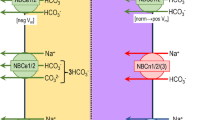
Abstract
Many anion channels, frequently referred as Cl− channels, are permeable to different anions in addition to Cl−. As the second-most abundant anion in the human body, HCO3− permeation via anion channels has many important physiological roles. In addition to its classical role as an intracellular pH regulator, HCO3− also controls the activity and stability of dissolved proteins in bodily fluids such as saliva, pancreatic juice, intestinal fluid, and airway surface liquid. Moreover, HCO3− permeation through these channels affects membrane potentials that are the driving forces for transmembrane transport of solutes and water in epithelia and affect neuronal excitability in nervous tissue. Consequently, aberrant HCO3− transport via anion channels causes a number of human diseases in respiratory, gastrointestinal, genitourinary, and neuronal systems. Notably, recent studies have shown that the HCO3− permeabilities of several anion channels are not fixed and can be altered by cellular stimuli, findings which may have both physiological and pathophysiological significance. In this review, we summarize recent progress in understanding the molecular mechanisms and the physiological roles of HCO3− permeation through anion channels. We hope that the present discussions can stimulate further research into this very important topic, which will provide the basis for human disorders associated with aberrant HCO3− transport.





Similar content being viewed by others
References
Aguilella-Arzo M, Andrio A, Aguilella VM, Alcaraz A (2009) Dielectric saturation of water in a membrane protein channel. Phys Chem Chem Phys 11(2):358–365. https://doi.org/10.1039/b812775a
Alka K, Casey JR (2014) Bicarbonate transport in health and disease. IUBMB Life 66(9):596–615. https://doi.org/10.1002/iub.1315
Alvaro D, Alpini G, Jezequel AM, Bassotti C, Francia C, Fraioli F, Romeo R, Marucci L, Le Sage G, Glaser SS, Benedetti A (1997) Role and mechanisms of action of acetylcholine in the regulation of rat cholangiocyte secretory functions. J Clin Invest 100(6):1349–1362. https://doi.org/10.1172/JCI119655
Arreola J, Melvin JE, Begenisich T (1995) Volume-activated chloride channels in rat parotid acinar cells. J Physiol 484(Pt 3):677–687. https://doi.org/10.1113/jphysiol.1995.sp020695
Ben-Ari Y, Gaiarsa JL, Tyzio R, Khazipov R (2007) GABA: a pioneer transmitter that excites immature neurons and generates primitive oscillations. Physiol Rev 87(4):1215–1284. https://doi.org/10.1152/physrev.00017.2006
Bormann J, Hamill OP, Sakmann B (1987) Mechanism of anion permeation through channels gated by glycine and gamma-aminobutyric acid in mouse cultured spinal neurons. J Physiol 385:243–286. https://doi.org/10.1113/jphysiol.1987.sp016493
Born M (1920) Volumen un hydrationwärme der ionen. Z Phys 1:45–48
Brunner JD, Lim NK, Schenck S, Duerst A, Dutzler R (2014) X-ray structure of a calcium-activated TMEM16 lipid scramblase. Nature 516(7530):207–212. https://doi.org/10.1038/nature13984
Caputo A, Caci E, Ferrera L, Pedemonte N, Barsanti C, Sondo E, Pfeffer U, Ravazzolo R, Zegarra-Moran O, Galietta LJ (2008) TMEM16A, a membrane protein associated with calcium-dependent chloride channel activity. Science 322(5901):590–594. https://doi.org/10.1126/science.1163518
Catalan M, Niemeyer MI, Cid LP, Sepulveda FV (2004) Basolateral ClC-2 chloride channels in surface colon epithelium: regulation by a direct effect of intracellular chloride. Gastroenterology 126(4):1104–1114. https://doi.org/10.1053/j.gastro.2004.01.010
