Abstract
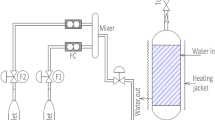
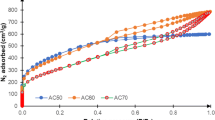
Powdered and granulated activated carbons from babassu were submitted to alkaline treatment with 2 mol L–1 NaOH solution, washed with distilled water to pH 8 and dried. The prepared carbons were tested as catalysts in the Claisen–Schmidt reaction between acetophenone and 4-nitrobenzaldehyde using methanol as solvent at room temperature for 24 h. Babassu granular carbon (BGC) and basic babassu granular carbon (BBGC) exhibited the highest conversions, of 69.26% and 66.59%, respectively. The kinetic study was carried out with BBGC, which led to values of kinetic constant k of 5.26 L mol–1 h–1 at 35 °C and 7.35 L mol–1 h–1 at 50 °C. The activation energy of the reaction was 18.91 kJ mol–1. This study proposes a second-order pseudo-homogeneous reaction catalyzed by BBGC. A continuous fixed-bed reactor operation was carried out with BBGC catalyst and the best conversion was 60% in 120 min using of 0.05 mol L–1 reactants concentration.




Similar content being viewed by others
Abbreviations
- BPC:
-
Babassu powdered carbon
- BBPCWW:
-
Basic babassu powdered carbon without washing
- BBPC:
-
Basic babassu powdered carbon
- BGC:
-
Babassu granular carbon
- BBGC:
-
Basic babassu granular carbon
- UBBPC:
-
Used basic babassu powdered carbon
- UBBGC:
-
Used basic babassu granular carbon
- ASAP:
-
Adsorption and desorption of N2
- TG/DTA:
-
Thermogravimetry and thermo-differential analysis
- FT-IR:
-
Infrared spectroscopy
- SEM/EDS:
-
Screening electron microscopy
- AES:
-
Atomic emission spectroscopy
- AAS:
-
Atomic absorption spectroscopy
References
Aguilera A, Alcantara AR, Marinas JM, Sinisterra JV (1987) Ba(OH)2 as the catalyst in organic reactions. Part XIV. Mechanism of Claisen–Schmidt condensation in solid–liquid conditions. Can J Chem 65(6):1165–1171. https://doi.org/10.1139/v87-195
Bermejo B, Couto Fraga A, Sousa-Aguiar EF (2019) The role of sulfonated activated carbons as catalysts for the hydrolysis of cellobiose. Braz J Chem Eng 36(1):309–315. https://doi.org/10.1590/0104-6632.20190361s20170412
Bhagat S, Sharma R, Sawant DM, Sharma L, Chakraborti AK (2006) LiOH·H2O as a Novel dual activation catalyst for highly efficient and easy synthesis of 1,3-diaryl-2-propenones by Claisen–Schmidt condensation under mild conditions. J Mol Catal Chem 244(1):20–24. https://doi.org/10.1016/j.molcata.2005.08.039
Bitencourt HR, Santos LS, Souza Filho APS (2007) Atividade alelopática de chalcona sintética, de seus precursores e de cetonas e aldeídos relacionados. Planta Daninha 25(4):747–753. https://doi.org/10.1590/S0100-83582007000400011
Boehm HP (2002) Surface oxides on carbon and their analysis: a critical assessment. Carbon 40(2):145–149. https://doi.org/10.1016/S0008-6223(01)00165-8
Cabrera M, Simoens M, Falchi G, Lavaggi ML, Piro OE, Castellano EE, Vidal A, Azqueta A, Monge A, De AC et al (2007) Synthetic chalcones, flavanones, and flavones as antitumoral agents: biological evaluation and structure-activity relationships. Bioorg Med Chem 15(10):3356–3367. https://doi.org/10.1016/j.bmc.2007.03.031
