Abstract
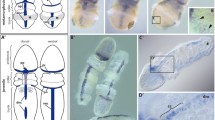
Crinoids are considered as the most basal extant echinoderms. They retain aboral nervous system with a nerve center, which has been degraded in the eleutherozoan echinoderms. To investigate the evolution of patterning of the nervous systems in crinoids, we examined temporal and spatial expression patterns of three neural patterning-related homeobox genes, six3, pax6, and otx, throughout the development of a feather star Anneissia japonica. These genes were involved in the patterning of endomesodermal tissues instead of the ectodermal neural tissues in the early planktonic stages. In the stages after larval attachment, the expression of these genes was mainly observed in the podia and the oral nervous systems instead of the aboral nerve center. Our results indicate the involvement of these three genes in the formation of oral nervous system in the common ancestor of the echinoderms and suggest that the aboral nerve center is not evolutionally related to the brain of other bilaterians.





Similar content being viewed by others
References
Ang S-L, Conlon RA, Jin O, Rossant J (1994) Positive and negative signals from mesoderm regulate the expression of mouse Otx2 in ectoderm explant. Development 120:2979–2989
Arenas-Mena C, Wong KS-Y (2007) HeOtx expression in an indirectly developing polychaete correlates with gastrulation by invagination. Dev Genes Evol 217:373–384. https://doi.org/10.1007/s00427-007-0150-7
Arendt D (2003) Evolution of eyes and photoreceptor cell types. Int J Dev Biol 47:563–571
Arendt D, Technau U, Wittbrodt J (2001) Evolution of the bilaterian larval foregut. Nature 409:81–85. https://doi.org/10.1038/35051075
Bottjer DJ, Davidson EH, Peterson KJ, Cameron RA (2006) Paleogenomics of echinoderms. Science 314(80):956–960. https://doi.org/10.1126/science.1132310
Brusca RC, Moore W, Shuster SM (2016) Invertebrates, 3rd edn. Sinauer, Sunderland
Callaerts P, Halder G, Gehring WJ (1997) Pax-6 in development and evolution. Annu Rev Neurosci 20:483–532
Clark AH, Clark AM (1967) A monograph of the existing crinoids 1(5). Bull US Nat Mus 82:1–860
Cohen BL, Améziane N, Eleaume M, de Forges BR (2004) Crinoid phylogeny: a preliminary analysis (Echinodermata: Crinoidea). Mar Biol 144:605–617. https://doi.org/10.1007/s00227-003-1212-7
Czerny T, Busslinger M (1995) DNA-binding and transactivation properties of Pax-6: three amino acids in the paired domain are responsible for the different sequence recognition of Pax-6 and BSAP (Pax-5). Mol Cell Biol 15:2858–2571
Dan JC, Dan K (1941) Early development of Comanthus japonicus. Japanese J Zool 9:565–574
Dan K, Ando Y, Sekiguchi K, Watanabe H (eds) (1988) Development of Invertebrates, Vol 2. Baifukan, Tokyo. [In Japanese]
Gan L, Mao C-A, Wikramanayake AH, Angerer LM, Angerer RC, Klein WH (1995) An orthodenticle-related protein from Strongylocentrotus purpuratus. Dev Biol 167:517–528
Garstang W (1894) Preliminary note on a new theory of the phylogeny of the Chordata. Zool Anzeiger 27:122–125
Gehring WJ, Ikeo K (1999) Pax 6: mastering eye morphogenesis and eye evolution. Trends Genet 15:371–377
Glardon S, Callaerts P, Halder G, Gehring WJ (1997) Conservation of Pax-6 in a lower chordate, the ascidian Phallusia mammillata. Development 124:817–825
Glardon S, Holland LZ, Gehring WJ, Holland ND (1998) Isolation and developmental expression of the amphioxus Pax-6 gene (AmphiPax-6): insights into eye and photoreceptor evolution. Development 125:2701–2710
Gonzalez P, Uhlinger KR, Lowe CJ (2017) The adult body plan of indirect developing hemichordates develops by adding a Hox-patterned trunk to an anterior larval territory. Curr Biol 27:87–95. https://doi.org/10.1016/j.cub.2016.10.047
Hara Y, Yamaguchi M, Akasaka K, Nakano H, Nonaka M, Amemiya S (2006) Expression patterns of Hox genes in larvae of the sea lily Metacrinus rotundus. Dev Genes Evol 216:797–809. https://doi.org/10.1007/s00427-006-0108-1
