Abstract
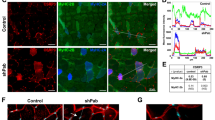
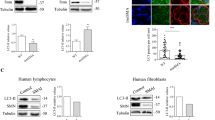
Spinal muscular atrophy (SMA) is caused by a deletion or mutation of the survival motor neuron 1 (SMN1) gene. Reduced SMN levels lead to motor neuron degeneration and muscular atrophy. SMN protein localizes to the cytoplasm and Cajal bodies. Moreover, in myofibrils from Drosophila and mice, SMN is a sarcomeric protein localized to the Z-disc. Although SMN participates in multiple functions, including the biogenesis of spliceosomal small nuclear ribonucleoproteins, its role in the sarcomere is unclear. Here, we analyzed the sarcomeric organization of SMN in human control and type I SMA skeletal myofibers. In control sarcomeres, we demonstrate that human SMN is localized to the titin-positive M-band and actin-positive I-band, and to SMN-positive granules that flanked the Z-discs. Co-immunoprecipitation assays revealed that SMN interacts with the sarcomeric protein actin, α-actinin, titin, and profilin2. In the type I SMA muscle, SMN levels were reduced, and atrophic (denervated) and hypertrophic (nondenervated) myofibers coexisted. The hypertrophied myofibers, which are potential primary targets of SMN deficiency, exhibited sites of focal or segmental alterations of the actin cytoskeleton, where the SMN immunostaining pattern was altered. Moreover, SMN was relocalized to the Z-disc in overcontracted minisarcomeres from hypertrophic myofibers. We propose that SMN could have an integrating role in the molecular components of the sarcomere. Consequently, low SMN levels might impact the normal sarcomeric architecture, resulting in the disruption of myofibrils found in SMA muscle. This primary effect might be independent of the neurogenic myopathy produced by denervation and contribute to pathophysiology of the SMA myopathy.





Similar content being viewed by others
Abbreviations
- SMN1 :
-
Survival Motor Neuron 1 gene
- SMN2 :
-
Survival Motor Neuron 2 gene
- SMN:
-
Survival motor neuron protein
- SMA:
-
Spinal muscular atrophy
- α-MNs:
-
α-Motor neurons
- snRNP:
-
Spliceosomal small nuclear ribonucleoproteins
- MOC:
-
Mander´s overlap coefficient
References
Agarkova I, Perriard J-C (2005) The M-band: an elastic web that crosslinks thick filaments in the center of the sarcomere. Trends Cell Biol 15:477–485
Ahmad S, Bhatia K, Kannan A, Gangwani L (2016) Molecular mechanisms of neurodegeneration in spinal muscular atrophy. J Exp Neurosci 10:39–49
Allen DL, Roy RR, Edgerton VR (1999) Myonuclear domains in muscle adaptation and disease. Muscle Nerve 22:1350–1360
Arnold A-S, Gueye M, Guettier-Sigrist S, Courdier-Fruh I, Coupin G, Poindron P, Gies J-P (2004) Reduced expression of nicotinic AChRs in myotubes from spinal muscular atrophy I patients. Lab Investig 84:1271–1278
Atkinson RA, Joseph C, Kelly G, Muskett FW, Frenkiel TA, Nietlispach D, Pastore A (2001) Ca 2+ -independent binding of an EF-hand domain to a novel motif in the α-actinin–titin complex. Nat Struct Biol 8:853–857
Bernabò P, Tebaldi T, Groen EJN, Lane FM, Perenthaler E, Mattedi F, Newbery HJ, Zhou H, Zuccotti P, Potrich V et al (2017) In vivo translatome profiling in spinal muscular atrophy reveals a role for SMN protein in ribosome biology. Cell Rep 21:953–965
Bevan AK, Hutchinson KR, Foust KD, Braun L, McGovern VL, Schmelzer L, Ward JG, Petruska JC, Lucchesi PA, Burghes AHM et al (2010) Early heart failure in the SMNDelta7 model of spinal muscular atrophy and correction by postnatal scAAV9-SMN delivery. Hum Mol Genet 19:3895–3905
Bolte S, Cordelières FP (2006) A guided tour into subcellular colocalization analysis in light microscopy. J Microsc 224:213–232
