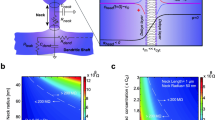
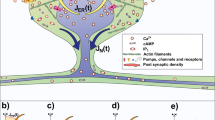
Abstract
Understanding the relationship between shape and function of dendritic spines is an elusive topic. Several modelling approaches have been used to investigate the interplay between spine geometry, calcium diffusion and electric signalling. We here use a second order finite element method to solve the Poisson–Nernst–Planck equations and describe electrodiffusion in dendritic spines. With this, we obtain relationships between dendritic geometry and calcic as well as electric responses to synaptic events. Our findings support the hypothesis that spine geometry plays a role shaping the electrical responses to synaptic events. Our method was also able to reveal the fine scale distribution of calcium in spines with irregular shapes.











Similar content being viewed by others
References
Babuska I, Suri M (1994) The p and h-p version of the finite element method, basic principles and properties. SIAM Rev 36(4):578–632
Belhamadia Y, Grenier J (2019) Modeling and simulation of hypothermia effects on cardiac electrical dynamics. PLoS ONE 14(5):1–23
Bourne JN, Harris KM (2008) Balancing structure and function at hippocampal dendritic spines. Annu Rev Neurosci 31:47–67
Brenner SC, Scott LR (2008) The mathematical theory of finite element methods. Springer, New York
Dione I, Deteix J, Briffard T, Chamberland E, Doyon N (2016) Improved simulation of electrodiffusion in the node of ranvier by mesh adaptation. PLoS ONE 11(8):2624–1635
García-López P, García-Marín V, Freire M (2007) The discovery of dendritic spines by Cajal in 1888 and its relevance in the present neuroscience. Prog Neurobiol 83:110–30
GIREF (2019) GIREF homepage. https://giref.ulaval.ca/. Accessed 10 Sept 2019
Hering H, Sheng M (2001) Dentritic spines: structure, dynamics and regulation. Nat Rev Neurosci 2:880–888
Hille B (1992) Ionic Channels of excitable membranes, 2nd edn. Sinauer Associates, Sunderland
Holcman D, Yuste R (2015) The new nanophysiology: regulation of ionic flow in neuronal subcompartments. Nat Rev 16:685–692
Holmes WR (1990) Is the function of dendritic spines to concentrate calcium? Brain Res 519:338–342
Holtmaat A, Svoboda K (2009) Experience-dependent structural synaptic plasticity in the mammalian brain. Nat Rev Neurosci 10:647–658
Jahr CE, Stevens CF (1993) Calcium permeability of the n-methyl-d-aspartate receptor channel in hippocampal neurons in culture. Proc Natl Acad Sci USA 90(24):11573–7
Jones E, Powell T (1969) Morphological variations in the dendritic spines of the neocortex. J Cell Sci 5(2):509–529
Koch C (1999) Biophysics of computation, information processing in single neuron. Oxford University Press, Oxford
Lopreore CL, Bartol TM, Coggan JS, Keller DX, Sosinsky GE, Ellisman MH, Sejnowski TJ (2008) Computational modeling of three-dimensional electrodiffusion in biological systems: application to the node of Ranvier. Biophys J 95(6):2624–2635
Mori Y (2009) From three-dimensional electrophysiology to the cable model: an asymptotic study. ArXiv e-prints
Neher E, Augustine GJ (1992) Calcium gradients and buffers in bovine chromaffin cells. J Physiol 450:273–301
Pannese E (2015) Neurocytology: fine structure of neurons, nerve processes, and neuroglial cells. Springer, Berlin
Pods J, Schonke J, Bastian P (2013) Electrodiffusion models of neurons and extracellular space using the Poisson–Nernst–Planck equations—numerical simulation of the intra- and extracellular potential for an axon model. Biophys J 105(1):242–254
Purves D (1997) Neuroscience. Sinauer Associates, Sunderland. Includes bibliographical references and index
Robinson R, Stokes R (2002) Electrolyte solutions. Dover Publications, Mineola
Sala F, Hernandez-Cruz A (1990) Calcium diffusion modeling in a spherical neuron relevance of buffering properties. Biophys J 57:313–324
Schiegg A, Gerstner W, Ritz R, van Hemmen JL (1995) Intracellular Ca2+ stores can account for the time course of LTP induction: a model of Ca2+ dynamics in dendritic spines. J Neurophysiol 74(3):1046–1055
Schneggenburger R, Zhou Z, Konnerth A, Neher E (1993) Fractional contribution of calcium to the cation current through glutamate receptor channels. Neuron 11(1):133–143
Sorra KE, Fiala JC, Harris KM (1998) Critical assessment of the involvement of perforations, spinules, and spine branching in hippocampal synapse formation. J Comp Neurol 398:225–240
Volfovsky N, Parnas H, Segal M, Korkotian E (1999) Geometry of dendritic spines affects calcium dynamics in hippocampal neurons: theory and experiments. J Neurophysiol 82(1):450–462
Wagner J, Keizer J (1994) Effects of rapid buffers on Ca2+ diffusion and Ca2+ oscillations. Biophys J 67:447–456
Yuste R (2010) Dendritic spines, vol 24. The MIT Press, Cambridge
Yuste R (2013) Electrical compartmentalization in dendritic spines. Annu Rev Neurosci 36:429–449
