Abstract
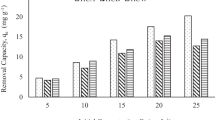
In this study, betel nut husk (BNH) was used to produce activated hydrochar using sodium hydroxide (NaOH). Activated BNH hydrochar (BNH-HAC) served as an adsorbent for methylene blue (MB) adsorption. The produced BNH-HAC was characterized by identifying its textural, morphological, and chemical properties. Batch equilibrium method was conducted to examine the factors that influenced MB adsorption by the BNH-HAC including initial concentration, contact time, solution pH, and temperature. The attained BNH-HAC revealed the BET surface area of 517.60 m2g−1 and mean pore diameter of 25.13 nm. The experimental data can be better characterized with the Freundlich isotherm model than the Langmuir isotherm model where, the maximum adsorption capacities attained were 324.4, 387.4, and 429.6 mg/g at 30 °C, 40 °C, and 50 °C, respectively, of BNH-HAC. Adsorption process regarding the BNH-HAC was regulated by film diffusion model, and its thermodynamic data revealed that the adsorption process is endothermic in character, and kinetic data better suited the pseudo-second order than the pseudo-first order model. The study findings confirmed that BNH-HAC can be an efficiently advanced adsorbent for the adsorption of MB.








Similar content being viewed by others
References
Banglapedia The National Encyclopedia of Bangladesh. http://en.banglapedia.org/index.php?title=Betel_Nut (accessed 13 April, 2020).
Boyd, G. E., Adamson, A. W., & Myers Jr., L. S. (1947). The exchange adsorption of ions from aqueous solutions by organic zeolites. II. Kinetics1. Journal of the American Chemical Society, 69(11), 2836–2848. https://doi.org/10.1021/ja01203a066.
Calugaru, I. L., Neculita, C. M., Genty, T., et al. (2020). Removal and recovery of Ni and Zn from contaminated neutral drainage by thermally activated dolomite and hydrothermally activated wood ash. Water Air and Soil Pollution, 231, 226. https://doi.org/10.1007/s11270-020-04600-3.
Cazetta, A. L., Vargas, A. M., Nogami, E. M., Kunita, M. H., Guilherme, M. R., Martins, A. C., & Almeida, V. C. (2011). NaOH-activated carbon of high surface area produced from coconut shell: kinetics and equilibrium studies from the methylene blue adsorption. Chemical Engineering Journal, 174(1), 117–125. https://doi.org/10.1016/j.cej.2011.08.058.
Chandra, J., George, N., & Narayanankutty, S. K. (2016). Isolation and characterization of cellulose nanofibrils from arecanut husk fibre. Carbohydrate Polymer, 142, 158–166. https://doi.org/10.1016/j.carbpol.2016.01.015.
Chang, J., Ma, J., Ma, Q., Zhang, D., Qiao, N., Hu, M., & Ma, H. (2016). Adsorption of methylene blue onto Fe3O4/activated montmorillonitenanocomposite. Applied Clay Science, 119, 132–140. https://doi.org/10.1016/j.clay.2015.06.038.
Crini, G. (2006). Non-conventional low-cost adsorbents for dye removal: a review. Bioresource Technology, 97(9), 1061–1085. https://doi.org/10.1016/j.biortech.2005.05.001.
Danso-Boateng, E., Nyktari, E., Wheatley, A. D., et al. (2020). Removal of organic pollutants from effluent of anaerobic digester using hydrochars produced from faecal simulant and sewage sludge. Water Air and Soil Pollut, 231, 192. https://doi.org/10.1007/s11270-020-04557-3.
Diaz, E., Manzano, F. J., Villamil, J., Rodriguez, J. J., & Mohedano, A. F. (2019). Low-cost activated grape seed-derived hydrochar through hydrothermal carbonization and chemical activation for sulfamethoxazole adsorption. Appllied Science, 9(23), 5127. https://doi.org/10.3390/app9235127.