Chan HC, Shi QX, Zhou CX, Wang XF, Xu WM, Chen WY, Chen AJ, Ni Y, Yuan YY (2006) Critical role of CFTR in uterine bicarbonate secretion and the fertilizing capacity of sperm. Mol Cell Endocrinol 250(1–2):106–113. https://doi.org/10.1016/j.mce.2005.12.032
Chen TY, Hwang TC (2008) CLC-0 and CFTR: chloride channels evolved from transporters. Physiol Rev 88(2):351–387. https://doi.org/10.1152/physrev.00058.2006
Chen H, Chatelain FC, Lesage F (2014) Altered and dynamic ion selectivity of K+ channels in cell development and excitability. Trends Pharmacol Sci 35(9):461–469. https://doi.org/10.1016/j.tips.2014.06.002
Chung MK, Guler AD, Caterina MJ (2008) TRPV1 shows dynamic ionic selectivity during agonist stimulation. Nat Neurosci 11(5):555–564. https://doi.org/10.1038/nn.2102
Cohn JA, Strong TV, Picciotto MR, Nairn AC, Collins FS, Fitz JG (1993) Localization of the cystic fibrosis transmembrane conductance regulator in human bile duct epithelial cells. Gastroenterology 105(6):1857–1864. https://doi.org/10.1016/0016-5085(93)91085-v
Curtis CM, Martin LC, Higgins CF, Colledge WH, Hickman ME, Evans MJ, MacVinish LJ, Cuthbert AW (1998) Restoration by intratracheal gene transfer of bicarbonate secretion in cystic fibrosis mouse gallbladder. Am J Physiol 274(6):G1053–G1060. https://doi.org/10.1152/ajpgi.1998.274.6.G1053
Dawson DC, Smith SS, Mansoura MK (1999) CFTR: mechanism of anion conduction. Physiol Rev 79(1 Suppl):S47–S75. https://doi.org/10.1152/physrev.1999.79.1.S47
Devor DC, Singh AK, Lambert LC, DeLuca A, Frizzell RA, Bridges RJ (1999) Bicarbonate and chloride secretion in Calu-3 human airway epithelial cells. J Gen Physiol 113(5):743–760. https://doi.org/10.1085/jgp.113.5.743
Di Fulvio M, Aguilar-Bryan L (2019) Chloride transporters and channels in beta-cell physiology: revisiting a 40-year-old model. Biochem Soc Trans 47(6):1843–1855. https://doi.org/10.1042/BST20190513
Domschke S, Domschke W, Rosch W, Konturek SJ, Sprugel W, Mitznegg P, Wunsch E, Demling L (1977) Inhibition by somatostatin of secretin-stimulated pancreatic secretion in man: a study with pure pancreatic juice. Scand J Gastroenterol 12(1):59–63
Dubyak GR (2004) Ion homeostasis, channels, and transporters: an update on cellular mechanisms. Adv Physiol Educ 28(1–4):143–154. https://doi.org/10.1152/advan.00046.2004
Duran C, Thompson CH, Xiao Q, Hartzell HC (2010) Chloride channels: often enigmatic, rarely predictable. Annu Rev Physiol 72:95–121. https://doi.org/10.1146/annurev-physiol-021909-135811
Durie PR, Forstner GG (1989) Pathophysiology of the exocrine pancreas in cystic fibrosis. J R Soc Med 82(Suppl 16):2–10
Dutta AK, Khimji AK, Kresge C, Bugde A, Dougherty M, Esser V, Ueno Y, Glaser SS, Alpini G, Rockey DC, Feranchak AP (2011) Identification and functional characterization of TMEM16A, a Ca2+-activated Cl- channel activated by extracellular nucleotides, in biliary epithelium. J Biol Chem 286(1):766–776. https://doi.org/10.1074/jbc.M110.164970
Dutzler R, Campbell EB, Cadene M, Chait BT, MacKinnon R (2002) X-ray structure of a ClC chloride channel at 3.0 A reveals the molecular basis of anion selectivity. Nature 415(6869):287–294. https://doi.org/10.1038/415287a
Eisbruch A, Rhodus N, Rosenthal D, Murphy B, Rasch C, Sonis S, Scarantino C, Brizel D (2003) How should we measure and report radiotherapy-induced xerostomia? Semin Radiat Oncol 13(3):226–234. https://doi.org/10.1016/S1053-4296(03)00033-X
Fahlke C (2000) Molecular mechanisms of ion conduction in ClC-type chloride channels: lessons from disease-causing mutations. Kidney Int 57(3):780–786. https://doi.org/10.1046/j.1523-1755.2000.00915.x
