Abstract
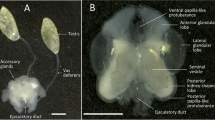
Myotis levis (yellowish myotis) is a small Neotropical insectivorous vespertilionid bat that provides valuable ecosystem services, such as control of disease vectors and agricultural pests. Aiming to describe the fluctuations of the reproductive organs throughout the year, the gonads and epididymis from 124 adult bats were histologically evaluated. These animals were captured in Santuário do Caraça, Minas Gerais, Brazil. After the initial screening, six bats per reproductive stage (in a representative month) had specific organs harvested for further investigation. The gonads, epididymis, accessory sex gland and brown adipose tissue were collected for biometric analyses. Furthermore, yellowish myotis testis was evaluated through histomorphometric and molecular assays, whereas blood samples were collected for hormonal analyses. The data were compared among the reproductive stages and correlated with rainfall distribution. As a result, we demonstrated that yellowish myotis presented a seasonal reproduction showing testis regression and rest, resembling the pattern exhibited by temperate-zone vespertilionid bats. During the Mature stage, after the peak of rainfall distribution, yellowish myotis testicles were fully developed for gamete production and maximum testosterone synthesis. These findings indicate a significant influence of this environmental factor on yellowish myotis reproduction. Following that, the accessory sex gland, brown adipose tissue and epididymis weights increased in the Regressed stage. The epididymis sperm storage occurred for at least 8 months and was observed in the Regressed, Rest and beginning of the Maturing stage. This reproductive fluctuation is interesting because the reactivation of the gonads coincided with the least amount of sperm in the epididymis.










Similar content being viewed by others
References
Abreu ACL, Palú L (2008) RPPN Santuário do Caraça. In: Vieira MCW (ed) RPPN: Reserva Particular do Patrimônio Natural em destaque na conservação da biodiversidade da Mata Atlântica. Conselho Nacional da Reserva da Biosfera da Mata Atlântica, São Paulo, pp 60–63
Anthony ELP (1988) Age determination in bats. In: Kunz TH (ed) Ecological and behavioral methods for the study of bats. Smithsonian Institution Press, Washington DC, pp 47–57
Araújo RA, Amaro BD, Talamoni SA, Godinho HP (2013) Seasonal reproduction of yellowish myotis, Myotis levis (Chiroptera: Vespertilionidae), from a Neotropical highland. J Morphol 274:1230–1238. https://doi.org/10.1002/jmor.20175
Barbour RW, Davis WH (1969) Bats of America. University Press of Kentucky, Lexington
Beguelini MR, Góes RM, Taboga SR, Morielle-Versute E (2013) Two periods of total testicular regression are peculiar events of the annual reproductive cycle of the black Myotis bat, Myotis nigricans (Chiroptera: Vespertilionidae). Reprod Fertil Dev 26:834–846. https://doi.org/10.1071/RD13109
Bernard RTF, Bojarski C, Millar RP (1991) Patterns of Leydig cell and LH gonadotroph activity, and plasma testosterone concentrations in the seasonally reproducing Schreibers’ long-fingered bat (Miniopterus schreibersii). J Reprod Fertil 91:479–492. https://doi.org/10.1530/jrf.0.0910479
Bernard RTF, Happold DCD, Happold M (1997) Sperm storage in a seasonally reproducing African vespertilionid, the banana bat (Pipistrellus nanus) from Malawi. J Zool 241:161–174. https://doi.org/10.1111/j.1469-7998.1997.tb05506.x
Clutton-Brock TH, Albon SD, Guinness FE (1989) Fitness costs of gestation and lactation in wild mammals. Nature 337:260–262. https://doi.org/10.1038/337260a0
