Abstract
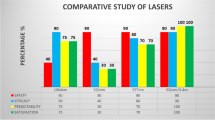
Tissue Biobanks represent an invaluable resource. Despite the majority of people supporting tissue donation, the actual rate remains low overall. Tissue discarded from surgical procedures represents a further avenue for collection for use in research. We aim to understand the information and consent requirements in a cohort of healthy, post-ophthalmic surgical subjects to optimise future tissue collection in living donors. Patients attending an ophthalmic clinic following refractive surgery for myopia (SMILE) were identified. Patient consent was implied with the completion of the provided survey. The questionnaire included gender, age range and education status. The majority of 31 subjects identified a benefit for future patients as the main motive for potential donation of discarded tissue (71%). Payment for the discarded tissue would not influence their decision in 77.4%. Explanation of the potential benefits of research was the most important information to consider before making a decision to donate. Only 12.9% of patients would have refused to include further information. Almost half of patients felt that the Biobank became the owner of tissue following donation. Current surgical patients may be more inclined to participate in research than the general public because of a sense of duty or an increased understanding of the role of research in evolving treatment. Despite minor uncertainty about the eventual use of the tissue and data, most subjects were positive to donation of discarded ocular tissue and de-identified information. Consent and education processes should be revised within an ophthalmic practice to minimise future patient anxiety.
Similar content being viewed by others
References
Banks E, Herbert N, Mather T, Rogers K, Jorm L (2012) Characteristics of Australian cohort study participants who do and do not take up an additional invitation to join a long-term biobank: The 45 and Up Study. BMC Res Notes 27(5):655. https://doi.org/10.1186/1756-0500-5-655
Bhandari V, Ganesh S, Brar S, Pandey R (2016) Application of the SMILE-derived glued lenticule patch graft in microperforations and partial-thickness corneal defects. Cornea 35(3):408–412
Capron AM, Delmonico FL, Dominguez-Gil B, Martin DE, Danovitch GM, Chapman J (2016) Statement of the declaration of istanbul custodian group regarding payments to families of deceased organ donors. Transplantation 100:2006–2009
Caulfield T, Burningham S, Joly Y, Master Z, Shabani M, Borry P et al (2014) A review of the key issues associated with the commercialization of biobanks. J Law Biosci 1(1):94–110
Caulfield T, Murdoch B (2017) Genes, cells, and biobanks: yes, there's still a consent problem. PLoS Biol 15:e2002654
Clark G, Lipworth W, Bokey L, Little JM, Kerridge IH (2006) An empirical study of tissue banking in Australia: Navigating regulatory and ethical challenges. J Law Med 1:102–109
Cynowiec J, Kim J, Qazi YA (2009) Incentivizing living organ donation. Curr Opin Organ Trans 14:201–205
Edwards T, Cadigan RJ, Evans JP, Henderson G (2014) Biobanks containing clinical specimens: defining characteristics, policies, and practices. Clin Biochem 47:245–251
Emery J, Kumar S, Smith H (1998) Patient understanding of genetic principles and their expectations of genetic services with the NHS: A qualitative study. Commun Genet 1:78–83
Fradgley EA, Chong SE, Cox ME, Paul CL, Gedye C (2018) Enlisting the willing: A study of healthcare professional-initiated and opt-in biobanking consent reveals improvement opportunities throughout the registration process. Eur J Cancer 89:36e41
Goddard KA, Smith KS, Chen C, McMullen C, Johnson C (2009) Biobank recruitment: motivations for nonparticipation. Biopreserv Biobank 7:119–121
Hewitt RE (2011) Biobanking: the foundation of personalized medicine. Curr Opin Oncol 23:112–119
Hoeyer K, Lynoe N (2006) Motivating donors to genetic research? Anthropological reasons to rethink the role of informed consent. Med Health Care Philos 9:13–23
Johnsson L, Helgesson G, Rafnar T, Halldosdottir I, Chia K-S, Eriksson S, Hansson MG (2010) Hypothetical and factual willingness to participate in biobank research. Eur J Hum Genet 18:1261–1264
Kettis-Lindblad A, Ring L, Viberth E, Hansson MG (2005) Genetic research and donation of tissue samples to biobanks. What do potential sample donors in the Swedish general public think? Eur J Publ Health 16:433–440
