Abstract
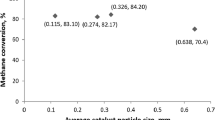
In the present work, three kinetic expressions for methane steam reforming with different methane and water partial pressures dependence were studied. Methane effectiveness factors were simulated for spherical uniform pellets (those customarily employed), for several temperatures and water/methane ratios (hereafter called R). It was found that all effectiveness factors decreased with temperature as expected for all the kinetics expressions studied. On the other hand, the evolution with R was strongly dependent on the kinetics analyzed, with an important change in effectiveness factors values, ranging from 30 to 400%. This implies that the most convenient active sites distribution might be dependent on R evolution throughout the reactor. Then, two MSR reactors were simulated, loaded with pelletized catalysts with uniform and eggshell in order to get uniform nomenclature active site distributions. It was found that the one loaded with eggshell in order to get uniform nomenclature pellets saves up to 30% catalysts mass with respect to the uniform catalyst distribution for the same CH4 conversion, reinforcing the importance of active sites distribution.






Similar content being viewed by others
Abbreviations
- DFT:
-
Density functional theory
- DRM:
-
Dry reforming of methane
- MSR:
-
Methane steam reforming
- C [mol m3]:
-
Molar concentration
- Cp [J mol−1 K−1]:
-
Molar heat capacity
- D [m2 s−1]:
-
Diffusivity
- DK [m2 s−1]:
-
Knudsen diffusivity
- F [mol s−1]:
-
Molar flow
- H [-]:
-
Heaviside step function
- ΔH [J mol−1]:
-
Heat of reaction
- k []:
-
Specific reaction constant
- K []:
-
Equilibrium constant
- L [m]:
-
Pellet size
- M [kg]:
-
Catalyst mass
- N [mol s−1 m−2]:
-
Molar flux
- P [atm]:
-
Pressure
- q [W s−1 m−2]:
-
Heat flux
- r [mol s−1 m−3]:
-
Reaction rate
- R [-]:
-
Water to methane feed ratio
- Rc [atm L mol−1 K−1]:
-
Universal gas constant
- T [K]:
-
Temperature
- z [m]:
-
Spatial coordinate
- α [-]:
-
Stoichiometric factor
- κ [W m−1 K−1]:
-
Thermal conductivity
- η [-]:
-
Effectiveness factor
- *:
-
Indicates dimensionless variable
- a:
-
Active phase left boundary
- b:
-
Active phase right boundary
- i:
-
Reaction
- j:
-
Species
- s:
-
At the surface conditions
References
Xu J, Froment G (1989) Methane steam reforming, methanation and water-gas shift: I. Intrinsic Kinet AIChE J 35:88–96
Bodrov N, Apelbaum L, Temkin M (1964) Kinetics of the reaction of methane wit steam on the surface of nickel. Kinetic Catal 5:696–705
De Deken J, Devos E, Froment G (1982) Steam reforming of natural gas: intrinsic kinetics, diffusional influences, and reactor desing. Chem Reaction Eng ACS Symp Ser 196:82–88
Elnashaie S, Adris A, Al-Ubaid A, Soliman M (1990) On the non-monotonic behaviour of methane—steam reforming kinetics. Ch Eng Sci 45:491–501
Hou K, Hughes R (2001) The kinetics of methane steam reforming over a Ni/α–Al2O3 catalyst. Ch Eng J 82:311–328
Mei D, Glezakou C, Lebarbier V, Kovarik L, Wan H, Albrecht K, Gerber M, Rousseau R, Dagle R (2014) Highly active and stable MgAl2O4-supported Rh and Ir catalysts for methane steam reforming: a combined experimental and theoretical study. J Catal 316:11–23
Wang Y, Chin Y, Rozmiarek R, Johnson B, Gao Y, Watson J, Tonkovich A, Van der Wiel D (2004) Highly active and stable Rh/MgOAl2O3 catalysts for methane steam reforming. Catal Today 98:575–581
Jones G, Jakobsen J, Shim S, Kleis J, Andersson M, Rossmeisl J, Abild-Pedersen F, Bligaard T, Helveg S, Hinnemann B, Rostrup-Nielsen J, Chorkendorff I, Sehested J, Nørskov J (2008) First principles calculations and experimental insight into methane steam reforming over transition metal catalysts. J Catal 259:147–160
Wei J, Iglesia E (2004) Structural requirements and reaction pathways in methane activation and chemical conversion catalyzed by rhodium. J Catal 225:116–127
Wei J, Iglesia E (2004) Isotopic and kinetic assessment of the mechanism of reactions of CH4 with CO2 or H2O to form synthesis gas and carbon on nickel catalysts. J Catal 224:370–383
Angeli S, Monteleone G, Giaconia A, Lemonidou A (2014) State-of-the-art catalysts for CH4 steam reforming at low temperature. Int J Hydrog Energy 39:1979–1997
Dong W, Roh H, Jun K, Park S, Oh Y (2002) Methane reforming over Ni/Ce-ZrO2 catalysts: effect of nickel content. Appl Catal A 226:63–72
Angeli S, Turchetti L, Monteleone G, Lemonidou A (2016) Catalyst development for steam reforming of methane and model biogas at low temperature. Appl Catal B Environ 181:34–46