Yuste R, Bonhoeffer T (2001) Morphological changes in dendritic spines associated with long-term synaptic plasticity. Annu Rev Neurosci 24:1071–89
Yuste R, Denk W (1995) Dendritic spines as basic functional units of neuronal integration. Nature 375:682–684
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
1.1 The multicompartmental model used for validation
Our multicompartemental model consisted of the sections: the spine head, the spine neck and the dendritic shaft. Membrane electric potential and free calcium concentration were obtained in each compartment by solving ordinary differential equations. Below are the equations solved in each compartment for the membrane potential given a synaptic sodium and calcium currents \(\mathrm {I}_{\mathrm {syn,Na}}\) and \(\mathrm {I}_{\mathrm {syn,Ca}}\).
Meanwhile the concentration of free calcium in each compartment evolves according to
where \(D=\mathrm {D}^{\mathrm{app}}_{\mathrm {Ca}}\cdot \mathrm {D}_{\mathrm {Ca}}\) is the effective diffusion coefficient of calcium ion. The electrical capacities of the compartments are given by
with the surfaces of the compartments being respectively given by
The electrical resistances of the neck and the dendrites are respectively given by
with the cytosol resistivity given by
where the sum is taken over all ionic concentrations, \(z_i\) is the valence of the ith specie, \(D_i\) is the diffusion coefficient and \(C_i\) is the resting concentration of the ith specie. The calcium released from store in the head and the dendrite are obtained by
where the fraction of store calcium concentration and of open channels are given by solving the following ODE’s
and
1.2 Multicompartment model used to determine the boundary condition of electric potential
As stated in this Appendix, the Dirichlet boundary condition for the electric potential at the open ends of the dendritic section were determined by a coarser multicompartment model. This model is similar to the one described in the previous subsection. However, the present model relies on more neural compartments, namely: one compartment for the spine head, one compartment for the spine neck, six compartments for the dentritic shaft and one compartment for the soma. The dynamic of membrane potential in the ith compartment (\(V_i\)) is given by
where \(\mathrm {Cap}\) is the membrane electric capacitance per unit area as determined in the previous subsection, \(\mathrm {Surf}_\mathrm{i}\) is the surface area of the ith compartment, \(\mathrm {I}_{\mathrm{i,mem}}\) is the transmembrane current in the ith compartment and \(\mathrm {I}_{\mathrm{i,long}}\) is the longitudinal current between compartment i and adjacent compartments. The transmembrane current is given by
where the first term describes the current through leak channels while the second describes the synaptic current.
The longitudinal current is given by
where \(\mathrm {CSA}_{\mathrm{i,k}}\) is the surface area of the common cross-section between adjacent compartments i and k, if the compartments i and k are not adjacent, then this is set to 0. The value of \(\mathrm {Res}_{\mathrm{i,k}}\) is defined only if the compartments i and k are adjacent and stands for the electrical resistance between compartments i and k. If i and k are adjacent dendritic compartments, we have
where \(\mathrm {R}^{\mathrm{cyt}}\) is the cytoplasmic resistivity defined in the previous subsection, \(\mathrm {L}^{\mathrm{dend}}\) is the length of the dendrite compartment and \(\mathrm {r}^{\mathrm{dend}}\) is the radius of the dendrite. The resistance between the soma and the adjacent dendritic compartment is given by
The resistance between the spine neck compartment and the adjacent dendritic dendrite compartment is given by
where \(\mathrm {L}^{\mathrm{neck}}\) is the length of the spine neck and \(\mathrm {r}^{\mathrm{neck}}\) is the radius of the spine neck. Finally, the resistance between the spine head and spine neck is also given by
Though not used, we also computed the dynamics of calcium concentration according to
where \(\mathrm {C}_\mathrm {i}\) is the calcium concentration in the ith compartment, \(\mathrm {I}_\mathrm{i}^{\mathrm {Ca,syn}}\) is the synaptic current in the ith compartment \(\mathrm {J}^{\mathrm {ext}}_\mathrm {i}\) is the \(\mathrm {Ca}^{2+}\) extrusion flux in the ith compartment and \(\mathrm {J}_\mathrm {i}^{\mathrm {Ca,long}}\) describes the longitudinal flux of \(\mathrm {Ca}^{2+}\) between adjacent compartments. The latter is given by
where \(\mathrm {\mathrm{J}_{\mathrm{i,k}}^{\mathrm{Ca}}}\) is the flux of \(\mathrm {Ca}^{2+}\) from compartment k to compartment i equal to zero if the compartments i and k are not adjacent and if the compartments are adjacent, this is given by
where \(\mathrm {dist}_{\mathrm{i,k}}\) is the distance between the center of the compartments (Table 3).