Fatima-Shad K, Barry PH (1993) Anion permeation in GABA- and glycine-gated channels of mammalian cultured hippocampal neurons. Proc Biol Sci 253(1336):69–75. https://doi.org/10.1098/rspb.1993.0083
Fitz JG, Basavappa S, McGill J, Melhus O, Cohn JA (1993) Regulation of membrane chloride currents in rat bile duct epithelial cells. J Clin Invest 91(1):319–328. https://doi.org/10.1172/JCI116188
Gaitan-Penas H, Gradogna A, Laparra-Cuervo L, Solsona C, Fernandez-Duenas V, Barrallo-Gimeno A, Ciruela F, Lakadamyali M, Pusch M, Estevez R (2016) Investigation of LRRC8-mediated volume-regulated anion currents in Xenopus oocytes. Biophys J 111(7):1429–1443. https://doi.org/10.1016/j.bpj.2016.08.030
Garnett JP, Hickman E, Burrows R, Hegyi P, Tiszlavicz L, Cuthbert AW, Fong P, Gray MA (2011) Novel role for pendrin in orchestrating bicarbonate secretion in cystic fibrosis transmembrane conductance regulator (CFTR)-expressing airway serous cells. J Biol Chem 286(47):41069–41082. https://doi.org/10.1074/jbc.M111.266734
Gee HY, Noh SH, Tang BL, Kim KH, Lee MG (2011) Rescue of DeltaF508-CFTR trafficking via a GRASP-dependent unconventional secretion pathway. Cell 146(5):746–760. https://doi.org/10.1016/j.cell.2011.07.021
Gee HY, Noh SH, Tang BL, Kim KH, Lee MG (2011) Rescue of ΔF508-CFTR trafficking via a GRASP-dependent unconventional secretion pathway. Cell 146(5):746–760. https://doi.org/10.1016/j.cell.2011.07.021
Gennari FJ, Weise WJ (2008) Acid-base disturbances in gastrointestinal disease. Clin J Am Soc Nephrol 3(6):1861–1868. https://doi.org/10.2215/CJN.02450508
Grubb BR, Gabriel SE (1997) Intestinal physiology and pathology in gene-targeted mouse models of cystic fibrosis. Am J Physiol 273(2 Pt 1):G258–G266. https://doi.org/10.1152/ajpgi.1997.273.2.G258
Gunawardana SC, Sharp GW (2002) Intracellular pH plays a critical role in glucose-induced time-dependent potentiation of insulin release in rat islets. Diabetes 51(1):105–113. https://doi.org/10.2337/diabetes.51.1.105
Han YF, Shewan AM, Thorn P (2016) HCO3− transport through Anoctamin/Transmembrane protein ANO1/TMEM16A in pancreatic Acinar cells regulates luminal pH. J Biol Chem 291(39):20345–20352. https://doi.org/10.1074/jbc.M116.750224
Hatefi Y, Hanstein WG (1969) Solubilization of particulate proteins and nonelectrolytes by chaotropic agents. Proc Natl Acad Sci U S A 62(4):1129–1136
Hirata K, Nathanson MH (2001) Bile duct epithelia regulate biliary bicarbonate excretion in normal rat liver. Gastroenterology 121(2):396–406. https://doi.org/10.1053/gast.2001.26280
Hogan DL, Crombie DL, Isenberg JI, Svendsen P, Schaffalitzky de Muckadell OB, Ainsworth MA (1997) CFTR mediates cAMP- and Ca2+-activated duodenal epithelial HCO3- secretion. Am J Physiol 272(4 Pt 1):G872–G878. https://doi.org/10.1152/ajpgi.1997.272.4.G872
Hohenester S, Wenniger LM, Paulusma CC, van Vliet SJ, Jefferson DM, Elferink RP, Beuers U (2012) A biliary HCO3- umbrella constitutes a protective mechanism against bile acid-induced injury in human cholangiocytes. Hepatology 55(1):173–183. https://doi.org/10.1002/hep.24691
Hubner CA, Holthoff K (2013) Anion transport and GABA signaling. Front Cell Neurosci 7:177. https://doi.org/10.3389/fncel.2013.00177
Hwang TC, Kirk KL (2013) The CFTR ion channel: gating, regulation, and anion permeation. Cold Spring Harb Perspect Med 3(1):a009498. https://doi.org/10.1101/cshperspect.a009498
Ishiguro H, Steward MC, Lindsay AR, Case RM (1996) Accumulation of intracellular HCO3- by Na+-HCO3- cotransport in interlobular ducts from guinea-pig pancreas. J Physiol 495(Pt 1):169–178
Ishiguro H, Steward MC, Wilson RW, Case RM (1996) Bicarbonate secretion in interlobular ducts from guinea-pig pancreas. J Physiol 495(Pt 1):179–191