Crichton EG (2000) Sperm storage and fertilization. In: Crichton EG, Krutzsch PH (eds) Reproductive biology of bats. Academic Press, San Diego, pp 295–320
Crichton EG, Krutzsch PH (2000) Reproductive biology of bats. Academic Press, San Diego
Cumming GS, Bernard RTF (1997) Rainfall, food abundance and timing of parturition in African bats. Oecologia 111:309–317. https://doi.org/10.1007/s004420050240
Dobrinski I, Ogawa T, Avarbock MR, Brinster RL (2001) Effect of the GnRH-agonist leuprolide on colonization of recipient testes by donor spermatogonial stem cells after transplantation in mice. Tissue Cell 33:200–207. https://doi.org/10.1054/tice.2001.0177
Duarte APG, Talamoni SA (2010) Reproduction of the large fruit-eating bat Artibeus lituratus (Chiroptera: Phyllostomidae) in a Brazilian Atlantic forest area. Mamm Biol 75:320–325. https://doi.org/10.1016/j.mambio.2009.04.004
Dwyer PD (1970) Latitude and breeding season in a polyestrous species of Myotis. J Mammal 51:405–410. https://doi.org/10.2307/1378506
Eisenberg JF, Redford KH (1999) Mammals of the Neotropics. . The Central Neotropics: Ecuador, Peru, Bolivia, Brazil. University of Chicago Press, Chicago
Encarnação JA, Dietz M, Kierdorf U (2004) Reproductive condition and activity pattern of male Daubenton's bats (Myotis daubentonii) in the summer habitat. Mamm Biol 69:163–172. https://doi.org/10.1078/1616-5047-00131
Entwistle AC, Racey PA, Speakman JR (1998) The reproductive cycle and determination of sexual maturity in male brown long-eared bats, Plecotus auritus (Chiroptera: Vespertilionidae). J Zool (Lond) 244:63–70. https://doi.org/10.1111/j.1469-7998.1998.tb00007.x
Farias TO, Notini AA, Talamoni SA, Godinho HP (2015) Testis Morphometry and stages of the seminiferous epithelium cycle in an epididymal sperm-storing Neotropical Vespertilionid, Myotis levis (Chiroptera). Anat Histol Embryol 44:361–369. https://doi.org/10.1111/ahe.12148
Fleming TH, Geiselman C, Kress WJ (2009) The evolution of bat pollination: a phylogenetic perspective. Ann Bot 104:1017–1043. https://doi.org/10.1093/aob/mcp197
Gardner AL (2008) Mammals of South America. Vol. 1. Marsupials, Xenarthrans, Shrews, and Bats. University of Chicago Press, Chicago
Gendt K, Atanassova N, Tan KA, França LR, Parreira GG, Mckinnell C, Sharpe RM, Saunders PTK, Mason JI, Hartung S, Ivell R, Denolet E, Verhoeven G (2005) Development and function of the adult generation of Leydig cells in mice with Sertoli cell-selective or total ablation of the androgen receptor. Endocrinology 146:4117–4126. https://doi.org/10.1210/en.2005-0300
Giulietti AM, Pirani JR, Harley RM (1997) Espinhaço range region, eastern Brazil. In: Davis SD, Heywood VH, Herrera-Macbryde O, Villa-Lobos J, Hamilton AC (eds) Centres of plant diversity, a guide and strategy for their conservation. Information Press, Oxford, pp 397–404
Gustafson AW (1979) Male reproductive patterns in hibernating bats. J Reprod Fertil 56:317–331. https://doi.org/10.1530/jrf.0.0560317
Gustafson AW (1987) Changes in Leydig cell activity during the annual testicular cycle of the bat Myotis lucifugus lucifugus: histology and lipid histochemistry. Am J Anat 178:312–325. https://doi.org/10.1002/aja.1001780403
Gustafson AW, Damassa DA (1985) Annual variations in plasma sex steroid-binding protein and testosterone concentrations in the adult male little brown bat: relation to the asynchronous recrudescence of the testis and accessory reproductive organs. Biol Reprod 33:1126–1137. https://doi.org/10.1095/biolreprod33.5.1126
Gustafson AW, Shemesh M (1976) Changes in plasma testosterone levels during the annual reproductive cycle of the hibernating bat, Myotis lucifugus lucifugus with a survey of plasma testosterone levels in adult male vertebrates. Biol Reprod 15:9–24. https://doi.org/10.1093/biolreprod/15.1.9
