Abstract
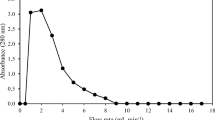
Lectins are proteins that bind to the carbohydrate moieties on surface of bacteria, erythrocytes and other cells of invertebrates causing agglutination and mediate in recognition of foreign substances. In the present study, we isolated and characterized a lectin molecule present in the hemolymph of Macrobrachium rosenbergii, an important cultured freshwater prawn. Lectin in serum samples of adult prawns was assessed through hemagglutination (HA) test using rabbit RBC that showed a titre ranging from 16 to 64. This serum hemagglutinin was confirmed as a C-type lectin based on its dependency on calcium ions towards binding to rabbit RBCs. The hemagglutinin was also found to be stable at the pH range of 5.0–10.0 and temperature range of 10–40 °C. Of various sugars and glycoproteins tested in hemagglutination inhibition assay, the serum lectin was found specific only to N-acetylneuraminic acid and fetuin with respective minimum inhibitory concentrations at 50 mM and 0.31 mg/ml. Further, the lectin was purified by affinity chromatography on rabbit erythrocyte stroma, which showed hemagglutination with rabbit RBC. In electrophoretic analyses, the purified lectin showed one band with molecular weight of ~ 427 kDa in native gradient PAGE, and its two constituent polypeptide chains of ~ 81 and ~ 73 kDa in SDS-PAGE. These polypeptides were analysed in MALDI-TOF/TOF mass spectrometry and identified as hemocyanins. It was hence, concluded that hemocyanin in M. rosenbergii possesses lectin-like activity.




Similar content being viewed by others
References
Nair CM, Salin KR (2012) Current status and prospects of farming the giant river prawn Macrobrachium rosenbergii (De Man) and the monsoon river prawn Macrobrachium malcolmsonii (H.M. Edwards) in India. Aquac Res 43:999–1014
Pillai D, Bonami JR (2012) A review on the diseases of freshwater prawns with special focus on white tail disease of Macrobrachium rosenbergii. Aquac Res 43:1029–1037
Burnett KG, Burnett LE (2015) Respiratory and metabolic impacts of crustacean immunity: are there implications for the insects? Integr Comp Biol 55:856–868
Mohanty J, Sahoo PK, Pillai BR, Mohanty S, Garnayak SK, Kumar S (2015) Purification and characterization of a β–glucan binding protein from the haemolymph of freshwater prawn Macrobrachium rosenbergii. Aquac Res 46:95–104
Lis H, Sharon N (1998) Lectins: carbohydrate-specific proteins that mediate cellular recognition. Chem Rev 98:637–674
Sánchez-Salgado JL, Pereyra MA, Agundis C, Vivanco-Rojas O, Sierra-Castillo C, Alpuche-Osorno JJ, Edgar Z (2017) Participation of lectins in crustacean immune system. Aquac Res 48:4001–4011
Wang XW, Wang JX (2013) Diversity and multiple functions of lectins in shrimp immunity. Dev Comp Immunol 39:27–38
Pohleven J, Štrukelj B, Kos J (2012) Affinity chromatography of lectins. In: Magdeldin S (ed) Affinity chromatography. Intech Open Publisher, Croatia, pp 49–74
Vazquez L, Masso F, Rosas P, Montano L, Zenteno E (1993) Purification and characterization of a lectin from Macrobrachium rosenbergii (Crustacea, Decapoda) hemolymph. Comp Biochem Physiol 105B:617–623
Vazquez L, Jaramillo L, Lascurain R, Cooper EL, Rosas P, Zenteno E (1996) Bacterial agglutination by the sialic acid specific serum lectin from Macrobrachium rosenbergii. Comp Biochem Physiol 113B:355–359
Agundis C, Pereyra A, Zenteno R, Brassart C, Sierra C, Vazquez L, Zenteno E (2000) Quantification of lectin in freshwater prawn (Macrobrachium rosenbergii) hemolymph by ELISA. Comp Biochem Physiol 127B:165–172