Ishiguro H, Yamamoto A, Nakakuki M, Yi L, Ishiguro M, Yamaguchi M, Kondo S, Mochimaru Y (2012) Physiology and pathophysiology of bicarbonate secretion by pancreatic duct epithelium. Nagoya J Med Sci 74(1–2):1–18
Jentsch TJ (2016) VRACs and other ion channels and transporters in the regulation of cell volume and beyond. Nat Rev Mol Cell Biol 17(5):293–307. https://doi.org/10.1038/nrm.2016.29
Jentsch TJ, Pusch M (2018) CLC chloride channels and transporters: structure, function, physiology, and disease. Physiol Rev 98(3):1493–1590. https://doi.org/10.1152/physrev.00047.2017
Jentsch TJ, Steinmeyer K, Schwarz G (1990) Primary structure of Torpedo marmorata chloride channel isolated by expression cloning in Xenopus oocytes. Nature 348(6301):510–514. https://doi.org/10.1038/348510a0
Jentsch TJ, Stein V, Weinreich F, Zdebik AA (2002) Molecular structure and physiological function of chloride channels. Physiol Rev 82(2):503–568. https://doi.org/10.1152/physrev.00029.2001
Joo NS, Irokawa T, Robbins RC, Wine JJ (2006) Hyposecretion, not hyperabsorption, is the basic defect of cystic fibrosis airway glands. J Biol Chem 281(11):7392–7398. https://doi.org/10.1074/jbc.M512766200
Jun I, Cheng MH, Sim E, Jung J, Suh BL, Kim Y, Son H, Park K, Kim CH, Yoon JH, Whitcomb DC, Bahar I, Lee MG (2016) Pore dilatation increases the bicarbonate permeability of CFTR, ANO1 and glycine receptor anion channels. J Physiol 594(11):2929–2955. https://doi.org/10.1113/JP271311
Jung J, Lee MG (2014) Role of calcium signaling in epithelial bicarbonate secretion. Cell Calcium 55(6):376–384. https://doi.org/10.1016/j.ceca.2014.02.002
Jung J, Lee MG (2015) Does calmodulin regulate the bicarbonate permeability of ANO1/TMEM16A or not? J Gen Physiol 145(1):75–77. https://doi.org/10.1085/jgp.201411283
Jung J, Nam JH, Park HW, Oh U, Yoon JH, Lee MG (2013) Dynamic modulation of ANO1/TMEM16A HCO3− permeability by Ca2+/calmodulin. Proc Natl Acad Sci U S A 110(1):360–365. https://doi.org/10.1073/pnas.1211594110
Kaila K, Pasternack M, Saarikoski J, Voipio J (1989) Influence of GABA-gated bicarbonate conductance on potential, current and intracellular chloride in crayfish muscle fibres. J Physiol 416:161–181. https://doi.org/10.1113/jphysiol.1989.sp017755
Kim Y, Jun I, Shin DH, Yoon JG, Piao H, Jung J, Park HW, Cheng MH, Bahar I, Whitcomb DC, Lee MG (2019) Regulation of CFTR bicarbonate channel activity by WNK1: implications for pancreatitis and CFTR-related disorders. Cell Mol Gastroenterol Hepatol 9:79–103. https://doi.org/10.1016/j.jcmgh.2019.09.003
Kunzelmann K (2015) TMEM16, LRRC8A, bestrophin: chloride channels controlled by Ca(2+) and cell volume. Trends Biochem Sci 40(9):535–543. https://doi.org/10.1016/j.tibs.2015.07.005
LaRusch J, Jung J, General IJ, Lewis MD, Park HW, Brand RE, Gelrud A, Anderson MA, Banks PA, Conwell D, Lawrence C, Romagnuolo J, Baillie J, Alkaade S, Cote G, Gardner TB, Amann ST, Slivka A, Sandhu B, Aloe A, Kienholz ML, Yadav D, Barmada MM, Bahar I, Lee MG, Whitcomb DC, North American Pancreatitis Study G (2014) Mechanisms of CFTR functional variants that impair regulated bicarbonate permeation and increase risk for pancreatitis but not for cystic fibrosis. PLoS Genet 10(7):e1004376. https://doi.org/10.1371/journal.pgen.1004376
Lee MG, Muallem S (2008) Physiology of duct cell secretion. In: Beger H, Buchler M, Kozarek R, Lerch M, Neoptolemos J, Warshaw A, Whitcomb D, Shiratori K (eds) Pancreas: an integrated textbook of basic science, medicine, and surgery. Blackwell Publishing, Oxford, pp 78–90