1.2.1 Variational formulation, finite element approximation and time discretization
The first two equations of (33) are respectively multiplied by test functions \(\phi \) and \(\varphi \) and integrated over the domain \(\varOmega \):
Applying integration by parts, we obtain
The functional space of the ionic concentrations and the electric potential is denoted by \({\mathscr {C}}\) which is defined as:
Hence we look for \(C_k \in H^1(\varOmega )\) such that \(C_k-C^0_k \in \mathrm {{\mathscr {C}}}\) for \(k=1,\ldots ,4\) and also \(V \in H^1(\varOmega )\) such that \(V-V_d \in \mathrm {{\mathscr {C}}}\). So we obtain the weak formulation
The domain \((\varOmega )\) is partitioned into a finite mesh \({\mathscr {T}}:\left\{ G\right\} \) of simplicial \({\mathscr {G}}\). We represent the unknown functions \(C_k\) and V and the test functions \(\phi \) and \(\varphi \) by piecewise polynomials on this mesh. Finite dimensional approximation space \({\mathscr {H}}\) of piecewise polynomials of degree n is considered on the generated mesh \({\mathscr {T}}\):
We therefore look for \(C_{k}\in H^1_n\) such that \(C_k-C_k^0\in \mathrm {{\mathscr {H}}}\) for all \(k=1,\ldots ,4\) and \(V \in H_n^1\) such that \(V-V_d \in \mathrm {{\mathscr {H}}}\) such that:
To discretize the time derivative in the first equation of equation (62), we employ the second order Backward Difference Formula method (BDF2) also known as the Gear time-stepping scheme.
1.2.2 Elementary variational formulation
The elementary variational formulation is obtained by integrating over an element K. We obtain
Ritz method (Brenner and Scott 2008) is applied on each element K by letting
where \(N_{i}^{K}\) and \(M_{i}^{K}\) are the interpolation functions on the element K for \(i=1,2,\ldots n_{d}\). Substituting the two equations above into Equations (65) and (66) and taking successively \(\phi =\phi _{i}^{K}\) and \(\varphi =\varphi _{i}^{K}\), we obtain
and
where \(Z=\mathrm {Ca}^{{2+}}_{\mathrm{store}}\) and it is obtained by solving Eqs. (48) and (50) using an implicit linear multi-step method (backward difference formula). From this, we obtain the elementary system
from which we finally obtain the following algebraic system
Observe that this system is non linear since there is a coupling term in (69). A Newton–Raphson method is used to solve the problem at time step \(r+1\). We solve iteratively the first order linearisation of the system.
to obtain \(U^{r+1}\) when the correction \(\delta _{U}\) is small enough. We resolve the issue with the required two initial values in Eqs. (67) and (67) by introducing \(\delta _{C}\), \(\delta _{V}\) (Dione et al. 2016) and neglecting the second order terms in the equations as
The numerical computations were implemented using finite element library MEF++ software (Version 5) developed at the laboratory of the Groupe Interdisciplinaire de Recherche en Éléments Finis (GIREF) at the department of mathematics and statistic, Université Laval.
The boundary of the domain consist of points, curves and surfaces which are grouped as a geometric entities on which we define the boundary conditions using iMEF++. iMEF++ is a software developed by GIREF.
Rights and permissions
About this article
Cite this article
Boahen, F., Doyon, N. Modelling dendritic spines with the finite element method, investigating the impact of geometry on electric and calcic responses. J. Math. Biol. 81, 517–547 (2020). https://doi.org/10.1007/s00285-020-01517-7
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00285-020-01517-7
Keywords
- Electrodiffusion
- Poisson–Nernst–Planck equations
- Dendritic spine
- Calcium signalling
- Finite element method