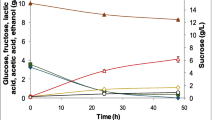
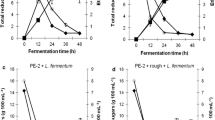
Abstract
In the bioethanol industry, the acid washing applied to yeast cell mass after fermentation cycles is not always effective in removing the unwanted bacteria. In fact, it poses safety hazards due to the handling of large volumes of sulfuric acid. We explored whether sulfuric acid could be replaced, partially or completely, in the acid washing step to control bacterial growth. To achieve this, we evaluated: (1) the effect of ethanol and pH on growth of Lactobacillus fermentum in MRS medium; (2) ethanol as a substitute to or adjuvant of sulfuric acid in the cell treatment against L. fermentum; (3) acid treatments of cells with or without 5% ethanol in cell-recycled batch fermentation with an industrial strain of Saccharomyces cerevisiae and contaminated with L. fermentum. There was a total loss of L. fermentum viability in the following cell treatments: pH 2.0 with 5% ethanol, pH 3.0 with 20% ethanol, only 22% ethanol and pH 2.0 with 3 mg L–1 Kamoran®. Switching the treatment from pH 2.0 with 5% ethanol to pH 2.0 during the fermentative cycles was effective in non-sterile must samples that were not contaminated with L. fermentum, because the native bacteria did not survive the acid treatment (pH 2.0).




Similar content being viewed by others
References
Agtarap A, Chamberlin JW, Pinkerton M, Steinrauf L (1967) The structure of monensic acid, a new biologically active compound. J Am Chem Soc 89:5737–5739. https://doi.org/10.1021/ja00998a062
Amorim HV, Basso LC, Lopes ML (2009) Sugar cane juice and molasses, beet molasses and sweet sorghum: composition and usage. In: Ingledew WM, Kelsall AGD, Kluhspies C (eds) The alcohol textbook. University Press, Nottingham, pp 39–46
Amorim HV, Lopes ML, Oliveira JVC, Buckeridge MS, Goldman GH (2011) Scientific challenges of bioethanol production in Brazil. Appl Microbiol Biotechnol 91:1267–1275. https://doi.org/10.1007/s00253-011-3437-6
Assad L (2017) Aproveitamento de resíduos do setor sucroalcooleiro desafia empresas e pesquisadores. Ciênc Cult 69:13–16. https://doi.org/10.21800/2317-66602017000400005
Bassi APG, Silva JCG, Reis VR, Ceccato-Antonini SR (2013) Effects of single and combined cell treatments based on low pH and high concentrations of ethanol on the growth and fermentation of Dekkera bruxellensis and Saccharomyces cerevisiae. World J Microbiol Biotechnol 29:1661–1676. https://doi.org/10.1007/s11274-013-1329-x
Bassi APG, Meneguello L, Paraluppi AL, Sanches BCP, Ceccato-Antonini SR (2018) Interaction of Saccharomyces cerevisiae–Lactobacillus fermentum–Dekkera bruxellensis and feedstock on fuel ethanol fermentation. Antonie Van Leeuwenhoek 111:1661–1672. https://doi.org/10.1007/s10482-018-1056-2
Basso TO, Lino FSO (2019) Clash of kingdoms: how do bacterial contaminants thrive in and interact with yeasts during ethanol production? In: Basso TP, Basso LC (eds) Fuel ethanol production from sugarcane. IntechOpen, London, pp 23–38
Basso LC, Amorim HV, Oliveira AJ, Lopes ML (2008) Yeast selection for fuel ethanol production in Brazil. FEMS Yeast Res 8:1155–1163. https://doi.org/10.1111/j.1567-1364.2008.00428.x
Basso TO, Gomes FS, Lopes ML, Amorim HV, Eggleston G, Basso LC (2014) Homo-and heterofermentative lactobacilli differently affect sugarcane-based fuel ethanol fermentation. Antonie Van Leeuwenhoek 105:169–177. https://doi.org/10.1007/s10482-013-0063-6