Etim, U. J., Umoren, S. A., & Eduok, U. M. (2016). Coconut coir dust as a low cost adsorbent for the removal of cationic dye from aqueous solution. Journal of Saudi Chemical Society, 20, S67–S76. https://doi.org/10.1016/j.jscs.2012.09.014.
Freundlich, H. (1907). Über die adsorption in lösungen. ZeitschriftfürphysikalischeChemie, 57(1), 385–470. https://doi.org/10.1515/zpch-1907-5723.
Hao, W., Björkman, E., Lilliestråle, M., & Hedin, N. (2014). Activated carbons for water treatment prepared by phosphoric acid activation of hydrothermally treated beer waste. Industrial & Engineering Chemistry, 53(40), 15389–15397. https://doi.org/10.1021/ie5004569.
Hernandes, P. T., Oliveira, M. L., Georgin, J., Franco, D. S., Allasia, D., &Dotto, G. L. (2019). Adsorptive decontamination of wastewater containing methylene blue dye using golden trumpet tree bark (Handroanthusalbus). Environ Science and Pollution Research, 26(31), 31924–31933.https://doi.org/10.1007/s11356-019-06353-x.
Ho, Y. S., & McKay, G. (1999). Pseudo-second order model for sorption processes. Process Biochemistry, 34(5), 451–465. https://doi.org/10.1016/S0032-9592(98)00112-5.
Islam, M. A., Benhouria, A., Asif, M., & Hameed, B. H. (2015a). Methylene blue adsorption on factory-rejected tea activated carbon prepared by conjunction of hydrothermal carbonization and sodium hydroxide activation processes. Journal of the Taiwan Institute of Chemical Engineers, 52, 57–64. https://doi.org/10.1016/j.jtice.2015.02.010.
Islam, M. A., Tan, I. A. W., Benhouria, A., Asif, M., & Hameed, B. H. (2015b). Mesoporous and adsorptive properties of palm date seed activated carbon prepared via sequential hydrothermal carbonization and sodium hydroxide activation. Chemical Engineering Journal, 270, 187–195. https://doi.org/10.1016/j.cej.2015.01.058.
Islam, M. A., Ahmed, M. J., Khanday, W. A., Asif, M., & Hameed, B. H. (2017a). Mesoporous activated carbon prepared from NaOH activation of rattan (Lacospermasecundiflorum) hydrochar for methylene blue removal. Ecotoxicology and Environmental Safety, 138, 279–285. https://doi.org/10.1016/j.ecoenv.2017.01.010.
Islam, M. A., Sabar, S., Benhouria, A., Khanday, W. A., Asif, M., & Hameed, B. H. (2017b). Nanoporous activated carbon prepared from karanj (Pongamiapinnata) fruit hulls for methylene blue adsorption. Journal of the Taiwan Institute of Chemical Engineers, 74, 96–104. https://doi.org/10.1016/j.jtice.2017.01.016.
Karagöz, S., Tay, T., Ucar, S., & Erdem, M. (2008). Activated carbons from waste biomass by sulfuric acid activation and their use on methylene blue adsorption. Bioresource Technology, 99(14), 6214–6222. https://doi.org/10.1016/j.biortech.2007.12.019.
Lagergren, S. K. (1898). About the theory of so-called adsorption of soluble substances. Sven. Vetenskapsakad. Handingarl, 24, 1–39. https://doi.org/10.1021/ja02242a004.
Langmuir, I. (1918). The adsorption of gases on plane surfaces of glass, mica and platinum. The Journal of the American Chemical Society, 40(9), 1361–1403.
Lawagon, C. P., Amon, R. E. C., Lawagon, C. P., & Amon, R. E. C. (2019). Magnetic rice husk ash 'cleanser' as efficient methylene blue adsorbent. Environmental Engineering Research, 25(5), 685–692. https://doi.org/10.4491/eer.2019.287.
