Abstract
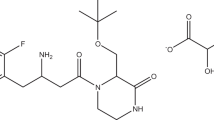
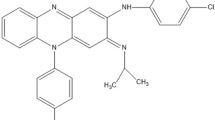
This paper deals with chromatographic method development for the simultaneous estimation of levocetrizine hydrochloride and montelukast sodium in their combined dosage form using analytical quality by design approach. The main objective for application of analytical quality by design approach was to identify conditions where an adequate separation could be achieved within a robust region. Critical method parameters that have the most influence on resolution and retardation factors of both drugs are volume of methanol and volume of modifier (triethylamine); these critical parameters were screened by using Taguchi orthogonal array (OA) design from the seven potential method parameters. Further 32 factorial design was used for establishing the relationship between critical process parameters: volume of methanol and volume of modifier (triethylamine) on critical quality attributes: resolution and retardation factors of both drugs. The chromatographic separation was performed using aluminium backed pre-coated silica gel 60 F254 as stationary phase and ethyl acetate:methanol:triethylamine (5:5:0.04, % v/v) as the mobile phase. The quantification was carried out at 234 nm wavelength. The Rf values were found to be 0.23 ± 0.02 and 0.55 ± 0.02 for levocetrizine hydrochloride and montelukast sodium, respectively. The method was validated as per ICH Q2(R1) guidelines where the method was found to be specific, linear, precise, robust and accurate in the range of 100–500 ng/band for levocetrizine hydrochloride and 200–1000 ng/band for montelukast sodium. The developed HPTLC method was successfully applied for the routine analysis of levocetrizine hydrochloride and montelukast sodium in their combined tablet dosage form.








Similar content being viewed by others
References
Tripathi KD (2003) Essential of medical pharmacology, 5th edn. Jaypee Brothers Medical Publishers, New Delhi
Sharma HL, Sharma KK (2011) Principles of pharmacology, 2nd edn. Paras Medical Publisher, Hyderabad
A. Waugh, A. Grant, Ross and Wilson Anatomy & Physiology in Health and Illness. 12th edn. Churchill Livingstone Elsevier, 2014
Wells BG, Dipiro J, Schwinghammer T, Hamilton C (2006) Pharmacotherapy handbook, 6th edn. McGraw–Hill Medical, New York, NY
Peraman R, Bhadraya K, Padmanabha RY (2015) Analytical quality by design: a tool for regulatory flexibility and robust analytics. Int J Anal Chem:1–9
Basavaiah K, Raghu M, Vinay K (2012) Simple and rapid spectrophotometric assay of levocetirizine in pharmaceuticals through charge-transfer complexation using chloranilic acid and 2,3-dichloro-5,6-dicyanoquinone as π-acceptors. B Chem Soc Ethiopia 26:319–328
Raghu MS, Basavaiah K (2012) Optimized and validated spectrophotometric methods for the determination of levocetirizine in pharmaceuticals based on charge transfer reaction. J Assoc Arab Univ Basic Appl Sci 12:33–41
Saeed AM, Sultana N, Hussain F (2009) Spectrophotometric method for quantitative determination of montelukast in bulk, pharmaceutical formulations and human serum. J Anal Chem 64:690–695
Patel NK, Pancholi S (2011) Spectrophotometric determination of montelukast sodium and levocetirizine dihydrochloride in tablet dosage form by AUC curve method. Pharma Chem 3:135–140
N.K. Patel, P. Chouhan, S.K. Paswan, P.K. Soni, Development and validation of a UV spectrophotometric method for simultaneous estimation of combination of montelukast sodium in presence of levocetirizine dihydrochloride. Pharm Lett. 6 (2014)
Hommoss R, Elzein H, Haidar S (2011) Determination of levocetirizine configurational stability in tablets using chiral HPLC method. Int J Pharm Pharm Sci 3:103–107
Munar A, Frazee C, Jones B, Garg U (2016) Cetirizine quantification by high-performance liquid chromatography–tandem mass spectrometry (HPLC–MS/MS). In: Gard U (ed) Clinical applications of mass spectrometry in drug analysis. Methods and Protocols, Springer, New York, NY, pp 115–120
Hancu G, Budău M, Papp LA, Rusu A, Mircia E (2014) Chiral separation of cetirizine enantiomers by cyclodextrin mediated capillary electrophoresis. Indones J Pharm 25:223
Morita M, Berton D, Boldin R, Barros F, Meurer E, Amarante A, Campos DR, Calafatti SA, Pereira R, Abib E Jr, Pedrazolli J Jr (2008) Determination of levocetirizine in human plasma by liquid chromatography–electrospray tandem mass spectrometry: Application to a bioequivalence study. J. Chromatogr. B 862:132–139
Roman J, Breier AR, Steppe M (2011) Stability indicating LC method to determination of sodium montelukast in pharmaceutical dosage form and its photodegradation kinetics. J. Chromatogr. Sci. 49:540–546
Garg LK, Reddy VS, Sait SS, Krishnamurthy T, Vali SJ, Reddy AM (2013) Quality by design: design of experiments approach prior to the validation of a stability-indicating HPLC method for montelukast. Chromatographia 76:1697–1706
Sripalakit P, Kongthong B, Saraphanchotiwitthaya A (2008) A simple bioanalytical assay for determination of montelukast in human plasma: application to a pharmacokinetic study. J Chromatogr B 869:38–44
Rathore A, Sathiyanarayanan L, Mahadik K (2010) Development of validated HPLC and HPTLC methods for simultaneous determination of levocetirizine dihydrochloride and montelukast sodium in bulk drug and pharmaceutical dosage form. Pharm Anal Acta 106:1–9
Somkuwar S, Pathak A (2012) Simultaneous estimation of levocetirizine dihydrochloride and montelukast sodium by RP-HPLC method. Pharmacia 1:90–94
Patel NK, Patel S, Pancholi S (2012) HPLC method development and validation for simultaneous estimation of montelukast sodium and levocetirizine dihydrochloride in pharmaceutical dosage forms. Int J Pharm Pharm Sci:4–2
Rote A, Niphade V (2011) Determination of Montelukast sodium and Levocetirizine dihydrochloride in combined tablet dosage form by HPTLC and first-derivative spectrophotometry. J Liq Chromatogr Relat Technol 34:155–167
International Conference on Harmonization, Validation of Analytical Procedure: Text and Methodology Q2 (R1), ICH Harmonised Tripartite Guideline, Geneva, 2005
Renger B, Végh Z, Ferenczi-Fodor K (2011) Validation of thin-layer and high-performance thin-layer chromatographic methods. J Chromatogr A 1218:2712–2721
Patel R, Patel M, Dubey N, Dubey N, Patel B (2012) HPTLC method development and validation: strategy to minimize methodological failures. J Food Drug Anal 20:794–804
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Desai, J.A., Dedhiya, P.P., Patel, H.B. et al. Application of analytical quality by design approach in method development for the simultaneous estimation of levocetrizine hydrochloride and montelukast sodium in their combined dosage form. JPC-J Planar Chromat 33, 119–129 (2020). https://doi.org/10.1007/s00764-020-00019-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00764-020-00019-y