Harada Y, Okai N, Taguchi S, Tagawa K, Humphreys T, Satoh N (2000) Developmental expression of the hemichordate otx ortholog. Mech Dev 91:337–339
Hejnol A, Martindale MQ (2008) Acoel development supports a simple planula-like urbilaterian. Phil Trans R Soc B 363:1493–1501. https://doi.org/10.1098/rstb.2007.2239
Hinman VF, Nguyen AT, Davidson EH (2003) Expression and function of a starfish Otx ortholog, AmOtx: a conserved role for Otx proteins in endoderm development that predates divergence of the eleutherozoa. Mech Dev 120:1165–1176. https://doi.org/10.1016/j.mod.2003.08.002
Hyman LH (1955) The invertebrates, vol IV, Echinodermata. McGraw-Hill, New York
Irvine SQ, Fonseca VC, Zompa MA, Antony R (2008) Cis-regulatory organization of the Pax6 gene in the ascidian Ciona intestinalis. Dev Biol 317:649–659. https://doi.org/10.1016/j.ydbio.2008.01.036
Janies DA, Voight JR, Daly M (2011) Echinoderm phylogeny including Xyloplax, a progenetic asteroid. Syst Biol 60:420–438. https://doi.org/10.1093/sysbio/syr044
Kozmik Z (2005) Pax genes in eye development and evolution. Curr Opin Genet Dev 15:430–438. https://doi.org/10.1016/j.gde.2005.05.001
Kozmik Z, Holland ND, Kreslova J, Oliveri D, Schubert M, Jonasova K, Holland LZ, Pestarino M, Benes V, Candiani S (2007) Pax-Six-Eya-Dach network during amphioxus development: conservation in vitro but context specificity in vivo. Dev Biol 306:143–159. https://doi.org/10.1016/j.ydbio.2007.03.009
Kubota H (1969) Development of Comanthus japonica I. From spawning to attachment. Jpn J Dev Biol 23:92–93 (in Japanese)
Kubota H (1970) Development of Comanthus japonica II. After setting. Jpn J Dev Biol 24:40–41 (in Japanese)
Lowe CJ, Wray GA (1997) Radical alterations in the roles of homeobox genes during echinoderm evolution. Nature 389:718–721. https://doi.org/10.1038/39580
Lowe CJ, Wu M, Salic A, Evans L, Lander ES, Stange-Thomann N, Gruber CE, Gerhart J, Kirschner M (2003) Anteroposterior patterning in hemichordates and the origins of the chordate nervous system. Cell 113:853–865
Marshall AM (1884) On the nervous system of Antedon rosaceus. Q J Microsc Sci 24:507–548
Martínez-Morales JR, Rodrigo I, Bovolenta P (2004) Eye development: a view from the retina pigmented epithelium. BioEssays 26:766–777. https://doi.org/10.1002/bies.20064
Mazza ME, Pang K, Martindale MQ, Finnerty JR (2007) Genomic organization, gene structure, and developmental expression of three clustered otx genes in the sea anemone Nematostella vectensis. J Exp Zool Part B Mol Dev Evol 308B:494–506. https://doi.org/10.1002/jez.b.21158
Mitsunaga-Nakatsubo K, Akasaka K, Sakamoto N, Takata K, Matsumura Y, Kitajima T, Kusunoki S, Shimada H (1998) Differential expression of sea urchin Otx isoform (HpOtxE and HpOtxL) mRNAs during early development. Int J Dev Biol 42:645–651
Morris VB, Zhao J-T, Shearman DCA, Byrne M, Frommer M (2004) Expression of an Otx gene in the adult rudiment and the developing central nervous system in the vestibula larva of the sea urchin Holopneustes purpurescens. Int J Dev Biol 48:17–22
Nakano H, Hibino T, Oji T, Hara Y, Amemiya S (2003) Larval stages of a living sea lily (stalked crinoid echinoderm). Nature 421:158–160. https://doi.org/10.1038/nature01236.1
Nakano H, Nakajima Y, Amemiya S (2009) Nervous system development of two crinoid species, the sea lily Metacrinus rotundus and the feather star Oxycomanthus japonicus. Dev Genes Evol 219:565–576. https://doi.org/10.1007/s00427-010-0317-5
Nielsen MG, Popodi EM, Minsuk S, Raff RA (2003) Evolutionary convergence in Otx expression in the pentameral adult rudiment in direct-developing sea urchins. Dev Genes Evol 213:73–82. https://doi.org/10.1007/s00427-003-0299-7
Omori A, Akasaka K, Kurokawa D, Amemiya S (2011) Gene expression analysis of Six3, Pax6, and Otx in the early development of the stalked crinoid Metacrinus rotundus. Gene Expr Patterns 11:48–56. https://doi.org/10.1016/j.gep.2010.09.002