Lee MG, Xu X, Zeng W, Diaz J, Wojcikiewicz RJ, Kuo TH, Wuytack F, Racymaekers L, Muallem S (1997) Polarized expression of Ca2+ channels in pancreatic and salivary gland cells. Correlation with initiation and propagation of [Ca2+]i waves. J Biol Chem 272(25):15765–15770. https://doi.org/10.1074/jbc.272.25.15765
Lee MG, Choi JY, Luo X, Strickland E, Thomas PJ, Muallem S (1999) Cystic fibrosis transmembrane conductance regulator regulates luminal Cl−/HCO3− exchange in mouse submandibular and pancreatic ducts. J Biol Chem 274(21):14670–14677
Lee MG, Wigley WC, Zeng W, Noel LE, Marino CR, Thomas PJ, Muallem S (1999) Regulation of Cl−/ HCO3− exchange by cystic fibrosis transmembrane conductance regulator expressed in NIH 3T3 and HEK 293 cells. J Biol Chem 274(6):3414–3421
Lee MG, Ohana E, Park HW, Yang D, Muallem S (2012) Molecular mechanism of pancreatic and salivary gland fluid and HCO3− secretion. Physiol Rev 92(1):39–74. https://doi.org/10.1152/physrev.00011.2011
Linsdell P, Evagelidis A, Hanrahan JW (2000) Molecular determinants of anion selectivity in the cystic fibrosis transmembrane conductance regulator chloride channel pore. Biophys J 78(6):2973–2982. https://doi.org/10.1016/s0006-3495(00)76836-6
Liu F, Zhang Z, Csanady L, Gadsby DC, Chen J (2017) Molecular structure of the human CFTR ion channel. Cell 169(1):85–95 e88. https://doi.org/10.1016/j.cell.2017.02.024
Lopes AG, Guggino WB (1987) Volume regulation in the early proximal tubule of the Necturus kidney. J Membr Biol 97(2):117–125. https://doi.org/10.1007/bf01869418
Low JT, Shukla A, Behrendorff N, Thorn P (2010) Exocytosis, dependent on Ca2+ release from Ca2+ stores, is regulated by Ca2+ microdomains. J Cell Sci 123(Pt 18):3201–3208. https://doi.org/10.1242/jcs.071225
Mall M, Bleich M, Schurlein M, Kuhr J, Seydewitz HH, Brandis M, Greger R, Kunzelmann K (1998) Cholinergic ion secretion in human colon requires coactivation by cAMP. Am J Physiol 275(6):G1274–G1281. https://doi.org/10.1152/ajpgi.1998.275.6.G1274
Mo L, Hellmich HL, Fong P, Wood T, Embesi J, Wills NK (1999) Comparison of amphibian and human ClC-5: similarity of functional properties and inhibition by external pH. J Membr Biol 168(3):253–264. https://doi.org/10.1007/s002329900514
Murek M, Kopic S, Geibel J (2010) Evidence for intestinal chloride secretion. Exp Physiol 95(4):471–478. https://doi.org/10.1113/expphysiol.2009.049445
Neyraud E, Bult JH, Dransfield E (2009) Continuous analysis of parotid saliva during resting and short-duration simulated chewing. Arch Oral Biol 54(5):449–456. https://doi.org/10.1016/j.archoralbio.2009.01.005
Park HW, Nam JH, Kim JY, Namkung W, Yoon JS, Lee JS, Kim KS, Venglovecz V, Gray MA, Kim KH, Lee MG (2010) Dynamic regulation of CFTR bicarbonate permeability by [Cl-]i and its role in pancreatic bicarbonate secretion. Gastroenterology 139(2):620–631. https://doi.org/10.1053/j.gastro.2010.04.004
Park E, Campbell EB, MacKinnon R (2017) Structure of a CLC chloride ion channel by cryo-electron microscopy. Nature 541(7638):500–505. https://doi.org/10.1038/nature20812
Pezzulo AA, Tang XX, Hoegger MJ, Abou Alaiwa MH, Ramachandran S, Moninger TO, Karp PH, Wohlford-Lenane CL, Haagsman HP, van Eijk M, Banfi B, Horswill AR, Stoltz DA, McCray PB Jr, Welsh MJ, Zabner J (2012) Reduced airway surface pH impairs bacterial killing in the porcine cystic fibrosis lung. Nature 487(7405):109–113. https://doi.org/10.1038/nature11130
Piala AT, Moon TM, Akella R, He H, Cobb MH, Goldsmith EJ (2014) Chloride sensing by WNK1 involves inhibition of autophosphorylation. Sci Signal 7(324):ra41. https://doi.org/10.1126/scisignal.2005050
Poroca DR, Pelis RM, Chappe VM (2017) ClC channels and transporters: structure, physiological functions, and implications in human chloride Channelopathies. Front Pharmacol 8:151. https://doi.org/10.3389/fphar.2017.00151