Bornemann A, Petersen MB, Schmalbruch H (1996) Fatal congenital myopathy with actin filament deposits. Acta Neuropathol 92:104–108
Bowerman M, Shafey D, Kothary R (2007) Smn depletion alters profilin II expression and leads to upregulation of the RhoA/ROCK pathway and defects in neuronal integrity. J Mol Neurosci 32:120–131
Bowerman M, Swoboda KJ, Michalski JP, Wang GS, Reeks C, Beauvais A, Murphy K, Woulfe J, Screaton RA, Scott FW et al (2012) Glucose metabolism and pancreatic defects in spinal muscular atrophy. Ann Neurol 72:256–268
Boyer JG, Ferrier A, Kothary R (2013) More than a bystander: the contributions of intrinsic skeletal muscle defects in motor neuron diseases. Front Physiol 4:356
Braun S, Warter JM, Poindron P, Croizat B, Lagrange MC (1995) Constitutive muscular abnormalities in culture in spinal muscular atrophy. Lancet 345:694–695
Bricceno KV, Martinez T, Leikina E, Duguez S, Partridge TA, Chernomordik LV, Fischbeck KH, Sumner CJ, Burnett BG (2014) Survival motor neuron protein deficiency impairs myotube formation by altering myogenic gene expression and focal adhesion dynamics. Hum Mol Genet
Brzustowicz LM, Lehner T, Castilla LH, Penchaszadeh GK, Wilhelmsen KC, Daniels R, Davies KE, Leppert M, Ziter F, Wood D et al (1990) Genetic mapping of chronic childhood-onset spinal muscular atrophy to chromosome 5q1 1.2?13.3. Nature 344:540–541
Burghes AHM, Beattie CE (2009) Spinal muscular atrophy: why do low levels of survival motor neuron protein make motor neurons sick? Nat Rev Neurosci 10:597–609
Burlet P, Huber C, Bertrandy S, Ludosky MA, Zwaenepoel I, Clermont O, Roume J, Delezoide AL, Cartaud J, Munnich A et al (1998) The distribution of SMN protein complex in human fetal tissues and its alteration in spinal muscular atrophy. Hum Mol Genet 7:1927–1933
Carvalho T, Almeida F, Calapez A, Lafarga M, Berciano MT, Carmo-Fonseca M (1999) The spinal muscular atrophy disease gene product, SMN: a link between snRNP biogenesis and the Cajal (coiled) body. J Cell Biol 147:715–728
Castillo-Iglesias MS, Berciano MT, Narcis JO, Val-Bernal JF, Rodriguez-Rey JC, Tapia O, Lafarga M (2019) Reorganization of the nuclear compartments involved in transcription and RNA processing in myonuclei of type I spinal muscular atrophy. Histochem Cell Biol 152:227–237
Centner, T., Fougerousse, F., Freiburg, A., Witt, C., Beckmann, J.S., Granzier, H., Trombitás, K., Gregorio, C.C., and Labeit, S. (2000). Molecular tools for the study of titin’s differential expression. In Advances in Experimental Medicine and Biology, pp. 35–52
Chaytow H, Huan H, Gillingwater TH, Faller KME (2018) The role of survival motor neuron protein (SMN) in protein homeostasis. Cell Mol Life Sci 75:3877–3894
Cifuentes-Diaz C, Frugier T, Tiziano FD, Lacène E, Roblot N, Joshi V, Moreau MH, Melki J (2001) Deletion of murine SMN exon 7 directed to skeletal muscle leads to severe muscular dystrophy. J Cell Biol 152:1107–1114
Coady TH, Lorson CL (2011) SMN in spinal muscular atrophy and snRNP biogenesis. Wiley Interdiscip Rev RNA 2:546–564
Coovert DD, Le TT, McAndrew PE, Strasswimmer J, Crawford TO, Mendell JR, Coulson SE, Androphy EJ, Prior TW, Burghes AHM (1997) The survival motor neuron protein in spinal muscular atrophy. Hum Mol Genet 6:1205–1214
Coque E, Raoul C, Bowerman M (2014) ROCK inhibition as a therapy for spinal muscular atrophy: understanding the repercussions on multiple cellular targets. Front Neurosci 8:271
Deguise MO, De Repentigny Y, McFall E, Auclair N, Sad S, Kothary R (2017) Immune dysregulation may contribute to disease pathogenesis in spinal muscular atrophy mice. Hum Mol Genet 26:801–819
Dubowitz V, and Sewry C (2007) Muscle biopsy: a practical approach. Saunders, Elsevier.
