Abstract
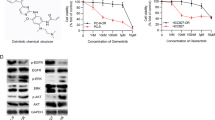
Lung adenocarcinoma (LUAD) is the top prevalent histological kind of lung cancer worldwide. Recent evidences have demonstrated that Sauchinone plays an anticancer role in tumor cell invasion and migration. Therefore, we performed this investigation to explain the potential role of Sauchinone in LUAD as well as the potential mechanism involved. Cell counting kit 8 (CCK-8) and transwell experiments were implemented to measure the proliferative, invasive and migratory abilities of LUAD cells. qRT-PCR and Western blot were performed to detect the transfection efficiency of si-EIF4EBP1s. Additionally, Western blot was also implemented to evaluate the effect of Sauchinone on EIF4EBP1 expression level as well as cell cycle-related proteins. Our findings showed that Sauchinone remarkably suppressed the proliferative ability of LUAD cells in a dose-dependent and time-dependent manner. EIF4EBP1 was a candidate target gene of Sauchinone. EIF4EBP1 expression was increased in LUAD tissues, and its high expression induced a poorer prognosis of LUAD patients. EIF4EBP1 expression was positively associated with cell cycle in LUAD. Sauchinone treatment attenuated EIF4EBP1 expression and cell cycle-related protein levels. Knockdown of EIF4EBP1 repressed the proliferation, invasion and migration of LUAD cells; furthermore, Sauchinone stimulation enforced its inhibitory effect. Meanwhile, the treatment of Sauchinone intensified the arrest of cell cycle induced by EIF4EBP1 knockdown. To sum up, our discovery indicated that Sauchinone exerts an anticancer role through down-regulating EIF4EBP1 and mediating cell cycle in LUAD.






Similar content being viewed by others
References
Senosain MF, Massion PP (2020) Intratumor heterogeneity in early lung adenocarcinoma. Front Oncol 10:349. https://doi.org/10.3389/fonc.2020.00349
Ruiz-Cordero R, Devine WP (2020) Targeted therapy and checkpoint immunotherapy in lung cancer. Surg Pathol Clin 13(1):17–33. https://doi.org/10.1016/j.path.2019.11.002
Zhang H, Guo L, Chen J (2020) Rationale for lung adenocarcinoma prevention and drug development based on molecular biology during carcinogenesis. OncoTargets Ther 13:3085–3091. https://doi.org/10.2147/ott.s248436
Zappa C, Mousa SA (2016) Non-small cell lung cancer: current treatment and future advances. Transl Lung Cancer Res 5(3):288–300. https://doi.org/10.21037/tlcr.2016.06.07
Lu Y, Ma J, Li Y, Huang J, Zhang S, Yin Z et al (2017) CDP138 silencing inhibits TGF-beta/Smad signaling to impair radioresistance and metastasis via GDF15 in lung cancer. Cell Death Dis 8(9):e3036. https://doi.org/10.1038/cddis.2017.434
Schiller JH, Harrington D, Belani CP, Langer C, Sandler A, Krook J et al (2002) Comparison of four chemotherapy regimens for advanced non-small-cell lung cancer. N Engl J Med 346(2):92–98. https://doi.org/10.1056/NEJMoa011954
Hu Y, Wang J (2019) Interactions between clopidogrel and traditional Chinese medicine. J Thromb Thrombolysis 48(3):491–499. https://doi.org/10.1007/s11239-019-01945-3
Ko EY, Moon A (2015) Natural products for chemoprevention of breast cancer. J Cancer Prev 20(4):223–231. https://doi.org/10.15430/jcp.2015.20.4.223
Xiao J, Wang J, Chen Y, Zhou Z, Gao C, Guo Z (2020) Sauchinone ameliorates intestinal inflammation and promotes Th17 cell production of IL-10 via Blimp-1. Biochem Biophys Res Commun 522(2):435–441. https://doi.org/10.1016/j.bbrc.2019.11.122