Cai C-Y, Rao L, Rao Y, Guo J-X, Xiao Z-Z, Cao J-Y, Huang Z-S, Wang B (2017) Analogues of xanthones-chalcones and bis-chalcones as α-glucosidase inhibitors and anti-diabetes candidates. Eur J Med Chem 130:51–59. https://doi.org/10.1016/j.ejmech.2017.02.007
Calvino V, Picallo M, López-Peinado AJ, Martín-Aranda RM, Durán-Valle CJ (2006) Ultrasound accelerated claisen-schmidt condensation: a green route to chalcones. Appl Surf Sci 252(17):6071–6074. https://doi.org/10.1016/j.apsusc.2005.11.006
Cerratinga: Instituto Sociedade, População e Natureza. https://cerratinga.org.br Acessed 7 Jul 2019
Chen M, Zhai L, Christensen SB, Theander TG, Kharazmi A (2001) Inhibition of fumarate reductase in Leishmania major and L. donovani by chalcones. Antimicrob Agents Chemother 45(7):2023–2029. https://doi.org/10.1128/AAC.45.7.2023-2029.2001
Chiang H-L, Huang CP, Chiang PC (2002) The surface characteristics of activated carbon as affected by ozone and alkaline treatment. Chemosphere 47(3):257–265. https://doi.org/10.1016/S0045-6535(01)00215-6
Clayden J, Warren S, Greeves N (2012) Organic chemistry, 2nd edn. Oxford Universiry Press, Oxford
Domínguez JN, Charris JE, Lobo G, de Gamboa ND, Moreno MM, Riggione F, Sanchez E, Olson J, Rosenthal PJ (2001) Synthesis of quinolinyl chalcones and evaluation of their antimalarial activity. Eur J Med Chem 36(6):555–560. https://doi.org/10.1016/S0223-5234(01)01245-4
Durán-Valle CJ, Fonseca IM, Calvino-Casilda V, Picallo M, López-Peinado AJ, Martín-Aranda RM (2005) Sonocatalysis and alkaline-doped carbons: an efficient method for the synthesis of chalcones in heterogeneous media. Catal Today 107–108:500–506. https://doi.org/10.1016/j.cattod.2005.07.063
Gasull EI, Silber JJ, Blanco SE, Tomas F, Ferretti FH (2000) A theoretical and experimental study of the formation mechanism of 4-x-chalcones by the Claisen-Schmidt reaction. J Mol Struct THEOCHEM 503(3):131–144. https://doi.org/10.1016/S0166-1280(99)00256-0
Gregg SJ, Sing KSW, Salzberg HW (1967) Adsorption surface area and porosity. J Electrochem Soc 114(11):279C. https://doi.org/10.1149/1.2426447
Guida A, Lhouty MH, Tichit D, Figueras F, Geneste P (1997) Hydrotalcites as base catalysts kinetics of claisen-schmidt condensation, intramolecular condensation of acetonylacetone and synthesis of chalcone. Appl Catal Gen 164(1):251–264. https://doi.org/10.1016/S0926-860X(97)00175-0
Hashimoto K, Kumagai N, Shibasaki M (2014) Self-assembled asymmetric catalyst engaged in a continuous-flow platform: an anti-selective catalytic asymmetric nitroaldol reaction. Org Lett 16(13):3496–3499. https://doi.org/10.1021/ol501432h
He Y, Jawad A, Li X, Atanga M, Rezaei F, Rownaghi AA (2016) Direct aldol and nitroaldol condensation in an aminosilane-grafted Si/Zr/Ti composite hollow fiber as a heterogeneous catalyst and continuous-flow reactor. J Catal 341:149–159. https://doi.org/10.1016/j.jcat.2016.07.001
Herencia F, Ferrándiz ML, Ubeda A, Guillén I, Dominguez JN, Charris JE, Lobo GM, Alcaraz MJ (1999) Novel anti-inflammatory chalcone derivatives inhibit the induction of nitric oxide synthase and cyclooxygenase-2 in mouse peritoneal macrophages. FEBS Lett 453(1–2):129–134. https://doi.org/10.1016/s0014-5793(99)00707-3