Poulsen JH, Fischer H, Illek B, Machen TE (1994) Bicarbonate conductance and pH regulatory capability of cystic fibrosis transmembrane conductance regulator. Proc Natl Acad Sci U S A 91(12):5340–5344. https://doi.org/10.1073/pnas.91.12.5340
Qu Z, Hartzell HC (2000) Anion permeation in Ca2+-activated Cl− channels. J Gen Physiol 116(6):825–844. https://doi.org/10.1085/jgp.116.6.825
Quinton PM (2001) The neglected ion: HCO3−. Nat Med 7(3):292–293. https://doi.org/10.1038/85429
Quinton PM (2008) Cystic fibrosis: impaired bicarbonate secretion and mucoviscidosis. Lancet 372(9636):415–417. https://doi.org/10.1016/S0140-6736(08)61162-9
Quinton PM (2010) Role of epithelial HCO3- transport in mucin secretion: lessons from cystic fibrosis. Am J Physiol Cell Physiol 299(6):C1222–C1233. https://doi.org/10.1152/ajpcell.00362.2010
Richardson C, Sakamoto K, de los Heros P, Deak M, Campbell DG, Prescott AR, Alessi DR (2011) Regulation of the NKCC2 ion cotransporter by SPAK-OSR1-dependent and -independent pathways. J Cell Sci 124(Pt 5):789–800. https://doi.org/10.1242/jcs.077230
Rivera C, Voipio J, Payne JA, Ruusuvuori E, Lahtinen H, Lamsa K, Pirvola U, Saarma M, Kaila K (1999) The K+/Cl- co-transporter KCC2 renders GABA hyperpolarizing during neuronal maturation. Nature 397(6716):251–255. https://doi.org/10.1038/16697
Romanenko VG, Catalan MA, Brown DA, Putzier I, Hartzell HC, Marmorstein AD, Gonzalez-Begne M, Rock JR, Harfe BD, Melvin JE (2010) Tmem16A encodes the Ca2+-activated Cl- channel in mouse submandibular salivary gland acinar cells. J Biol Chem 285(17):12990–13001. https://doi.org/10.1074/jbc.M109.068544
Rome L, Grantham J, Savin V, Lohr J, Lechene C (1989) Proximal tubule volume regulation in hyperosmotic media: intracellular K+, Na+, and Cl. Am J Physiol 257(6 Pt 1):C1093–C1100. https://doi.org/10.1152/ajpcell.1989.257.6.C1093
Roos A, Boron WF (1981) Intracellular pH. Physiol Rev 61(2):296–434
Roux B, Berneche S, Egwolf B, Lev B, Noskov SY, Rowley CN, Yu H (2011) Ion selectivity in channels and transporters. J Gen Physiol 137(5):415–426. https://doi.org/10.1085/jgp.201010577
Sagheddu C, Boccaccio A, Dibattista M, Montani G, Tirindelli R, Menini A (2010) Calcium concentration jumps reveal dynamic ion selectivity of calcium-activated chloride currents in mouse olfactory sensory neurons and TMEM16b-transfected HEK 293T cells. J Physiol 588(Pt 21):4189–4204. https://doi.org/10.1113/jphysiol.2010.194407
Schroeder BC, Cheng T, Jan YN, Jan LY (2008) Expression cloning of TMEM16A as a calcium-activated chloride channel subunit. Cell 134(6):1019–1029. https://doi.org/10.1016/j.cell.2008.09.003
Seidler UE (2013) Gastrointestinal HCO3- transport and epithelial protection in the gut: new techniques, transport pathways and regulatory pathways. Curr Opin Pharmacol 13(6):900–908. https://doi.org/10.1016/j.coph.2013.10.001
Shah VS, Ernst S, Tang XX, Karp PH, Parker CP, Ostedgaard LS, Welsh MJ (2016) Relationships among CFTR expression, HCO3- secretion, and host defense may inform gene- and cell-based cystic fibrosis therapies. Proc Natl Acad Sci U S A 113(19):5382–5387. https://doi.org/10.1073/pnas.1604905113
Shah VS, Meyerholz DK, Tang XX, Reznikov L, Abou Alaiwa M, Ernst SE, Karp PH, Wohlford-Lenane CL, Heilmann KP, Leidinger MR, Allen PD, Zabner J, McCray PB Jr, Ostedgaard LS, Stoltz DA, Randak CO, Welsh MJ (2016) Airway acidification initiates host defense abnormalities in cystic fibrosis mice. Science 351(6272):503–507. https://doi.org/10.1126/science.aad5589