Feldkötter M, Schwarzer V, Wirth R, Wienker TF, Wirth B (2002) Quantitative analyses of SMN1 and SMN2 based on real-time lightCycler PCR: fast and highly reliable carrier testing and prediction of severity of spinal muscular atrophy. Am J Hum Genet 70:358–368
Förthmann B, Brinkmann H, Ratzka A, Stachowiak MK, Grothe C, Claus P (2013) Immobile survival of motoneuron (SMN) protein stored in Cajal bodies can be mobilized by protein interactions. Cell Mol Life Sci 70:2555–2568
Gautel M, Mues A, Young P (1999) Control of sarcomeric assembly: the flow of information on titin. Rev Physiol Biochem Pharmacol 138:97–137
Giesemann T, Rathke-Hartlieb S, Rothkegel M, Bartsch JW, Buchmeier S, Jockusch BM, Jockusch H (1999) A role for polyproline motifs in the spinal muscular atrophy protein SMN. Profilins bind to and colocalize with smn in nuclear gems. J Biol Chem 274:37908–37914
Goebel HH, Anderson JR, Hübner C, Oexle K, Warlo I (1997) Congenital myopathy with excess of thin myofilaments. Neuromuscul Disord 7:160–168
Gokhin DS, Lewis RA, McKeown CR, Nowak RB, Kim NE, Littlefield RS, Lieber RL, Fowler VM (2010) Tropomodulin isoforms regulate thin filament pointed-end capping and skeletal muscle physiology. J Cell Biol 189:95–109
Görgens SW, Eckardt K, Jensen J, Drevon CA, Eckel J (2015) Exercise and regulation of adipokine and myokine production. Prog Mol Biol Transl Sci 135:313–336
Greensmith L, Vrbová G (1997) Disturbances of neuromuscular interaction may contribute to muscle weakness in spinal muscular atrophy. Neuromuscul Disord 7:369–372
Groen EJN, Perenthaler E, Courtney NL, Jordan CY, Shorrock HK, van der Hoorn D, Huang Y-T, Murray LM, Viero G, Gillingwater TH (2018) Temporal and tissue-specific variability of SMN protein levels in mouse models of spinal muscular atrophy. Hum Mol Genet 27:2851–2862
Guettier-Sigrist S, Hugel B, Coupin G, Freyssinet J-M, Poindron P, Warter J-M (2002) Possible pathogenic role of muscle cell dysfunction in motor neuron death in spinal muscular atrophy. Muscle Nerve 25:700–708
Hall A (1998) Rho GTPases and the actin cytoskeleton. Science 279(80):509–514
Hall ZW, Ralston E (1989) Nuclear domains in muscle cells. Cell 59:771–772
Hamilton G, Gillingwater TH (2013) Spinal muscular atrophy: going beyond the motor neuron. Trends Mol Med 19:40–50
Hebert MD, Szymczyk PW, Shpargel KB, Matera AG (2001) Coilin forms the bridge between Cajal bodies and SMN, the spinal muscular atrophy protein. Genes Dev 15:2720–2729
Heier CR, Satta R, Lutz C, Didonato CJ (2010) Arrhythmia and cardiac defects are a feature of spinal muscular atrophy model mice. Hum Mol Genet 19:3906–3918
Hensel N, Claus P (2018) The actin cytoskeleton in SMA and ALS: how does it contribute to motoneuron degeneration? Neuroscientist 24:54–72
Hensel N, Stockbrügger I, Rademacher S, Broughton N, Brinkmann H, Grothe C, Claus P (2014) Bilateral crosstalk of rho- and extracellular-signal-regulated-kinase (ERK) pathways is confined to an unidirectional mode in spinal muscular atrophy (SMA). Cell Signal 26:540–548
Hindi SM, Tajrishi MM, Kumar A (2013) Signaling mechanisms in mammalian myoblast fusion. Sci. Signal 6:re2
Ince, P.G., and Wharton, S.B. (2007). Chapter 5 cytopathology of the motor neuron. In Handbook of Clinical Neurology, pp 89–119