Hsieh HK, Tsao LT, Wang JP, Lin CN (2000) Synthesis and anti-inflammatory effect of chalcones. J Pharm Pharmacol 52(2):163–171. https://doi.org/10.1211/0022357001773814
Izquierdo J, Ayats C, Henseler AH, Pericàs MA (2015) A Polystyrene-supported 9-amino(9-deoxy)epi quinine derivative for continuous flow asymmetric michael reactions. Org Biomol Chem 13(14):4204–4209. https://doi.org/10.1039/C5OB00325C
Jin W, Santos JL, Pastor-Perez L, Gu S, Centeno MA, Reina TR (2019) Noble metal supported on activated carbon for “hydrogen free” HDO reactions: exploring economically advantageous routes for biomass valorisation. ChemCatChem 11(17):4434–4441. https://doi.org/10.1002/cctc.201900841
Jioui I, Dânoun K, Solhy A, Jouiad M, Zahouily M, Bilal E, Len C, Fihri A (2016) Modified fluorapatite as highly efficient catalyst for the synthesis of chalcones via Claisen–Schmidt condensation reaction. Arch Ouverte HAL. https://doi.org/10.1016/j.jiec.2016.06.003
Lawrence NJ, Patterson RP, Ooi LL, Cook D, Ducki S (2006) Effects of alpha-substitutions on structure and biological activity of anticancer chalcones. Bioorg Med Chem Lett 16(22):5844–5848. https://doi.org/10.1016/j.bmcl.2006.08.065
Li Z, Zhao H, Han H, Liu Y, Song J, Guo W, Chu W, Sun Z (2017) Graphene-supported ZnO nanoparticles: an efficient heterogeneous catalyst for the Claisen–Schmidt condensation reaction without additional base. Tetrahedron Lett 58(42):3984–3988. https://doi.org/10.1016/j.tetlet.2017.09.011
Mallikarjun KG (2004) Antiviral activity of substituted chalcones and their respective Cu(ii), Ni(ii) and Zn(ii) complexes. J Chem 2(1):58–61. https://doi.org/10.1155/2005/461302
McMurry JE (2014) Organic chemistry with biological applications, 3rd edn. Cengage Learning, Boston
Modzelewska A, Pettit C, Achanta G, Davidson NE, Huang P, Khan SR (2006) Anticancer activities of novel chalcone and bis-chalcone derivatives. Bioorg Med Chem 14(10):3491–3495. https://doi.org/10.1016/j.bmc.2006.01.003
Nondek L, Málek J (1980) Kinetics of condensation of benzaldehyde and its derivatives with acetone and methyl ethyl ketone catalysed by aluminium oxide | Scinapse | Academic search engine for paper. Collect Czech Chem Commun. https://doi.org/10.1135/cccc19801812
Nowakowska Z (2007) A review of anti-infective and anti-inflammatory chalcones. Eur J Med Chem 42(2):125–137. https://doi.org/10.1016/j.ejmech.2006.09.019
Ozdemir Z, Kandilci HB, Gümüşel B, Caliş U, Bilgin AA (2007) Synthesis and studies on antidepressant and anticonvulsant activities of some 3-(2-furyl)-pyrazoline derivatives. Eur J Med Chem 42(3):373–379. https://doi.org/10.1016/j.ejmech.2006.09.006
Qi X, Lai M, Zhu M-J, Peng J-B, Ying J, Wu X-F (2019) 1-arylvinyl formats: a new CO source and ketone source in carbonylative synthesis of chalcone derivatives. ChemCatChem. https://doi.org/10.1002/cctc.201900011
Quaglio D, Zhdanovskaya N, Tobajas G, Cuartas V, Balducci S, Christodoulou MS, Fabrizi G, Gargantilla M, Priego E-M, Carmona-Pestaña Á et al (2019) Chalcones and chalcone-mimetic derivatives as notch inhibitors in a model of T cell acute lymphoblastic leukemia. ACS Med Chem Lett 10(4):639–643. https://doi.org/10.1021/acsmedchemlett.8b00608