Pani AM, Mullarkey EE, Aronowicz J, Assimacopoulos S, Grove EA, Lowe CJ (2012) Ancient deuterostome origins of vertebrate brain signalling centres. Nature 483:289–294. https://doi.org/10.1038/nature10838
Paul CRC, Smith AB (1984) The early radiation and phylogeny of echinoderms. Biol Rev 59:443–481
Reichert H (2009) Evolutionary conservation of mechanisms for neural regionalization, proliferation and interconnection in brain development. Biol Lett 5:112–116. https://doi.org/10.1098/rsbl.2008.0337
Rouse GW, Jermiin LS, Wilson NG, Eeckhaut I, Lanterbecq D, Oji T, Young CM, Browning T, Cisternas P, Helgen LE, Stuckey M, Messing CG (2013) Fixed, free, and fixed: the fickle phylogeny of extant Crinoidea (Echinodermata) and their Permian-Triassic origin. Mol Phylogenet Evol 66:161–181. https://doi.org/10.1016/j.ympev.2012.09.018
Scouras A, Smith MJ (2006) The complete mitochondrial genomes of the sea lily Gymnocrinus richeri and the feather star Phanogenia gracilis: signature nucleotide bias and unique nad4L gene rearrangement within crinoids. Mol Phylogenet Evol 39:323–334. https://doi.org/10.1016/j.ympev.2005.11.004
Shibata TF, Sato A, Oji T, Akasaka K (2008) Development and growth of the feather star Oxycomanthus japonicus to sexual maturity. Zool Sci 25:1075–1083. https://doi.org/10.2108/zsj.25.1075
Shoguchi E, Harada Y, Numakunai T, Satoh N (2000) Expression of the otx gene in the ciliary bands during sea cucumber embryogenesis. genesis 27:58–63
Stierwald M, Yanze N, Bamert RP, Kammermeier L, Schmid V (2004) The Sine oculis/Six class family of homeobox genes in jellyfish with and without eyes: development and eye regeneration. Dev Biol 274:70–81. https://doi.org/10.1016/j.ydbio.2004.06.018
Tomsa JM, Langeland JA (1999) Otx expression during lamprey embryogenesis provides insights into the evolution of the vertebrate head and jaw. Dev Biol 207:26–37. https://doi.org/10.1006/dbio.1998.9163
Ullrich-Luter EM, Dupont S, Arboleda E, Hausen H, Arnone MI (2011) Unique system of photoreceptors in sea urchin tube feet. Proc Natl Acad Sci 108:8367–8372. https://doi.org/10.1073/pnas.1018495108
Wada S, Katsuyama Y, Sato Y, Itoh C, Saiga H (1996) Hroth, an orthodenticle-related homeobox gene of the ascidian, Halocynthia roretzi: its expression and putative roles in the axis formation during embryogenesis. Mech Dev 60:59–71
Williams NA, Holland PWH (1996) Old head on young shoulders. Nature 383:490
Zuber ME, Gestri G, Viczian AS, Barsacchi G, Harris WA (2003) Specification of the vertebrate eye by a network of eye field transcription factors. Development 130:5155–5167. https://doi.org/10.1242/dev.00723
Acknowledgments
We are grateful to Ms. Toko Tsurugaya (Urawa University) for her support in culturing and handling A. japonica. We also appreciate Mr. Minoru Sekimoto, Mr. Mamoru Sekifuji, Mr. Hisanori Kohtsuka, and Ms. Natsuko Sugii (Misaki Marine Biological Station) for their help in collecting and culturing of A. japonica, and we are obliged to Dr. Yoko Nakajima (Keio University), Dr. Daisuke Kurokawa, and Dr. Mariko Kondo (Misaki Marine Biological Station) for their valuable comments.
Funding
This work was supported in part by the Global COE Program (Integrative Life Science Based on the Study of Biosignaling Mechanisms), MEXT, Japan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants performed by any of the authors. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Additional information
Communicated by Mark Q. Martindale
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Omori, A., Shibata, T.F. & Akasaka, K. Gene expression analysis of three homeobox genes throughout early and late development of a feather star Anneissia japonica. Dev Genes Evol 230, 305–314 (2020). https://doi.org/10.1007/s00427-020-00665-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00427-020-00665-6