Shan J, Liao J, Huang J, Robert R, Palmer ML, Fahrenkrug SC, O'Grady SM, Hanrahan JW (2012) Bicarbonate-dependent chloride transport drives fluid secretion by the human airway epithelial cell line Calu-3. J Physiol 590(21):5273–5297. https://doi.org/10.1113/jphysiol.2012.236893
Sheppard DN, Rich DP, Ostedgaard LS, Gregory RJ, Smith AE, Welsh MJ (1993) Mutations in CFTR associated with mild-disease-form Cl− channels with altered pore properties. Nature 362(6416):160–164. https://doi.org/10.1038/362160a0
Smith JJ, Welsh MJ (1992) cAMP stimulates bicarbonate secretion across normal, but not cystic fibrosis airway epithelia. J Clin Invest 89(4):1148–1153. https://doi.org/10.1172/JCI115696
Smith SS, Steinle ED, Meyerhoff ME, Dawson DC (1999) Cystic fibrosis transmembrane conductance regulator. Physical basis for lyotropic anion selectivity patterns. J Gen Physiol 114(6):799–818. https://doi.org/10.1085/jgp.114.6.799
Sondo E, Caci E, Galietta LJV (2014) The TMEM16A chloride channel as an alternative therapeutic target in cystic fibrosis. Int J Biochem Cell Biol 52:73–76. https://doi.org/10.1016/j.biocel.2014.03.022
Song Y, Salinas D, Nielson DW, Verkman AS (2006) Hyperacidity of secreted fluid from submucosal glands in early cystic fibrosis. Am J Physiol Cell Physiol 290(3):C741–C749. https://doi.org/10.1152/ajpcell.00379.2005
Staley KJ (2006) Wrong-way chloride transport: is it a treatable cause of some intractable seizures? Epilepsy Curr 6(4):124–127. https://doi.org/10.1111/j.1535-7511.2006.00119.x
Staley KJ, Soldo BL, Proctor WR (1995) Ionic mechanisms of neuronal excitation by inhibitory GABAA receptors. Science 269(5226):977–981. https://doi.org/10.1126/science.7638623
Steward MC, Ishiguro H, Case RM (2005) Mechanisms of bicarbonate secretion in the pancreatic duct. Annu Rev Physiol 67:377–409. https://doi.org/10.1146/annurev.physiol.67.031103.153247
Tang L, Fatehi M, Linsdell P (2009) Mechanism of direct bicarbonate transport by the CFTR anion channel. J Cyst Fibros 8(2):115–121. https://doi.org/10.1016/j.jcf.2008.10.004
Thaysen JH, Thorn NA, Schwartz IL (1954) Excretion of sodium, potassium, chloride and carbon dioxide in human parotid saliva. Am J Physiol 178(1):155–159
Thul R, Falcke M (2004) Release currents of IP3 receptor channel clusters and concentration profiles. Biophys J 86(5):2660–2673. https://doi.org/10.1016/S0006-3495(04)74322-2
Voss FK, Ullrich F, Munch J, Lazarow K, Lutter D, Mah N, Andrade-Navarro MA, von Kries JP, Stauber T, Jentsch TJ (2014) Identification of LRRC8 heteromers as an essential component of the volume-regulated anion channel VRAC. Science 344(6184):634–638. https://doi.org/10.1126/science.1252826
Wakabayashi M, Mori T, Isobe K, Sohara E, Susa K, Araki Y, Chiga M, Kikuchi E, Nomura N, Mori Y, Matsuo H, Murata T, Nomura S, Asano T, Kawaguchi H, Nonoyama S, Rai T, Sasaki S, Uchida S (2013) Impaired KLHL3-mediated ubiquitination of WNK4 causes human hypertension. Cell Rep 3(3):858–868. https://doi.org/10.1016/j.celrep.2013.02.024
Wang XF, Zhou CX, Shi QX, Yuan YY, Yu MK, Ajonuma LC, Ho LS, Lo PS, Tsang LL, Liu Y, Lam SY, Chan LN, Zhao WC, Chung YW, Chan HC (2003) Involvement of CFTR in uterine bicarbonate secretion and the fertilizing capacity of sperm. Nat Cell Biol 5(10):902–906. https://doi.org/10.1038/ncb1047
Weinstein AM (2010) A mathematical model of rat ascending Henle limb. III. Tubular function. Am J Physiol Renal Physiol 298(3):F543–F556. https://doi.org/10.1152/ajprenal.00232.2009
Xiao Q, Cui Y (2014) Acidic amino acids in the first intracellular loop contribute to voltage- and calcium- dependent gating of anoctamin1/TMEM16A. PLoS One 9(6):e99376. https://doi.org/10.1371/journal.pone.0099376