Griswold MD (1998) The central role of Sertoli cells in spermatogenesis. Semin Cell Dev Biol 9:411–416. https://doi.org/10.1006/scdb.1998.0203
Happold DCD, Happold M (1989) Reproduction of Angola free-tailed bats (Tadarida condylura) and little free-tailed bats (Tadarida pumila) in Malawi (Central Africa) and elsewhere in Africa. Reproduction 85:133–149. https://doi.org/10.1530/jrf.0.0850133
Happold DCD, Happold M (1990) Reproductive strategies of bats in Africa. J Zool 222:557–583. https://doi.org/10.1111/j.1469-7998.1990.tb06014.x
Hess RA, França LR (2007) Spermatogenesis and cycle of the seminiferous epithelium. In: Cheng CY (ed) Molecular mechanism in spermatogenesis. Springer, New York, pp 1–15
Hosken DJ, Blackberry MA, Stewart TB, Stucki AF (1998) The male reproductive cycle of three species of Australian vespertilionid bat. J Zool 245:261–270. https://doi.org/10.1111/j.1469-7998.1998.tb00100.x
Ismail N, Morales CR (1992) Effects of vitamin a deficiency on the inter-Sertoli cell tight junctions and on the germ cell population. Microsc Res Tech 20:43–49. https://doi.org/10.1002/jemt.1070200106
Janzen DH, Schoener TW (1968) Differences in insect abundance and diversity between wetter and drier sites during a tropical dry season. Ecology 49:96–110. https://doi.org/10.2307/1933565
Johnson L, Varner DD, Tatum ME, Scrutchfield WL (1991) Season but not age affects Sertoli cell number in adult stallions. Biol Reprod 45:404–410. https://doi.org/10.1095/biolreprod45.3.404
Jones G, Jacobs DS, Kunz TH, Wilig MR, Racey PA (2009) Carpe noctem: the importance of bats as bioindicators. Endanger Species Res 8:93–115. https://doi.org/10.3354/esr00182
Krutzsch PH (1975) Reproduction of the canyon bat, Pipistrellus hesperus, in southwestern United States. Am J Anat 143:163–200. https://doi.org/10.1002/aja.1001430203
Krutzsch PH (1979) Male reproductive patterns in nonhibernating bats. J Reprod Fertil 56:333–344. https://doi.org/10.1530/jrf.0.0560333
Krutzsch PH (2000) Anatomy, physiology and Cyclicity of the male reproductive tract. In: Crichton EG, Krutzsch PH (eds) Reproductive biology of bats. Academic Press, San Diego, pp 91–155
Krutzsch PH (2009) The reproductive biology of the cave myotis (Myotis velifer). Acta Chiropterol 11:89–104. https://doi.org/10.3161/150811009x465712
Krutzsch PH, Crichton EG (1986) Reproduction of the male eastern pipistrelle, Pipistrellus subflavus, in the North-Eastern United States. J Reprod Fertil 76:91–104. https://doi.org/10.1530/jrf.0.0760091
Krutzsch PH, Wells WW (1960) Androgenic activity in the interscapular brown adipose tissue of the male hibernating bat (Myotis lucifugus). Proc Soc Exp Biol Med 105:578–581. https://doi.org/10.3181/00379727-105-26182
Kunz TH, De Torrez EB, Bauer D, Lobova T, Fleming TH (2011) Ecosystem services provided by bats. Ann N Y Acad Sci 1223:1–38. https://doi.org/10.1111/j.1749-6632.2011.06004.x
Kurohmaru M, Saruwatari T, Kimura J, Mukohyama M, Watanabe G, Taya K, Hayashi Y (2002) Seasonal changes in spermatogenesis of the Japanese lesser horseshoe bat, Rhinolophus cornutus from a morphological viewpoint. Okajimas Folia Anat Jpn 79:93–100. https://doi.org/10.2535/ofaj.79.93
Lara NLM, Costa GMJ, Avelar GF, Lacerda SMSN, Hess RA, França LR (2018) Testis physiology-overview and histology. In: Skinner MK (ed) Encyclopedia of reproduction. Academic Press, New York, pp 105–116
LaVal RK (1973) A revision of the Neotropical bats of the genus Myotis. Sci Bull Los Angeles Co Mus 15:1–54
Maine JJ, Boyles JG (2015) Bats initiate vital agroecological interactions in corn. Proc Natl Acad Sci 112:12438–12443. https://doi.org/10.1073/pnas.1505413112
