Abstract
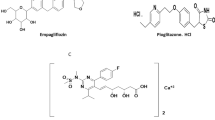
A sensitive, accurate, precise, and robust high-performance thin-layer chromatography (HPTLC) method has been developed for the simultaneous quantification of empagliflozin, linagliptin, and metformin HCl in bulk and synthetic mixture. The method applied HPTLC aluminum plates pre-coated with silica gel 60 F254 as the stationary phase along with n-butanol:water:glacial acetic acid (6:3:1, v/v) as the mobile phase. The Rf values of empagliflozin, linagliptin, and metformin HCl were found to be 0.73, 0.52, and 0.33, respectively. Densitometric analysis was carried out in the absorbance mode at 223 nm. The method was obtained linear in the range of 0.1–0.7 μg/band for empagliflozin, 0.05–0.35 μg/band for linagliptin, and 10–70 μg/band for metformin HCl, respectively. From synthetic mixture, the percentage (%) amounts of all three drugs were found to be more than 98% by the proposed HPTLC method. The method was validated as per the International Conference on Harmonisation (ICH) guideline Q2(R1).







Similar content being viewed by others
References
Drug bank, Empagliflozin, https://www.drugbank.ca/drugs/DB09038 Accessed 9 July 2019
Pubchem, Empagliflozin, https://pubchem.ncbi.nlm.nih.gov/compound/Empagliflozin Accessed15September 2019
Drug bank, Linagliptin https://www.drugbank.ca/drugs/DB08882 Accessed 16 July2019
Drug bank Metformin HCl, https://www.drugbank.ca/drugs/DB00331 Accessed 8 July2019
Approval date for the combination, https://www.biospace.com/article/releases/u-s-fda-accepts-new-drug-application-for-triple-combination-tablet-for-adults-with-type-2-diabetes/ Accessed 16 July 2019
Bhole RP, Wankhede SB, Pandey M (2017) Stability indicating HPTLC method for simultaneous estimation of empagliflozin and linagliptin in pharmaceutical formulation. Ana Chem Lett 7(1):76–85. https://doi.org/10.1080/22297928.2017.1279567
Srivani J, Umamahesh B, Veeresham C (2015) Development and validation of stability indicating HPTLC method for simultaneous determination of linagliptin and metformin. Int J Pharm Pharm Sci 8(1):112–115
Bhole RP, Wankhede SB, Zambare YB, Wankhede TS (2017) High performance thin layer chromatographic determination of linagliptin in pharmaceutical formulations and in biological samples. Int J Pharm Chem Ana 4(1):13–19
Reddy CM, Mubeen G, Pal M (2015) HPTLC Method for estimation of metformin hydrochloride. Biomed Pharmacol J 1(2):445–448
Pubchem, Linagliptin, https://pubchem.ncbi.nlm.nih.gov/compound/Linagliptin Accessed:16 July2019
Patel VB, Patel JS, Shah DA (2018) Estimation of metformin hydrochloride and teneligliptin in pharmaceutical formulation by high performance thin layer chromatography. Indian Drugs 55(10):34–39
ICH Guidelines Q2 (R1), Validation of analytical procedures: text and methodology, Geneva, Switzerland, November 2005
Acknowledgments
All authors are very thankful SICART, Vallabh Vidyanagar, and Indukaka Ipcowala College of Pharmacy, New Vallabh Vidyanagar, for providing necessary facilities to carry out research work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Patel, I.M., Chhalotiya, U.K., Jani, H.D. et al. Densitometric simultaneous estimation of combination of empagliflozin, linagliptin and metformin hydrochloride used in the treatment of type 2 diabetes mellitus. JPC-J Planar Chromat 33, 109–118 (2020). https://doi.org/10.1007/s00764-020-00026-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00764-020-00026-z