Yamada J, Okabe A, Toyoda H, Kilb W, Luhmann HJ, Fukuda A (2004) Cl- uptake promoting depolarizing GABA actions in immature rat neocortical neurones is mediated by NKCC1. J Physiol 557(Pt 3):829–841. https://doi.org/10.1113/jphysiol.2004.062471
Yamada K, Park HM, Rigel DF, DiPetrillo K, Whalen EJ, Anisowicz A, Beil M, Berstler J, Brocklehurst CE, Burdick DA, Caplan SL, Capparelli MP, Chen G, Chen W, Dale B, Deng L, Fu F, Hamamatsu N, Harasaki K, Herr T, Hoffmann P, Hu QY, Huang WJ, Idamakanti N, Imase H, Iwaki Y, Jain M, Jeyaseelan J, Kato M, Kaushik VK, Kohls D, Kunjathoor V, LaSala D, Lee J, Liu J, Luo Y, Ma F, Mo R, Mowbray S, Mogi M, Ossola F, Pandey P, Patel SJ, Raghavan S, Salem B, Shanado YH, Trakshel GM, Turner G, Wakai H, Wang C, Weldon S, Wielicki JB, Xie X, Xu L, Yagi YI, Yasoshima K, Yin J, Yowe D, Zhang JH, Zheng G, Monovich L (2016) Small-molecule WNK inhibition regulates cardiovascular and renal function. Nat Chem Biol 12(11):896–898. https://doi.org/10.1038/nchembio.2168
Yang YD, Cho H, Koo JY, Tak MH, Cho Y, Shim WS, Park SP, Lee J, Lee B, Kim BM, Raouf R, Shin YK, Oh U (2008) TMEM16A confers receptor-activated calcium-dependent chloride conductance. Nature 455(7217):1210–1215. https://doi.org/10.1038/nature07313
Yang H, Kim A, David T, Palmer D, Jin T, Tien J, Huang F, Cheng T, Coughlin SR, Jan YN, Jan LY (2012) TMEM16F forms a Ca2+-activated cation channel required for lipid scrambling in platelets during blood coagulation. Cell 151(1):111–122. https://doi.org/10.1016/j.cell.2012.07.036
Yu K, Lujan R, Marmorstein A, Gabriel S, Hartzell HC (2010) Bestrophin-2 mediates bicarbonate transport by goblet cells in mouse colon. J Clin Invest 120(5):1722–1735. https://doi.org/10.1172/JCI41129
Yu Y, Kuan AS, Chen TY (2014) Calcium-calmodulin does not alter the anion permeability of the mouse TMEM16A calcium-activated chloride channel. J Gen Physiol 144(1):115–124. https://doi.org/10.1085/jgp.201411179
Zhang GH, Cragoe EJ Jr, Melvin JE (1992) Regulation of cytoplasmic pH in rat sublingual mucous acini at rest and during muscarinic stimulation. J Membr Biol 129(3):311–321
Zhang Z, Chen J (2016) Atomic structure of the cystic fibrosis transmembrane conductance regulator. Cell 167(6):1586–1597 e1589. https://doi.org/10.1016/j.cell.2016.11.014
Zhang Z, Liu F, Chen J (2018) Molecular structure of the ATP-bound, phosphorylated human CFTR. Proc Natl Acad Sci U S A 115(50):12757–12762. https://doi.org/10.1073/pnas.1815287115
Acknowledgments
The authors would like to thank Dong Soo Jang and the Medical Illustration & Design at Yonsei University for providing excellent support with medical illustrations.
Funding
This work was funded by grants 2013R1A3A2042197 (MGL) and 2020R1C1C1009363 (DHS) from the Basic Research Program and grants 2016M3A9B5941215 and 2019M3E5D5066690 (JJ) from the Bio & Medical Technology Program of the National Research Foundation of Korea, funded by the Korean government (MSIT & MOHW), and by the National Institutes of Health, USA, awards P41GM103712 and P30DA035778 (MHC).
Author information
Authors and Affiliations
Contributions
DHS and MGL designed and wrote the first draft of the manuscript. MK, YK, IJ, JJ, and JHN discussed, amended, and finalized the manuscript. MHC developed the model for WNK1-CFTR interaction.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shin, D.H., Kim, M., Kim, Y. et al. Bicarbonate permeation through anion channels: its role in health and disease. Pflugers Arch - Eur J Physiol 472, 1003–1018 (2020). https://doi.org/10.1007/s00424-020-02425-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-020-02425-x