Mcwilliam AN (1988) The reproductive cycle of male long-fingered bats, Miniopterus minor (Chiroptera: Vespertilionidae), in a seasonal environment of the African tropics. J Zool 216:119–129. https://doi.org/10.1111/j.1469-7998.1988.tb02419.x
Medway L (1972) Reproductive cycles of the flat-headed bats Tylonycteris pachypus and T. robustula (Chiroptera: Vespertilioninae) in a humid equatorial environment. Zool J Linnean Soc 51:33–61. https://doi.org/10.1111/j.1096-3642.1972.tb00772.x
Miller RE (1939) The reproductive cycle in male bats of the species Myotis lucifugus lucifugus and Myotis grisescens. J Morphol 64:267–295. https://doi.org/10.1002/jmor.1050640205
Miranda JMD, Bernardi IP, Passos FC (2011) Chave ilustrada para a determinação dos morcegos da Região Sul do Brasil. João MD Miranda, Curitiba
Moreira AMM, Pereira CCA (2004) Levantamento Topoclimático da RPPN Santuário do Caraça. Cad Geo 23:43–50
Myers P (1977) Patterns of reproduction of four species of vespertilionid bats in Paraguay. Univ Calif Publ Zool 107:1–41
Nowak RW (1994) Walker’s bats of the world. Johns Hopkins University Press, Baltimore
Ogawa T, Dobrinski I, Avarbock MR, Brinster RL (1998) Leuprolide, a gonadotropin-releasing hormone agonist, enhances colonization after spermatogonial transplantation into mouse testes. Tissue Cell 30:583–588. https://doi.org/10.1016/S0040-8166(98)80039-6
O’Shea TJ, Vaughan TA (1980) Ecological observations on an east African bat community. Mammalia 44:485–496. https://doi.org/10.1515/mamm.1980.44.4.485
Pearson OP, Koford MR, Pearson AK (1952) Reproduction of the lump-nosed bat (Corynorhinus rafinesquei) in California. J Mammal 33:273–320. https://doi.org/10.2307/1375769
Pfeiffer B, Mayer F (2012) Spermatogenesis, sperm storage and reproductive timing in bats. J Zool 289:77–85. https://doi.org/10.1111/j.1469-7998.2012.00970.x
Puga CC, Beguelini MR, Morielle-Versute E, Vilamaior PS, Taboga SR (2016) The effects of castration followed testosterone supplementation in prostatic complex of Artibeus planirostris (Chiroptera: Phyllostomidae). Tissue Cell 48:252–264. https://doi.org/10.1016/j.tice.2016.02.006
Racey PA (1972) Viability of bat spermatozoa after prolonged storage in the epididymis. J Reprod Fertil 28:309–311. https://doi.org/10.1530/jrf.0.0280309
Racey PA (1974) The reproductive cycle in male noctule bats, Nyctalus noctula. J Reprod Fertil 41:169–182. https://doi.org/10.1530/jrf.0.0410169
Racey PA (1979) The prolonged storage and survival of spermatozoa in Chiroptera. J Reprod Fertil 56:391–402. https://doi.org/10.1530/jrf.0.0560391
Racey PA (1982) Ecology of bat reproduction. In: Kunz TH (ed) Ecology of bats. Plenum Press, New York, pp 57–104
Racey PA, Tam WH (1974) Reproduction in male Pipistrellus pipistrellus (Mammalia: Chiroptera). J Zool 172:101–122. https://doi.org/10.1111/j.1469-7998.1974.tb04096.x
Racey PA, Entwistle AC (2000) Life-history and reproductive strategies of bats. In: Crichton EG, Krutzsch PH (eds) Reproductive biology of bats. Academic Press, San Diego, pp 363–414
Rasband WS (2014) ImageJ, U.S. National Institutes of Health, Bethesda, Maryland, USA, http://imagej.nih.gov/ij/, 1997–2014. Accessed 6 May 2019
Rautenbach IL, Kemp AC, Scholtz CH (1988) Fluctuations in availability of arthropods correlated with microchiropteran and avian predator activities. Koedoe 31:77–90. https://doi.org/10.4102/koedoe.v31i1.486
Russo D, Jones G (2015) Bats as bioindicators: an introduction. Mamm Biol 80:157–158. https://doi.org/10.1016/j.mambio.2015.03.005f
Sá AJr, Carvalho LG, Silva FF, Alves MC (2012) Application of the Köppen classification of climatic zoning in the state of Minas Gerais, Brazil. Theor Appl Climatol 108:1–7. https://doi.org/10.1007/s00704-011-0507-8