Jokela M, Huovinen S, Raheem O, Lindfors M, Palmio J, Penttilä S, Udd B (2016) Distinct muscle biopsy findings in genetically defined adult-onset motor neuron disorders. PLoS One 11:e0151376
Kariya S, Park G-H, Maeno-Hikichi Y, Leykekhman O, Lutz C, Arkovitz MS, Landmesser LT, Monani UR (2008) Reduced SMN protein impairs maturation of the neuromuscular junctions in mouse models of spinal muscular atrophy. Hum Mol Genet 17:2552–2569
Kim EK, Choi E-J (2017) SMN1 functions as a novel inhibitor for TRAF6-mediated NF-κB signaling. Biochim Biophys Acta - Mol Cell Res 1864:760–770
Kontrogianni-Konstantopoulos A, Ackermann MA, Bowman AL, Yap SV, Bloch RJ (2009) Muscle giants: molecular scaffolds in sarcomerogenesis. Physiol Rev 89:1217–1267
Lafarga M, Casafont I, Bengoechea R, Tapia O, Berciano MT (2009) Cajal’s contribution to the knowledge of the neuronal cell nucleus. Chromosoma 118:437–443
Lange, S., Pinotsis, N., Agarkova, I., and Ehler, E. (2019). The M-band: the underestimated part of the sarcomere. Biochim. Biophys. Acta - Mol. Cell Res
Lee JH, Jun H-S (2019) Role of Myokines in regulating skeletal muscle mass and function. Front Physiol 10:42
Lefebvre S, Bürglen L, Reboullet S, Clermont O, Burlet P, Viollet L, Benichou B, Cruaud C, Millasseau P, Zeviani M (1995) Identification and characterization of a spinal muscular atrophy-determining gene. Cell 80:155–165
Li DK, Tisdale S, Lotti F, Pellizzoni L (2014) SMN control of RNP assembly: from post-transcriptional gene regulation to motor neuron disease. Semin Cell Dev Biol 32:22–29
Liu Q, Dreyfuss G (1996) A novel nuclear structure containing the survival of motor neurons protein. EMBO J 15:3555–3565
Lorson CL, Strasswimmer J, Yao JM, Baleja JD, Hahnen E, Wirth B, Le T, Burghes AH, Androphy EJ (1998) SMN oligomerization defect correlates with spinal muscular atrophy severity. Nat Genet 19:63–66
Luo L, Jan LY, Jan YN (1997) Rho family small GTP-binding proteins in growth cone signalling. Curr Opin Neurobiol 7:81–86
Martínez-Hernández R, Soler-Botija C, Also E, Alias L, Caselles L, Gich I, Bernal S, Tizzano EF (2009) The developmental pattern of myotubes in spinal muscular atrophy indicates prenatal delay of muscle maturation. J Neuropathol Exp Neurol 68:474–481
Matera AG, Wang Z (2014) A day in the life of the spliceosome. Nat Rev Mol Cell Biol 15:108–121
Melki J, Sheth P, Abdelhak S, Burlet P, Bachelot MF, Lathrop MG, Frezal J, Munnich A (1990) Mapping of acute (type I) spinal muscular atrophy to chromosome 5q12-q14. The French Spinal Muscular Atrophy Investigators. Lancet (London, England) 336:271–273
Monani UR, Lorson CL, Parsons DW, Prior TW, Androphy EJ, Burghes AH, McPherson JD (1999) A single nucleotide difference that alters splicing patterns distinguishes the SMA gene SMN1 from the copy gene SMN2. Hum Mol Genet 8:1177–1183
Mutsaers CA, Wishart TM, Lamont DJ, Riessland M, Schreml J, Comley LH, Murray LM, Parson SH, Lochmüller H, Wirth B et al (2011) Reversible molecular pathology of skeletal muscle in spinal muscular atrophy. Hum Mol Genet 20:4334–4344
Navascues J, Berciano MT, Tucker KE, Lafarga M, Matera AG (2004) Targeting SMN to Cajal bodies and nuclear gems during neuritogenesis. Chromosoma 112:398–409
Newham DJ, McPhail G, Mills KR, Edwards RH (1983) Ultrastructural changes after concentric and eccentric contractions of human muscle. J Neurol Sci 61:109–122