Liu YC, Williams GP, George BL, Soh YQ, Seah XY, Peh GSL, Yam GHF, Mehta JS (2017) Corneal lenticule storage before reimplantation. Mol Vis 23:753–764
Master Z, Nelson E, Murdoch B, Caulfield T (2012) Biobanks, consent and claims of consensus. Nat Methods 9:885–888
McKibbin M, Ahmed M, Allsop MJ, Downey L, Gale R, Grant HL, Potrata B, Willis TA, Hewison J (2014) Current understanding of genetics and genetic testing and information needs and preferences of adults with inherited retinal disease. Eur J Hum Genet 22(9):1058–1062
Murphy J, Scott J, Kaufman D, Geller G, LeRoy L, Hudson K (2008) Public expectations for return of results from large-cohort genetic research. Am J Bioeth 8(11):36–43
National Health and Medical Research Council (NHMRC) (2010) Biobanks Information Paper 2010, NHMRC, Canberra
Otlowski M (2009) Principles and Practice in Biobank Governance. In: Kaye J, Stranger M (eds) 5. Ashgate, Surrey, pp 79–92
Parker LS (2006) Best laid plans for offering results go awry. The American Journal of Bioethics 6:22–23
Patel A (2011) Tissue banking for research—bench to bedside and back—myth, reality or fast fading reality at the dawn of a personalised healthcare era. Cell Tissue Bank 12:19–21
Rahm AK, Wrenn M, Carroll NM, Feigelson HS (2013) Biobanking for research: a survey of patient population attitudes and understanding. J Commun Genet 4:445–450
Reichardt JO (2010) Donor compensation: an ethical imperative! Trans Proc 42:124–125
Riau AK, Lui YC, Yam GHF, Mehta JS (2020) Stromal keratophakia: corneal inlay implantation. Prog Retinal Eye Res 75:100780
Riegman PHJ, van Veen E-B (2011) Biobanking residual tissues. Hum Genet 130:357–368
Riordan F, Papoutsi C, Reed JE, Marston C, Bell D, Majeed A (2015) Patient and public attitudes towards informed consent models and levels of awareness of electronic health records in the UK. Int J Med Inform 84:237–247
Simon CM, L’heureux J, Murray JC, Winokur P, Weiner G, Newbury E et al (2011) Active choice but not too active: public perspectives on biobank consent models. Genet Med 13:821–831
Skeie JM, Tsand SH, Vande Zande R, Fickbohm MM, Shah SS, Vallone JG, Mahajan VB (2014) A Biorepository for Ophthalmic Surgical Specimens. Proteomics Clin Appl 8:209–217
The National Health and Medical Research Council, the Australian Research Council and Universities Australia. National Statement on Ethical Conduct in Human Research 2007 (Updated 2018). Commonwealth of Australia, Canberra
Tupasela A, Sihvo S, Snell K, Jallinoja P, Aro AR, Hemminki E (2010) Attitudes towards biomedical use of tissue sample collections, consent, and biobanks among Finns. Scand J Publ Health 38:46–52
De Vries RG, Tomlinson T, Kim HM, Krenz C, Haggerty D, Ryan KA, Kim SYH (2017) Understanding the public’s reservations about broad consent and study-by-study consent for donations to a biobank: results of a national Survey. PLoS ONE 11:e0159113
Williams GP, Wu B, Liu YC, Teo E, Nyein CL, Peh G, Tan DT, Mehta JS (2018) Hyperopic refractive correction by LASIK, SMILE or lenticule reimplantation in a non-human primate model. PLoS ONE 13(3):e0194209
Willison D, Schwartz L, Abelson J (2007) Alternatives to project-specific consent for access to personal information for health research: What is the opinion of the Canadian public? J Am Med Inform Assoc 14(6):706–712
Willison DJ, Steeves V, Charles C, Schwartz L, Ranford J, Agarwal G, Cheng J, Thabane L (2009) Consent for use of personal information for health research: do people with potentially stigmatizing health conditions and the general public differ in their opinions? BMC Med Ethics 24(10):10
Womack C, Gray NM (2009) Banking human tissue for research: vision to reality. Cell Tissue Bank 10:267–270
Funding
Gerard Sutton receives support from Sydnovate, Sydney Medical School, University of Sydney for non-related research. No funding was provided for this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ghabcha, M., Sutton, G., Petsoglou, C. et al. Donation of discarded ocular tissue in patients undergoing SMILE laser refractive surgery: developing appropriate guidelines. Cell Tissue Bank 21, 605–613 (2020). https://doi.org/10.1007/s10561-020-09850-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-020-09850-3