Sharifi M, Akmali V, Ghorbani R (2008) Evidence of sperm storage in Myotis capaccinii (Chiroptera: Vespertilionidae) in western Iran. Iran J Vet Med 2:47–52
Sharifi M, Ghorbani R, Akmali V (2004) Reproductive cycle in Pipistrellus kuhlii (Chiroptera, Vespertilionidae) in western Iran. Mammalia 68:323–327. https://doi.org/10.1515/mamm.2004.031
Simmons NB (2005) Order Chiroptera. In: Wilson DE, Reeder DM (eds) Mammal species of the world: a taxonomic and geographic reference. Johns Hopkins University Press, Baltimore, pp 312–529
Speakman JR, Racey PA (1986) The influence of body condition on sexual development of male brown long-eared bats (Plecotus auritus) in the wild. J Zool 210:515–525. https://doi.org/10.1111/j.1469-7998.1986.tb03653.x
Tanaka T, Kanatsu-Shinohara M, Lei Z, Rao CV, Shinohara T (2016) The luteinizing hormone-testosterone pathway regulates mouse spermatogonial stem cell self-renewal by suppressing WNT5A expression in Sertoli cells. Stem Cell Rep 7:279–291. https://doi.org/10.1016/j.stemcr.2016.07.005
Tarulli GA, Stanton PG, Lerchl A, Meachem SJ (2006) Adult Sertoli cells are not terminally differentiated in the Djungarian hamster: effect of FSH on proliferation and junction protein organization. Biol Reprod 74:798–806. https://doi.org/10.1095/biolreprod.105.050450
Van Pelt AMM, Van Dissel-Emiliani FMF, Gaemers IC, Van Der Burg MJM, Tanke HJ, Rooij DG (1995) Characteristics of a spermatogonia and preleptotene spermatocytes in the vitamin A-deficient rat testis. Biol Reprod 53:570–578. https://doi.org/10.1095/biolreprod53.3.570
Zirkin BR, Papadopoulos V (2018) Leydig cells: formation, function, and regulation. Biol Reprod 99:101–111. https://doi.org/10.1093/biolre/ioy059
Zukal J, Pikula J, Bandouchova H (2015) Bats as bioindicators of heavy metal pollution: history and prospect. Mamm Biol 80:220–227. https://doi.org/10.1016/j.mambio.2015.01.001
Weir BJ, Rowlands IW (1973) Reproductive strategies of mammals. Annu Rev Ecol Evol Syst 4:139–163
Wilson DE (1979) Reproductive patterns. In: Baker RJ, Jones JK, Carter DC (eds) Biology of bats of the New World family Phyllostomatidae, Part III. Special Publications of the Museum of Texas Tech University, Lubbock, pp 317–378
Acknowledgements
The support of the Federal University of Minas Gerais (UFMG) and the Image Acquisition and Processing Center (CAPI-ICB/UFMG) was of great importance. We thank the Brazilian Chico Mendes Institute for Biodiversity Conservation (ICMBIO) for providing the licence to capture the animals and the staff of the Reserva Particular do Patrimônio Natural Santuário do Caraça for allowing us to collect the bats. We also thank Raíssa Araújo, Pedro Viana, Mara Lívia dos Santos, Fr. Lauro Palú and Dr. Aline Abreu for their scientific, technical and logistical assistance.
Funding
We thank the Coordination for the Improvement of Higher Education Personnel (CAPES), the National Council for Scientific and Technological Development (CNPq) and the Foundation to Support Research of the State of Minas Gerais (FAPEMIG) for their financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted (Ethics Committee on Animal Use from the Federal University of Minas Gerais - CEUA document 386/2017).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 218 kb)
Rights and permissions
About this article
Cite this article
Farias, T.D.O., Figueiredo, A.F.A., Wnuk, N.T. et al. Male reproductive morphofunctional evaluation of a Neotropical sperm-storing vespertilionid bat (Myotis levis) in an environmental context. Cell Tissue Res 382, 639–656 (2020). https://doi.org/10.1007/s00441-020-03242-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-020-03242-5