Bae HB, Li M, Son JK, Seo CS, Chung SH, Kim SJ et al (2010) Sauchinone, a lignan from Saururus chinensis, reduces tumor necrosis factor-alpha production through the inhibition of c-raf/MEK1/2/ERK 1/2 pathway activation. Int Immunopharmacol 10(9):1022–1028. https://doi.org/10.1016/j.intimp.2010.06.001
Gao Y, Zhao H, Li Y (2018) Sauchinone prevents IL-1β-induced inflammatory response in human chondrocytes. J Biochem Mol Toxicol 32(3):e22033. https://doi.org/10.1002/jbt.22033
Kim HY, Choi TW, Kim HJ, Kim SM, Park KR, Jang HJ et al (2011) A methylene chloride fraction of Saururus chinensis induces apoptosis through the activation of caspase-3 in prostate and breast cancer cells. Phytomedicine Int J Phytother Phytopharmacol 18(7):567–574. https://doi.org/10.1016/j.phymed.2010.10.013
He Z, Dong W, Li Q, Qin C, Li Y (2018) Sauchinone prevents TGF-β-induced EMT and metastasis in gastric cancer cells. Biomed Pharmacother 101:355–361. https://doi.org/10.1016/j.biopha.2018.02.121
Soma H, Yabe I, Takei A, Fujiki N, Yanagihara T, Sasaki H (2008) Associations between multiple system atrophy and polymorphisms of SLC1A4, SQSTM1, and EIF4EBP1 genes. Mov Disord 23(8):1161–1167. https://doi.org/10.1002/mds.22046
Kim YY, Von Weymarn L, Larsson O, Fan D, Underwood JM, Peterson MS et al (2009) Eukaryotic initiation factor 4E binding protein family of proteins: sentinels at a translational control checkpoint in lung tumor defense. Can Res 69(21):8455–8462. https://doi.org/10.1158/0008-5472.can-09-1923
Musa J, Orth MF, Dallmayer M, Baldauf M, Pardo C, Rotblat B et al (2016) Eukaryotic initiation factor 4E-binding protein 1 (4E-BP1): a master regulator of mRNA translation involved in tumorigenesis. Oncogene 35(36):4675–4688. https://doi.org/10.1038/onc.2015.515
Karlsson E, Pérez-Tenorio G, Amin R, Bostner J, Skoog L, Fornander T et al (2013) The mTOR effectors 4EBP1 and S6K2 are frequently coexpressed, and associated with a poor prognosis and endocrine resistance in breast cancer: a retrospective study including patients from the randomised Stockholm tamoxifen trials. Breast Cancer Res BCR 15(5):R96. https://doi.org/10.1186/bcr3557
Karlsson E, Waltersson MA, Bostner J, Pérez-Tenorio G, Olsson B, Hallbeck AL et al (2011) High-resolution genomic analysis of the 11q13 amplicon in breast cancers identifies synergy with 8p12 amplification, involving the mTOR targets S6K2 and 4EBP1. Genes Chromosom Cancer 50(10):775–787. https://doi.org/10.1002/gcc.20900
Zhu J, Wang M, Hu D (2020) Development of an autophagy-related gene prognostic signature in lung adenocarcinoma and lung squamous cell carcinoma. PeerJ 8:e8288. https://doi.org/10.7717/peerj.8288
Kim YW, Lee SM, Shin SM, Hwang SJ, Brooks JS, Kang HE et al (2009) Efficacy of sauchinone as a novel AMPK-activating lignan for preventing iron-induced oxidative stress and liver injury. Free Radic Biol Med 47(7):1082–1092. https://doi.org/10.1016/j.freeradbiomed.2009.07.018
Kim YW, Jang EJ, Kim CH, Lee JH (2017) Sauchinone exerts anticancer effects by targeting AMPK signaling in hepatocellular carcinoma cells. Chem Biol Interact 261:108–117. https://doi.org/10.1016/j.cbi.2016.11.016