Li, Y., Meas, A., Shan, S., Yang, R., & Gai, X. (2016). Production and optimization of bamboo hydrochars for adsorption of Congo red and 2-naphthol. Bioresource Technology, 207, 379–386. https://doi.org/10.1016/j.biortech.2016.02.012.
Li, Y., Tsend, N., Li, T., Liu, H., Yang, R., Gai, X., & Shan, S. (2019). Microwave assisted hydrothermal preparation of rice straw hydrochars for adsorption of organics and heavy metals. Bioresource Technology, 273, 136–143. https://doi.org/10.1016/j.biortech.2018.10.056.
Lima, H. H., Maniezzo, R. S., Llop, M. E., Kupfer, V. L., Arroyo, P. A., Guilherme, M. R., & Rinaldi, A. W. (2019). Synthesis and characterization of pecan nutshell-based adsorbent with high specific area and high methylene blue adsorption capacity. Journal of Molecular Liquids, 276, 570–576. https://doi.org/10.1016/j.molliq.2018.12.010.
Ling Pua, F., Sajab, M. S., Chia, C. H., Zakaria, S., Rahman, I. A., & Salit, M. S. (2013). Alkaline-treated cocoa pod husk as adsorbent for removing methylene blue from aqueous solutions. Journal of Environmental Chemical Engineering, 1(3), 460–465. https://doi.org/10.1016/j.jece.2013.06.012.
Moreira, M. A., Ciuffi, K. J., Rives, V., Vicente, M. A., Trujillano, R., Gil, A., & de Faria, E. H. (2017). Effect of chemical modification of palygorskite and sepiolite by 3-aminopropyltriethoxisilane on adsorption of cationic and anionic dyes. Applied Clay Science, 135, 394–404. https://doi.org/10.1016/j.clay.2016.10.022.
Naushad, M., Alqadami, A. A., Al Othman, Z. A., Alsohaimi, I. H., Algamdi, M. S., & Aldawsari, A. M. (2019). Adsorption kinetics, isotherm and reusability studies for the removal of cationic dye from aqueous medium using arginine modified activated carbon. Journal of Molecular Liquids, 293, 111442. https://doi.org/10.1016/j.molliq.2019.111442.
Njoku, V. O., Islam, M. A., Asif, M., & Hameed, B. H. (2014). Preparation of mesoporous activated carbon from coconut frond for the adsorption of carbofuran insecticide. Journal of Analytical and Applied Pyrolysis, 110, 172–180. https://doi.org/10.1016/j.jaap.2014.08.020.
Pathania, D., Sharma, S., & Singh, P. (2017). Removal of methylene blue by adsorption onto activated carbon developed from Ficus carica bast. Arabian Journal of Chemistry, 10, S1445–S1451. https://doi.org/10.1016/j.arabjc.2013.04.021.
Qian, W. C., Luo, X. P., Wang, X., Guo, M., & Li, B. (2018). Removal of methylene blue from aqueous solution by modified bamboo hydrochar. Ecotoxicology and Environmental Safety, 157, 300–306. https://doi.org/10.1016/j.ecoenv.2018.03.088.
Ramesh, S., Sundararaju, P., Banu, K. S. P., Karthikeyan, S., Doraiswamy, U., & Soundarapandian, K. (2019). Hydrothermal carbonization of arecanut husk biomass: fuel properties and sorption of metals. Environmental Science and Pollution Research, 26(4), 3751–3761. https://doi.org/10.1007/s11356-018-3888-8.
Rosas, J. M., Bedia, J., Rodríguez-Mirasol, J., & Cordero, T. (2009). HEMP-derived activated carbon fibers by chemical activation with phosphoric acid. Fuel, 88(1), 19–26. https://doi.org/10.1016/j.fuel.2008.08.004.
Senthilkumaar, S., Varadarajan, P. R., Porkodi, K., & Subbhuraam, C. V. (2005). Adsorption of methylene blue onto jute fiber carbon: kinetics and equilibrium studies. Journal of Colloid and Interface Science, 284(1), 78–82. https://doi.org/10.1016/j.jcis.2004.09.027.
