonite nanocomposites. Environ Sci Pollut Res 20:6242–6255. https://doi.org/10.1007/s11356-013-1672-3
Abdelwahab H, Hassan S, Mostafa M, El Sadek M (2016) Synthesis and characterization of glutamic-chitosan hydrogel for copper and nickel removal from wastewater. Molecules 21:684. https://doi.org/10.3390/molecules21060684
Lv Q, Hu X, Zhang X, Huang L, Liu Z, Sun G (2019) Highly efficient removal of trace metal ions by using poly (acrylic acid) hydrogel adsorbent. Mater Des 107934:107934. https://doi.org/10.1016/j.matdes.2019.107934
Kasgöz H, Özgümüs S, Orbay M (2003) Modified polyacrylamide hydrogels and their application in removal of heavy metal ions. Polymer 44:1785–1793. https://doi.org/10.1016/S0032-3861(03)00033-8
Tasdelen B, Çifçi DI, Meriç S (2017) Preparation of N-isopropylacrylamide/itaconic acid/pumice highly swollen composite hydrogels to explore their removal capacity of methylene blue. Colloids Surf A Physicochem Eng Asp 519:245–253. https://doi.org/10.1016/j.colsurfa.2016.11.003
Acknowledgements
J.A. Cortés and A.G. Alvarado gratefully a knowledge the finacial support from PROSNI program, Universidad de Guadalajara.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cortés, J.A., Zárate-Navarro, M.A. & Alvarado, A.G. Study and comparison of several methods to remove Ni(II) ions in aqueous solutions using poly(acrylamide-co-itaconic acid) hydrogels. J Polym Res 27, 238 (2020). https://doi.org/10.1007/s10965-020-02225-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-020-02225-y