Gong QZ, Xiao D, Feng F, Wen XD, Qu W (2018) ent-Sauchinone as potential anticancer agent inhibiting migration and invasion of human liver cancer cells via suppressing the STAT3 signaling pathway. Chem Biodivers 15(4):e1800024. https://doi.org/10.1002/cbdv.201800024
Lee HW, Lee EH, Lee JH, Kim JE, Kim SH, Kim TG et al (2015) Prognostic significance of phosphorylated 4E-binding protein 1 in non-small cell lung cancer. Int J Clin Exper Pathol 8(4):3955–3962
Wang Z, Feng X, Molinolo AA (2019) 4E-BP1 Is a tumor suppressor protein reactivated by mTOR inhibition in head and neck cancer. Cancer Res 79(7):1438–1450. https://doi.org/10.1158/0008-5472.can-18-1220
Vo DT, Karanam NK, Ding L, Saha D, Yordy JS, Giri U et al (2019) miR-125a-5p Functions as tumor suppressor microrna and is a marker of locoregional recurrence and poor prognosis in head and neck cancer. Neoplasia (New York, NY) 21(9):849–862. https://doi.org/10.1016/j.neo.2019.06.004
Sava GP, Fan H, Coombes RC, Buluwela L, Ali S (2020) CDK7 inhibitors as anticancer drugs. Cancer Metastas Rev. https://doi.org/10.1007/s10555-020-09885-8
Hu H, Xu H, Lu F, Zhang J, Xu L, Xu S et al (2020) Exosome-derived miR-486-5p regulates cell cycle, proliferation and metastasis in lung adenocarcinoma via targeting NEK2. Front Bioeng Biotechnol 8:259. https://doi.org/10.3389/fbioe.2020.00259
Zheng C, Li X, Ren Y, Yin Z, Zhou B (2019) Long noncoding RNA RAET1K enhances CCNE1 expression and cell cycle arrest of lung adenocarcinoma cell by sponging miRNA-135a-5p. Front Genet 10:1348. https://doi.org/10.3389/fgene.2019.01348
Li JP, Yang YX, Liu QL, Pan ST, He ZX, Zhang X et al (2015) The investigational Aurora kinase A inhibitor alisertib (MLN8237) induces cell cycle G2/M arrest, apoptosis, and autophagy via p38 MAPK and Akt/mTOR signaling pathways in human breast cancer cells. Drug Des Dev Ther 9:1627–1652. https://doi.org/10.2147/dddt.s75378
Gentric G, Maillet V, Paradis V, Couton D, L'Hermitte A, Panasyuk G et al (2015) Oxidative stress promotes pathologic polyploidization in nonalcoholic fatty liver disease. J Clin Investig 125(3):981–992. https://doi.org/10.1172/jci73957
Wang J, Wu A, Xu Y, Liu J, Qian X (2009) M(2)-A induces apoptosis and G(2)-M arrest via inhibiting PI3K/Akt pathway in HL60 cells. Cancer Lett 283(2):193–202. https://doi.org/10.1016/j.canlet.2009.03.039
Canaud G, Bonventre JV (2015) Cell cycle arrest and the evolution of chronic kidney disease from acute kidney injury. Nephrol Dial Trans 30(4):575–583. https://doi.org/10.1093/ndt/gfu230
Nakajima K, Crisma AR, Silva GB, Rogero MM, Fock RA, Borelli P (2014) Malnutrition suppresses cell cycle progression of hematopoietic progenitor cells in mice via cyclin D1 down-regulation. Nutrition (Burbank, Los Angeles County, Calif) 30(1):82–89. https://doi.org/10.1016/j.nut.2013.05.029
Funding
This study was supported by Science and Technology Program of Sichuan Province in 2018 (No.:2018JY0415).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest regarding the publication of this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, SQ., Feng, J., Yang, M. et al. Sauchinone: a prospective therapeutic agent-mediated EIF4EBP1 down-regulation suppresses proliferation, invasion and migration of lung adenocarcinoma cells. J Nat Med 74, 777–787 (2020). https://doi.org/10.1007/s11418-020-01435-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11418-020-01435-4