Shi, Y., Zhang, T., Ren, H., Kruse, A., & Cui, R. (2018). Polyethylene imine modified hydrochar adsorption for chromium (VI) and nickel (II) removal from aqueous solution. Bioresource Technology, 247, 370–379. https://doi.org/10.1016/j.biortech.2017.09.107.
Sing, K. S. W., Everett, D. H., Haul, R. A. W., Moscou, L., Pierotti, R. A., Rouquerol, J., & Siemieniewska, T. (1985). Physical and biophysical chemistry division commission on colloid and surface chemistry including catalysis. Pure and Applied Chemistry, 57(4), 603–619.
Subramani, B. S., Shrihari, S., Manu, B., & Babunarayan, K. S. (2019). Evaluation of pyrolyzed areca husk as a potential adsorbent for the removal of Fe2+ ions from aqueous solutions. Journal of Environmental Management, 246, 345–354. https://doi.org/10.1016/j.jenvman.2019.04.122.
Thommes, M., Kaneko, K., Neimark, A. V., Olivier, J. P., Rodriguez-Reinoso, F., Rouquerol, J., & Sing, K. S. (2015). Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC technical report). Pure and Applied Chemistry, 87(9-10), 1051–1069.
Tran, H. N., You, S. J., & Chao, H. P. (2017). Insight into adsorption mechanism of cationic dye onto agricultural residues-derived hydrochars: negligible role of π-π interaction. Korean Journal of Chemical Engineer, 34(6), 1708–1720. https://doi.org/10.1007/s11814-017-0056-7.
Weber, W. J., & Morris, J. C. (1963). Kinetics of adsorption on carbon from solution. Journal of the Sanitary Engineering Division, 89(2), 31–60.
Yu, X. Y., Luo, T., Zhang, Y. X., Jia, Y., Zhu, B. J., Fu, X. C., & Huang, X. J. (2011). Adsorption of lead (II) on O2-plasma-oxidized multiwalled carbon nanotubes: thermodynamics, kinetics, and desorption. ACS Applied Materials and Interfaces, 3(7), 2585–2593. https://doi.org/10.1021/am2004202.
Yusriah, L., Sapuan, S. M., Zainudin, E. S., & Mariatti, M. (2014). Characterization of physical, mechanical, thermal and morphological properties of agro-waste betel nut (Areca catechu) husk fibre. Journal of Cleaner Production, 72, 174–180. https://doi.org/10.1016/j.jclepro.2014.02.025.
Zhou, N., Chen, H., Feng, Q., Yao, D., Chen, H., Wang, H., & Lu, X. (2017). Effect of phosphoric acid on the surface properties and Pb (II) adsorption mechanisms of hydrochars prepared from fresh banana peels. Journal of Cleaner Production, 165, 221–230. https://doi.org/10.1016/j.jhazmat.2019.12158.
Zhu, X., Liu, Y., Qian, F., Zhou, C., Zhang, S., & Chen, J. (2015). Role of hydrochar properties on the porosity of hydrochar-based porous carbon for their sustainable application. ACS Sustainable Chemistry & Engineering, 3(5), 833–840. https://doi.org/10.1021/acssuschemeng.5b00153.
Funding
This research was funded by Ministry of Education, Government of Bangladesh, under advanced research in education scheme (Grant No. 37. 20. 0000. 004. 003. 020. 2016). Bangladesh Council of Scientific and Industrial Research (BCSIR) and University Teknologi MARA, Malaysia cooperated in SEM-EDX, CHNS, and BET analysis respectively.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tabassum, M., Bardhan, M., Novera, T.M. et al. NaOH-Activated Betel Nut Husk Hydrochar for Efficient Adsorption of Methylene Blue Dye. Water Air Soil Pollut 231, 398 (2020). https://doi.org/10.1007/s11270-020-04762-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-020-04762-0