Beckner M, Ivey ML, Phisher TG (2011) Microbial contamination of fuel ethanol fermentations. Lett Appl Microbiol 53:387–394. https://doi.org/10.1111/j.1472-765X.2011.03124.x
Bonatelli ML, Quecine MC, Silva MS, Labate CA (2017) Characterization of the contaminant bacterial communities in sugarcane first-generation industrial ethanol production. FEMS Microbiol Lett 364:fnx159. https://doi.org/10.1093/femsle/fnx159
Braga LPP, Alves RF, Dellias MTF, Navarrete AA, Basso TO, Tsai SM (2017) Vinasse fertirrigation alters soil resistome dynamics: an analysis based on metagenomic profiles. BioData Min 10:17. https://doi.org/10.1186/s13040-017-0138-4
Cao H, Wei D, Yang Y, Shang Y, Li G, Zhou Y, Ma Q, Xu Y (2017) Systems-level understanding of etanol-induced stresses and adaptation in E. coli. Sci Rep 7:1–15. https://doi.org/10.1038/srep44150
Carvalho-Netto OV, Carazzolle MF, Mofatto LS, Teixeira PJPL, Noronha MF, Calderón LAL, Mieczkowski PA, Argueso JL, Pereira GAG (2015) Saccharomyces cerevisiae transcriptional reprograming due to bacterial contamination during industrial scale bioethanol production. Microb Cell Fact 14:1–13. https://doi.org/10.1186/s12934-015-0196-6
Ceballos-Schiavone CHM (2009) Tratamento térmico do caldo de cana-de-açúcar visando a redução de contaminantes bacterianos—Lactobacillus na produção de etanol e eficiência de tratamento do fermento por etanol. Dissertation, Escola Superior de Agricultura “Luiz de Queiroz”-Universidade de São Paulo
Ceccato-Antonini SR (2018) Conventional and nonconventional strategies for controlling bacterial contamination in fuel ethanol fermentations. World J Microbiol Biotechnol 34:80. https://doi.org/10.1007/s11274-018-2463-2
Costa OYA, Souto BM, Tupinambé DD, Bergmann JC, Kyaw CM, Kruger RH, Barreto CC, Quirino BF (2015) Microbial diversity in sugarcane ethanol production in a Brazilian distillery using a culture-independent method. J Ind Microbiol Biotechnol 42:73–84. https://doi.org/10.1007/s10295-014-1533-1
Costa MAS, Cerri BC, Ceccato-Antonini SR (2018) Ethanol addition enhances acid treatment to eliminate Lactobacillus fermentum from the fermentations process for fuel ethanol production. Lett Appl Microbiol 66:77–85. https://doi.org/10.1111/lam.12819
Della-Bianca BE, Hulster E, Pronk JT, van Maris AJA, Gombert AK (2014) Physiology of the fuel ethanol strain Saccharomyces cerevisiae PE-2 at low pH indicates a context-dependent performance relevant for industrial applications. FEMS Yeast Res 14:1196–1205. https://doi.org/10.1111/1567-1364.12217
Dellias MTF, Borges CD, Lopes ML, Cruz SH, Amorim HV, Tsai SM (2018) Biofilm formation and antimicrobial sensitivity of lactobacilli contaminants from sugarcane-based fuel ethanol fermentation. Antonie Van Leeuwenhoek 111:1631–1644. https://doi.org/10.1007/s10482-018-1050-8
Fernandes MLM (1989) Efeito do etanol no crescimento de bactérias láticas. Dissertation, Universidade Técnica de Lisboa
Gallo CR (1989) Determinação da microbiota bacteriana de mosto e de dornas de fermentação alcoólica. Ph.D. Thesis, Universidade de Campinas
Gutknecht J, Walter A (1981) Transport of protons and hydrochloric acid through lipid bilayer membranes. Biochem Biophys Acta 641:183–188. https://doi.org/10.1016/0005-2736(81)90582-4
Herigstad B, Hamilton M, Heersink J (2001) How to optimize the drop late method for enumerating bacteria. J Microbiol Methods 44:121–129. https://doi.org/10.1016/S0167-7012(00)00241-4
Ingram LO (1990) Ethanol tolerance in bacteria. Crit Rev Biotechnol 9:305–319. https://doi.org/10.3109/07388558909036741