Nicholson-Dykstra S, Higgs HN, Harris ES (2005) Actin dynamics: growth from dendritic branches. Curr Biol 15:R346–R357
Nölle A, Zeug A, van Bergeijk J, Tönges L, Gerhard R, Brinkmann H, Al Rayes S, Hensel N, Schill Y, Apkhazava D et al (2011) The spinal muscular atrophy disease protein SMN is linked to the rho-kinase pathway via profilin. Hum Mol Genet 20:4865–4878
North, K.N., and Laing, N.G. (2008). Skeletal muscle alpha-actin diseases. In Advances in Experimental Medicine and Biology, pp 15–27
Ogawa C, Usui K, Aoki M, Ito F, Itoh M, Kai C, Kanamori-Katayama M, Hayashizaki Y, Suzuki H (2007) Gemin2 plays an important role in stabilizing the survival of motor neuron complex. J Biol Chem 282:11122–11134
Ogino S, Wilson RB (2004) Spinal muscular atrophy: molecular genetics and diagnostics. Expert Rev Mol Diagn 4:15–29
Ono, S. (2007). Mechanism of depolymerization and severing of actin filaments and its significance in cytoskeletal dynamics. In International Review of Cytology, pp. 1–82
Ono S (2010) Dynamic regulation of sarcomeric actin filaments in striated muscle. Cytoskeleton 67:677–692
Otter S, Grimmler M, Neuenkirchen N, Chari A, Sickmann A, Fischer U (2007) A comprehensive interaction map of the human survival of motor neuron (SMN) complex. J Biol Chem 282:5825–5833
Paavilainen VO, Bertling E, Falck S, Lappalainen P (2004) Regulation of cytoskeletal dynamics by actin-monomer-binding proteins. Trends Cell Biol 14:386–394
Paul PK, Gupta SK, Bhatnagar S, Panguluri SK, Darnay BG, Choi Y, Kumar A (2010) Targeted ablation of TRAF6 inhibits skeletal muscle wasting in mice. J Cell Biol 191:1395–1411
Pedersen BK, Febbraio MA (2008) Muscle as an endocrine organ: focus on muscle-derived Interleukin-6. Physiol Rev 88:1379–1406
Pellizzoni L, Yong J, Dreyfuss G (2002) Essential role for the SMN complex in the specificity of snRNP assembly. Science 298:1775–1779
Rajendra TK, Gonsalvez GB, Walker MP, Shpargel KB, Salz HK, Matera AG (2007) A Drosophila melanogaster model of spinal muscular atrophy reveals a function for SMN in striated muscle. J Cell Biol 176:831–841
Rochette C, Gilbert N, Simard L (2001) SMN gene duplication and the emergence of the SMN2 gene occurred in distinct hominids: SMN2 is unique to Homo sapiens. Hum Genet 108:255–266
Rossoll W, Jablonka S, Andreassi C, Kröning A-K, Karle K, Monani UR, Sendtner M (2003) Smn, the spinal muscular atrophy-determining gene product, modulates axon growth and localization of beta-actin mRNA in growth cones of motoneurons. J Cell Biol 163:801–812
Shababi M, Habibi J, Yang HT, Vale SM, Sewell WA, Lorson CL (2010) Cardiac defects contribute to the pathology of spinal muscular atrophy models. Hum Mol Genet 19:4059–4071
Shafey D, Côté PD, Kothary R (2005) Hypomorphic Smn knockdown C2C12 myoblasts reveal intrinsic defects in myoblast fusion and myotube morphology. Exp Cell Res 311:49–61
Sharma MC, Goebel HH (2005) Protein aggregate myopathies. Neurol India 53:273–279
Sharma A, Lambrechts A, Hao LT, Le TT, Sewry CA, Ampe C, Burghes AHM, Morris GE (2005) A role for complexes of survival of motor neurons (SMN) protein with gemins and profilin in neurite-like cytoplasmic extensions of cultured nerve cells. Exp Cell Res 309:185–197
