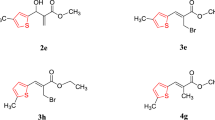
In this work, we report the first-time assessment of o-eugenol, 6-allyl-2-methoxyphenol, and their selected derivatives, against Mycobacterium tuberculosis H37RV, using the MABA susceptibility test. The bromo, nitro, O-alkylated, and reduced derivatives were obtained by standard methods and were characterized by spectroscopic and mass spectral data. Structure–activity relationships were investigated, with the most active derivatives being 4,5-dibromo-2-methoxy-6-propylphenol (139 μM) and 2-methoxy-3-nitro-6-propylphenol (237 μM). This study provides important information on the rational design of new lead anti-TB drugs based on o-eugenol derivatives.

Similar content being viewed by others
References
P. Manitto, D. Monti, and P. Gramatica, J. Chem. Soc., Perkin Trans. 1, 1, 727 (1974).
T. S. Kaufman, J. Braz. Chem. Soc., 26 (6), 1055 (2015).
C. E. Greenhalf, D. J. Nowakowski, N. Yates, I. Shield, and A. V. Bridgwater, Biomass Bioenergy, 56, 247 (2013).
G. T. McQuate, Y. S. Keum, C. D. Sylva, Q. X. Li, and E. B. Jang, J. Econ. Entomol., 97 (3), 862 (2004).
Y. -S. Keum, G. T. McQuate, and Q. X. Li, Biochem. Syst. Ecol., 35 (4), 188 (2007).
H. Cao, L. Zuguang, and C. Xiaozhen, Chin. J. Chem., 29 (10), 2187 (2011).
D.V. Fonseca, P. R. R. Salgado, H. C. A. Neto, A. M. F. O. Golzio, M. R. D. C. Filho, C. G. F. Melo, F. C. Leite, M. R. Piuvezam, L. C. M. Pordeus, J. M. B. Filho, and R. N. Almeida, Int. Immunopharmacol., 38, 402 (2016).
Global Tuberculosis Report 2017, Switzerland: World Health Organization (WHO), 2017. ISBN: 978-92-4-156551-6.
C. Lienhardt, M. Raviglione, M. Spigelamn, R. Hafner, E. Jaramillo, M. Hoelscher, A. Zumla, and J. Gheuens, J. Infect. Dis., 205, S241 (2012).
C. F. H. Allen and J. W. Gates, Org. Synth., 25, 49 (1945).
S. G. Franzblau, R. S. Witzig, J. C. McLaughlin, P. Torres, G. Madico, A. Hernandez, M. T. Degnan, M. B. Cook, V. K. Quenzer, R. M. Ferguson, and R. H. Gilman, J. Clin. Microbiol., 36 (2), 362 (1998).
P. Strazzolini, A. G. Giumanini, A. Runcio, and M. Scuccato, J. Org. Chem., 63 (4), 952 (1998).
C. Wermuth, D. Aldous, P. Raboisson, and D. Rognan, The Practice of Medicinal Chemistry, Academic Press, Amsterdam, 2015, 902 pp.
Y.-L. Lin, J.-Y. Chengy, and Y.-H. Chu, Tetrahedron, 63, 10949 (2007).
J. D. Vanitha and C. N. Paramasivan, Mycobacteriology, 49 (3), 179 (2004).
R. S. Reis, I. Neves, S. L. S. Lourenco, L. S. Fonseca, and M. C. S. Lourenco, J. Clin. Microbiol., 42 (5), 2247 (2004).
ACKNOWLEDGMENT
The authors A. M. Moraes and M. V. N. de Souza are thankful to CNPq for financial support.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Published in Khimiya Prirodnykh Soedinenii, No. 4, July–August, 2020, pp. 549–554.
Rights and permissions
About this article
Cite this article
Moraes, A.M., da Silva, E.T., Wardell, J.L. et al. Synthesis and First-Time Assessment of o-Eugenol Derivatives against Mycobacterium tuberculosis. Chem Nat Compd 56, 633–638 (2020). https://doi.org/10.1007/s10600-020-03110-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-020-03110-2