Lopes ML, Paulillo SCL, Godoy A, Cherubin RA, Lorenzi MS, Giometti FHC, Bernardino CD, Amorim Neto HB, Amorim HV (2016) Ethanol production in Brazil: a bridge between science and industry. Braz J Microbiol 47:64–76. https://doi.org/10.1016/j.bjm.2016.10.003
Lowicki D, Huczyńsk A (2013) Structure and antimicrobial properties of monensin A and its derivatives: summary of the achievements. Biomed Res Int 2013:1–14. https://doi.org/10.1155/2013/742149
Lucena BTL, Santos BM, Moreira JLS, Moreira APB, Nunes AC, Azevedo V, Miyoshi A, Thompson FL, Morais MA Jr (2010) Diversity of lactic acid bacteria of the bioethanol process. BMC Microbiol 10:298–306. https://doi.org/10.1186/1471-2180-10-298
Lund P, Tramonti A, de Biase D (2014) Coping with low pH: molecular strategies in neutralophilic bacteria. FEMS Microbiol Rev 38:1091–1125. https://doi.org/10.1111/1574-6976.12076
Mendonça AA, Lucena BTL, Morais MMC, Morais MA Jr (2016) First identification of Tn916-like element in industrial strains of Lactobacillus vini that spread the tet-M resistance gene. FEMS Microbiol Lett 363:fnv240. https://doi.org/10.1093/femsle/fnv240
Mendonça AA, Silva PKN, Calazans TLS, Souza RB, Pita WB, Elsztein C, Morais MA Jr (2019) Lactobacillus vini: mechanistic response to stress by medium acidification. Microbiology 165:26–36. https://doi.org/10.1099/mic.0.000738
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428. https://doi.org/10.1021/ac60147a030
Pittet V, Morrow K, Ziola B (2011) Ethanol tolerance of lactic acid bacteria, including relevance of the exopolysaccharide gene gtf. J Am Soc Brew Chem 69:57–61. https://doi.org/10.1094/ASBCJ-2011-0124-01
Ratzke C, Gore J (2018) Modifying and reacting to the environmental pH can drive bacterial interactions. PLOS Biol 16:e2004248. https://doi.org/10.1371/journal.pbio.2004248
Reis VR, Antonangelo ATBF, Bassi APG, Colombi D, Ceccato-Antonini SR (2017) Bioethanol strains of Saccharomyces cerevisiae characterised by microsatellite and stress resistance. Braz J Microbiol 48:268–274. https://doi.org/10.1016/j.bjm.2016.09.017
Russell JB (1987) A proposed mechanism of monensin action in inhibiting ruminal bacterial growth: effects on ion flux and proton motive force. J Anim Sci 64:1519–1525. https://doi.org/10.2527/jas1987.6451519x
Sanz JL, Rodriguez N, Amils R (1996) The action of antibiotics on the anaerobic digestion process. Appl Microbiol Biotechnol 46:587–592. https://doi.org/10.1007/s002530050865
Souza RB, dos Santos BM, Rodrigues de Souza RF, Silva PKN, Lucena BTL, Morais MA Jr (2012) The consequences of Lactobacillus vini and Dekkera bruxellensis as contaminants of the sugarcane-based ethanol fermentation. J Ind Microbiol Biotechnol 39:1645–1650. https://doi.org/10.1007/s10295-012-1167-0
Tiukova I, Eberhard T, Passoth V (2014) Interaction of Lactobacillus vini with the ethanol-producing yeasts Dekkera bruxellensis and Saccharomyces cerevisiae. Biotechnol Appl Biochem 61:40–44. https://doi.org/10.1002/bab.1135
Acknowledgements
This study was supported by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior-Brasil (CAPES)-Finance Code 001.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
da Silva-Neto, J.M., Covre, E.A., Rosa, B.C. et al. Can ethanol partially or fully replace sulfuric acid in the acid wash step of bioethanol production to fight contamination by Lactobacillus fermentum?. Braz. J. Chem. Eng. 37, 323–332 (2020). https://doi.org/10.1007/s43153-020-00033-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43153-020-00033-x