Abstract
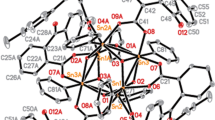
The reaction of perfluorinated three-mercury anticrown (o-C6F4Hg)3 (1) with 1,2-dichloroethane (DCE) affords the complex {[(o-C6F4Hg)3]2(ClCH2CH2Cl)}(2) containing two anticrown molecules per DCE molecule. According to the X-ray diffraction data, the DCE molecule in complex 2 acts as a bridge between molecules 1, each DCE chlorine atom being coordinated to a single mercury center of the neighboring anticrown molecules.
Similar content being viewed by others
References
V. B. Shur, I. A. Tikhonova, in Encyclopedia of Supramolecular Chemistry, Eds J. L. Atwood, J. W. Steed, Marcel Dekker, Inc., New York, 2004, p. 68.
T. J. Wedge, M. F. Hawthorne, Coord. Chem. Rev., 2003, 240, 111.
V. B. Shur, I. A. Tikhonova, Russ. Chem. Bull., 2003, 52, 2539.
M. R. Haneline, R. E. Taylor, F. P. Gabbaï, Chem. Eur. J., 2003, 21, 5188.
T. J. Taylor, C. N. Burress, F. P. Gabbaï, Organometallics, 2007, 26, 5252.
J. D. Wuest, Acc. Chem. Res., 1999, 32, 81.
M. F. Hawthorne, Z. Zheng, Acc. Chem. Res., 1997, 30, 267.
I. A. Tikhonova, D. A. Gribanyov, K. I. Tugashov, F. M. Dolgushin, A. S. Peregudov, D. Yu. Antonov, V. I. Rosenberg, V. B. Shur, J. Organomet. Chem., 2010, 695, 1949.
I. A. Tikhonova, D. A. Gribanyov, K. I. Tugashov, F. M. Dolgushin, A. F. Smol’yakov, A. S. Peregudov, Z. S. Klemenkova, V. B. Shur, ARKIVOC, 2011, No. 8, 172.
A. S. Filatov, A. K. Greene, E. A. Jackson, L. T. Scott, M. A. Petrukhina, J. Organomet. Chem., 2011, 696, 2877.
M. Fleischmann, C. Heindl, M. Seidl, G. Balázs, A. V. Virovets, E. V. Peresypkina, M. Tsunoda, F. P. Gabbaï, M. Scheer, Angew. Chem., Int. Ed., 2012, 51, 9918.
K. I. Tugashov, D. A. Gribanyov, F. M. Dolgushin, A. F. Smol’yakov, A. S. Peregudov, M. Kh. Minacheva, B. N. Strunin, I. A. Tikhonova, V. B. Shur, J. Organomet. Chem., 2013, 747, 167.
S. P. Fischer, E. W. Reinheimer, J. Chem. Crystallogr., 2013, 43, 478.
S. P. Fischer, E. W. Reinheimer, J. Chem. Crystallogr., 2014, 44, 123.
R. Castañeda, S. Draguta, A. Yakovenko, M. Fonari, T. Timofeeva, Acta Cryst., Sect. E, 2014, 70, m164.
A. Muñoz-Castro, Phys. Chem. Chem. Phys., 2014, 16, 7578.
M. Ponce-Vargas, A. Muñoz-Castro, J. Phys. Chem. C, 2014, 118, 28244.
H. Schmidbaur, A. Schier, Organometallics, 2015, 34, 2048.
S. P. Fisher, R. H. Groeneman, E. W. Reinheimer, J. Coord. Chem., 2015, 68, 3589.
K. I. Tugashov, D. A. Gribanyov, F. M. Dolgushin, A. F. Smol’yakov, A. S. Peregudov, I. A. Tikhonova, V. B. Shur, Organometallics, 2015, 34, 1530.
K. I. Tugashov, D. A. Gribanyov, F. M. Dolgushin, A. F. Smol’yakov, A. S. Peregudov, M. Kh. Minacheva, I. A. Tikhonova, V. B. Shur, Organometallics, 2016, 35, 2197.