Simone C, Ramirez A, Bucchia M, Rinchetti P, Rideout H, Papadimitriou D, Re DB, Corti S (2016) Is spinal muscular atrophy a disease of the motor neurons only: pathogenesis and therapeutic implications? Cell Mol Life Sci 73:1003–1020
Singh RN, Howell MD, Ottesen EW, Singh NN (2017) Diverse role of survival motor neuron protein. Biochim Biophys Acta - Gene Regul Mech 1860:299–315
Sjöström M, Squire JM (1977) Fine structure of the A-band in cryo-sections. The structure of the A-band of human skeletal muscle fibres from ultra-thin cryo-sections negatively stained. J Mol Biol 109:49–68
Somers E, Lees RD, Hoban K, Sleigh JN, Zhou H, Muntoni F, Talbot K, Gillingwater TH, Parson SH (2016) Vascular defects and spinal cord hypoxia in spinal muscular atrophy. Ann Neurol 79:217–230
Suarez C, Carroll RT, Burke TA, Christensen JR, Bestul AJ, Sees JA, James ML, Sirotkin V, Kovar DR (2015) Profilin regulates F-actin network homeostasis by favoring formin over Arp2/3 complex. Dev Cell 32:43–53
Sumner CJ (2007) Molecular mechanisms of spinal muscular atrophy. J Child Neurol 22:979–989
Szunyogova E, Zhou H, Maxwell GK, Powis RA, Francesco M, Gillingwater TH, Parson SH (2016) Survival motor neuron (SMN) protein is required for normal mouse liver development. Sci Rep 6:34635
Tapia O, Bengoechea R, Palanca A, Arteaga R, Val-Bernal JF, Tizzano EF, Berciano MT, Lafarga M (2012) Reorganization of Cajal bodies and nucleolar targeting of coilin in motor neurons of type I spinal muscular atrophy. Histochem Cell Biol 137:657–667
Tapia O, Narcís JO, Riancho J, Tarabal O, Piedrafita L, Calderó J, Berciano MT, Lafarga M (2017) Cellular bases of the RNA metabolism dysfunction in motor neurons of a murine model of spinal muscular atrophy: role of Cajal bodies and the nucleolus. Neurobiol Dis 108:83–99
Tisdale S, Pellizzoni L (2015) Disease mechanisms and therapeutic approaches in spinal muscular atrophy. J Neurosci 35:8691–8700
Vitte J, Fassier C, Tiziano FD, Dalard C, Soave S, Roblot N, Brahe C, Saugier-Veber P, Bonnefont JP, Melki J (2007) Refined characterization of the expression and stability of the SMN gene products. Am J Pathol 171:1269–1280
Walker MP, Rajendra TK, Saieva L, Fuentes JL, Pellizzoni L, Matera AG (2008) SMN complex localizes to the sarcomeric Z-disc and is a proteolytic target of calpain. Hum Mol Genet 17:3399–3410
Zhang R, So BR, Li P, Yong J, Glisovic T, Wan L, Dreyfuss G (2011) Structure of a key intermediate of the SMN complex reveals Gemin2’s crucial function in snRNP assembly. Cell 146:384–395
Acknowledgments
The authors wish to thank Raquel García-Ceballos for technical assistance.
Funding
This work was supported by the following grants: “Centro de Investigación Biomédica en Red sobre Enfermedades Neurodegenerativas” (CIBERNED; CB06/05/0037), Spain, and “Instituto de Investigación Valdecilla” (IDIVAL, Next-Val NVAL17/22), Spain.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare that they have no conflict of interest. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Berciano, M.T., Castillo-Iglesias, M.S., Val-Bernal, J.F. et al. Mislocalization of SMN from the I-band and M-band in human skeletal myofibers in spinal muscular atrophy associates with primary structural alterations of the sarcomere. Cell Tissue Res 381, 461–478 (2020). https://doi.org/10.1007/s00441-020-03236-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-020-03236-3