Ramalho SD, Bernardes A, Demetrius G, Noda-Perez C, Vieira PC, Dos Santos CY, da Silva JA, de Moraes MO, Mousinho KC (2013) Synthetic chalcone derivatives as inhibitors of cathepsins K and B, and their cytotoxic evaluation. Chem Biodiv. https://doi.org/10.1002/cbdv.201200344(PubMed: NCBI)
Tajuddeen N, Isah MB, Suleiman MA, van Heerden FR, Ibrahim MA (2018) The chemotherapeutic potential of chalcones against leishmaniases: a review. Int J Antimicrob Agents 51(3):311–318. https://doi.org/10.1016/j.ijantimicag.2017.06.010
Tamuly C, Saikia I, Hazarika M, Bordoloi M, Hussain N, Das MR, Deka K (2015) Bio-derived ZnO nanoflower: a highly efficient catalyst for the synthesis of chalcone derivatives. RSC Adv 5(12):8604–8608. https://doi.org/10.1039/C4RA14225J
Tsubogo T, Ishiwata T, Kobayashi S (2013) Asymmetric carbon–carbon bond formation under continuous-flow conditions with chiral heterogeneous catalysts. Angew Chem Int Ed Engl 52(26):6590–6604. https://doi.org/10.1002/anie.201210066
Tsukiyama R-I, Katsura H, Tokuriki N, Kobayashi M (2002) Antibacterial activity of licochalcone A against spore-forming bacteria. Antimicrob Agents Chemother 46(5):1226–1230. https://doi.org/10.1128/AAC.46.5.1226-1230.2002
Vedrine, J. C. Metal Oxides in Heterogeneous Catalysis - 1st Edition Elsevier, 618 (2018).
Vogel S, Ohmayer S, Brunner G, Heilmann J (2008) Natural and non-natural prenylated chalcones: synthesis, cytotoxicity and anti-oxidative activity. Bioorg Med Chem 16(8):4286–4293. https://doi.org/10.1016/j.bmc.2008.02.079
Wagh DP, Yadav GD (2018) Green synthesis of α-methylcinnamaldehyde via Claisen–Schmidt condensation of benzaldehyde with propanal over Mg–Zr mixed oxide supported on HMS. Mol Catal 459:119–128. https://doi.org/10.1016/j.mcat.2018.08.014
Winter C, Caetano JN, Araújo ABC, Chaves AR, Ostroski IC, Vaz BG, Pérez CN, Alonso CG (2016) Activated carbons for chalcone production: Claisen–Schmidt condensation reaction. Chem Eng J 303:604–610. https://doi.org/10.1016/j.cej.2016.06.058
Yadav GD, Yadav AR (2014) Novelty of Claisen–Schmidt condensation of biomass-derived furfural with acetophenone over solid super base catalyst. RSC Adv 4(109):63772–63778. https://doi.org/10.1039/C4RA09194A
Zhang H, Han M, Chen T, Xu L, Yu L (2017) Poly(N-Isopropylacrylamide-Co-l-proline)-catalyzed Claisen–Schmidt and knoevenagel condensations: unexpected enhanced catalytic activity of the polymer catalyst. RSC Adv 7(76):48214–48221. https://doi.org/10.1039/C7RA09412D
Zhao D, Ding K (2013) Recent advances in asymmetric catalysis in flow. ACS Catal 3(5):928–944. https://doi.org/10.1021/cs300830x
Acknowledgements
The authors are grateful to IQ-UFG, for the technical-scientific support; to CAPES and CNPq, for the financial assistance; to LabMic IF-UFG for SEM/EDS analysis; and to Tobasa Bioindustrial de Babaçu S.A. for the activated carbons gently donated.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Catharin, C.W., Chaves, A.R., de Souza, P.S. et al. Babassu activated carbon as catalyst for chalcone production by Claisen–Schmidt reaction: kinetic study, mechanism proposal and continuous flow bed reactor. Braz. J. Chem. Eng. 37, 415–423 (2020). https://doi.org/10.1007/s43153-020-00034-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43153-020-00034-w