M. Tsunoda, M. Fleischmann, J. S. Jones, N. Bhuvanesh, M. Scheer, F. P. Gabbaï, Dalton Trans., 2016, 45, 5045.
T. Lasanta, J. M. López-de-Luzuriaga, M. Monge, M. E. Olmos, D. Pascual, Dalton Trans., 2016, 45, 6334.
K. I. Tugashov, D. A. Gribanyov, F. M. Dolgushin, A. F. Smol’yakov, A. S. Peregudov, Z. S. Klemenkova, O. V. Matvienko, I. A. Tikhonova, V. B. Shur, Organometallics, 2017, 36, 2437.
K. I. Tugashov, S. M. Yunusov, E. S. Kalyuzhnaya, F. M. Dolgushin, A. A. Yakovenko, I. A. Tikhonova, V. B. Shur, Russ. Chem. Bull., 2018, 67, 45.
N. A. Semenov, D. E. Gorbunov, M. V. Shakhova, G. E. Salnikov, I. Y. Bagryanskaya, V. V. Korolev, J. Beckmann, N. P. Gritsan, A. V. Zibarev, Chem. Eur. J., 2018, 24, 12983.
K. I. Tugashov, D. A. Gribanyov, F. M. Dolgushin, A. F. Smol’yakov, A. S. Peregudov, Z. S. Klemenkova, I. G. Barakovskaya, I. A. Tikhonova, V. B. Shur, Organometallics, 2019, 38, 2910.
P. Sartory, A. Golloch, Chem. Ber., 1968, 101, 2004.
M. C. Ball, D. S. Brown, A. G. Massey, D. A. Wickens, J. Organomet. Chem., 1981, 206, 265.
K. I. Tugashov, I. A. Tikhonova, F. M. Dolgushin, A. Yu. Volkonskii, I. G. Barakovskaya, A. G. Buyanovskaya, V. B. Shur, Tez. dokl. Vseross. konf “Khimiya ftora” posvyashchennoi 100-letiyu so dnya rozhdeniya akademika I. L. Knunyantsa [All-Russian Conference “Fluorine Chemistry” devoted to the 100th Anniversary of Academician I. L. Knunyants], (June 5–9, 2006, Moscow), Moscow, 2006, P-75 (in Russian).
M. Fleischmann, J. S. Jones, F. P. Gabbaï, M. Scheer, Chem. Sci., 2015, 6, 132.
O. Elbjeirami, C. N. Burress, F. P. Gabbaï, M. A. Omary, J. Phys. Chem. C, 2007, 111, 9522.
A. J. Canty, G. B. Deacon, Inorg. Chim. Acta, 1980, 45, L 225.
P. Pyykkö, M. Straka, Phys. Chem. Chem. Phys., 2000, 2, 2489.
S. S. Batsanov, Russ. J. Inorg. Chem., 1991, 36, 1694.
Yu. K. Grishin, Yu. A. Strelenko, L. A. Margulis, Yu. A. Ustynyuk, L. S. Golovchenko, A. S. Peregudov, D. N. Kravtsov, Dokl. Chem., 1979, 249, 1008.
APEX II software package; Bruker AXS, Inc.: 5465, East Cheryl Parkway, Madison, WI 5317, 2005.
G. M. Sheldrick, Acta Crystallogr., Sect. C, 2015, C71, 3.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Academician of the Russian Academy of Sciences I. L. Eremenko on the occasion of his 70th birthday.
This work was financially supported by the Russian Foundation for Basic Research (Project No. 19-03-00875). The X-ray diffraction studies, spectroscopic measurements, and elemental analyses were performed with the financial support from the Ministry of Science and Higher Education of the Russian Federation using equipment of the Center for Molecular Composition Studies of the A. N. Nesmeyanov Institute of Organoelement Compounds of the Russian Academy of Sciences.
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 6, pp. 1092–1095, June, 2020.
Rights and permissions
About this article
Cite this article
Tugashov, K.I., Yunusov, S.M., Kalyuzhnaya, E.S. et al. Coordination chemistry of anticrowns. Synthesis and structure of the binuclear complex of perfluorinated three-mercury anticrown (o-F6Hg)3 with 1,2-dichloroethane. Russ Chem Bull 69, 1092–1095 (2020). https://doi.org/10.1007/s11172-020-2872-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-020-2872-x