Laosiripojana N, Assabumrungrat S (2005) Methane steam reforming over Ni/Ce–ZrO2 catalyst: influences of Ce–ZrO2 support on reactivity, resistance toward carbon formation, and intrinsic reaction kinetics. Appl Catal A 290:200–211
Iglesias I, Baronetti G, Mariño F (2017) Ni/Ce0.95M0.05O2−d (M=Zr, Pr, La) for methane steam reforming at mild conditions. Int J Hydrog Energy 42:29735–29744
Craciun R, Shereck B, Gorte R (1998) Kinetic studies of methane steam reforming on ceria-supported Pd. Catal Lett 51:149–153
Carrasco J, López-Durán D, Liu Z, Duchon T, Evans J, Senanayake S, Crumlin E, Matolín V, Rodríguez J, Ganduglia-Pirovano M (2015) In situ and theoretical studies for the dissociation of water on an active Ni/CeO2 catalyst: importance of strong metal-support interactions for the cleavage of O–H bonds. Angew Chem Int Ed 54:3917–3923
Liu Z, Grinter D, Lustemberg P, Nguyen-Phan T, Zhou Y, Luo S, Waluyo I, Crumlin EJ, Stacchiola D, Zhou J, Carrasco J, Busnengo H, Ganduglia-Pirovano H, Senanayake S, Rodriguez J (2016) Dry reforming of Methane on a highly-active Ni–CeO2 catalyst: effects of metal-support interactions on C–H bond breaking. Angew Chem Int Ed 55:7455–7459
Liu Z, Lustemberg P, Gutiérrez R, Carey J, Palomino R, Vorokhta M, Grinter D, Ramírez P, Matolín V, Nolan M, Ganduglia-Pirovano M, Senanayake S, Rodriguez J (2017) In situ investigation of methane dry reforming on metal/ceria (111) surfaces: metal–support interactions and C−H bond activation at low temperature. Angew Chem Int Ed 56:13041–13046
Huang T, Lin H, Yu T (2005) A Comparison of oxygen-vacancy effect on activity behaviors of carbon dioxide and steam reforming of methane over supported nickel catalysts. Catal Lett 105:239–247
Iglesias I, Baronetti G, Mariño F (2017) Nickel-based doped ceria-supported catalysts for steam reforming of methane at mild conditions. Energy Sour Part A 39:129–133
Halabi M, de Croon M, van der Schaaf J, Cobden P, Schouten J (2010) Intrinsic kinetics of low temperature catalytic methane–steam reforming and water–gas shift over Rh/CeαZr1−αO2 catalyst. Appl Catal A 389:80–91
Murkin C, Brightling J (2016) Eighty years of steam reforming. Johnson Matthey Technol Rev 60:263–269
Cho E, Koo K, Lee H, Park Y, Yoon W, Ko C (2017) Preparation of egg-shell-type Ni/Ru bimetal alumina pellet catalysts: steam methane reforming for hydrogen production. Int J Hydrog Energy 42:18350–18357
Kim Y, Cho E, Ko C (2019) Preparation of Ni-based egg-shell-type catalyst on cylinder-shaped alumina pellets and its application for hydrogen production via steam methane reforming. Int J Hydrog Energ 44:5314–5323
National Research Council, A Workshop Summary (2012) The role of the chemical sciences in finding alternatives to critical resources. The National Academies Press, Washington, DC
Augustine R (1955) Heterogeneous catalysis for the synthetic chemist, 1st edn. Marcel Dekker, Inc., New York
Elnashaie S, Elshishini S (1993) Modelling, simulation and optimization of industrial fixed bed catalytic reactors. OPA, Amsterdam, p 425
Khafizov M, Park I, Chernatynskiy A, He L, Lin J, Moore J, Swank D, Lillo T, Phillpot S, El-Azab A, Hurley D (2014) Thermal conductivity in nanocrystalline ceria thin films. J Am Ceram Soc 97:562–569
Munro R (1997) Evaluated material properties for a sintered alpha-alumina. J Am Ceram Soc 80:1919–1928
Reid R, Prausnitz J, Pohling B (1987) The properties of gases and liquids, 4th edn. McGraw-Hill, New York
Wilke C (1950) Diffusional properties of multicomponent gases. Chem Eng Prog 46:95–99
Cho E, Yu Y, Kim Y, Phan T, Park D, Ko C (2020) Egg-shell-type Ni supported on MgAl2O4 pellets as catalyst for steam methane reforming: Enhanced coke-resistance and pellet stability. Catal Today 352:157–165
Long Y, Li K, Gu Z, Wei Y, Lu C, Lin S, Yang K, Cheng X, Tian D, He F, Wang H (2020) Ce–Fe–Zr–O/MgO coated monolithic oxygen carriers for chemical looping reforming of methane to co-produce syngas and H2. Chem Eng J 388:124190–124203
Balzarotti R, Ambrosetti M, Beretta A, Groppi G, Tronconi E (2020) Investigation of packed conductive foams as a novel reactor configuration for methane steam reforming. Chem Eng J 391:123494–123506
Funding
Authors thank to Universidad of Buenos Aires and Consejo Nacional de Investigaciones Científicas y Técnicas CONICET for their financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
Authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Iglesias, I., Poggio-Fraccari, E. & Giunta, P. Study of effectiveness factors with non-uniform catalyst distributions for methane steam reforming. Reac Kinet Mech Cat 130, 713–726 (2020). https://doi.org/10.1007/s11144-020-01822-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11144-020-01822-8