Zhu H, Du J, Hui KM, Liu P, Chen J, Xiu Y, Yao W, Wu T, Meng Q, Gu W, Ren Q, Wang W (2013) Diversity of lectins in Macrobrachium rosenbergii and their expression patterns under spiroplasma MR-1008 stimulation. Fish Shellfish Immunol 35:300–309
Arockiaraj J, Chaurasia MK, Kumaresan V, Palanisamy R, Harikrishnan R, Pasupuleti M, Kasi M (2015) Macrobrachium rosenbergii mannose binding lectin: synthesis of MrMBL-N20 and MrMBL-C16 peptides and their antimicrobial characterization, bioinformatics and relative gene expression analysis. Fish Shellfish Immunol 43:364–374
Huang X, Li W, Jin M, Ma FT, Huang Y, Shi YR, Zhao LL, Feng JL, Ren Q, Wang W (2016) Single CRD containing lectin from Macrobrachium rosenbergii (MrLec) participates in innate immunity against pathogen infections. Fish Shellfish Immunol 51:282–290
Huang X, Han K, Li T, Wang W, Ren Q (2018) Novel L-type lectin from fresh water prawn, Macrobrachium rosenbergii participates in antibacterial and antiviral immune responses. Fish Shellfish Immunol 77:304–311
Sudhakar GRL, Smith VRP, Rama Devi P, Prakash Vincent SG (2012) First report on a N-acetylneuraminic acid specific lectin from the marine alpheid shrimp Alpheus digitalis Complex De Haan 1844 (Crustacea: Decapoda: Alpheidae). Ital J Zool 79:482–491
Alpuche J, Pereyra A, Agundis C, Rosas C, Pascual C, Slomianny MC, Vazquez L, Zenteno E (2005) Purification and characterization of a lectin from the white shrimp Litopenaeus setiferus (Crustacea decapoda) hemolymph. Biochim Biophys Acta 1724:86–93
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Wardi AH, Michos GA (1972) Alcian blue staining of glycoproteins in acrylamide disc electrophoresis. Anal Biochem 49:607–609
Sahoo PK, Pillai BR, Mohanty J, Kumari J, Mohanty S, Mishra BK (2007) Differential affinity of natural haemagglutinin of Macrobrachium rosenbergii towards vertebrate erythrocytes: effect of sex, size and moult stage on haemagglutination titre. Indian J Exp Biol 45:830–833
Pereyra A, Agundis C, Barrera B, Alpuche J, Sierra C, Zenteno R, Zenteno E, Vázquez L (2009) The use of monoclonal antibodies anti-lectin from freshwater prawn Macrobrachium rosenbergii (DeMan, 1879) in the recognition of protein with lectin activity in Decapod's hemolymph. Prep Biochem Biotechnol 39:308–322
Jayaraj SS, Thiagaraja R, Arumugam M, Vincent S (2010) Physico-chemical characterization of bacterial and hemagglutinins from the serum of the mud crab Scylla serrata. Invertebr Surviv J 7:79–88
Acharya S, Mohanty J, Sahoo PK (2004) Humoral defence factors in Indian river prawn, Macrobrachium malcolmsonii. Fish Shellfish Immunol 17:137–147
Figueroa-Soto CG, de la Barca AMC, Vazquez-Moreno L, Higuera-Ciapara I, Yepiz-Plascencia G (1997) Purification of hemocyanin from white shrimp (Penaeus vannamei Boone) by immobilized metal affinity chromatography. Comp Biochem Physiol 117B:203–208
Jiewkok A, Tsukimura B, Utarabhand P (2015) Purification and molecular cloning of hemocyanin from Fenneropenaeus merguiensis (de Man, 1888): response to Vibrio harveyi exposure. J Crustacean Biol 35:659–669
Burmester T (2001) Molecular evolution of the arthropod hemocyanin superfamily. Mol Biol Evol 18:184–195
Zhang Z, Li R, Aweya JJ, Wang F, Zhong M, Zhang Y (2019) Identification and characterization of glycosylation sites on Litopenaeus vannamei hemocyanin. FEBS Lett 593:820–830
van Holde KE, Miller KI, Decker H (2001) Hemocyanins and invertebrate evolution. J Biol Chem 276:15563–15566
Coates CJ, Nairn J (2014) Diverse immune functions of hemocyanins. Dev Comp Immunol 45:43–55
Yao T, Zhao MM, He J, Han T, Peng W, Zhang H, Wang JY, Jiang JZ (2019) Gene expression and phenoloxidase activities of hemocyanin isoforms in response to pathogen infections in abalone Haliotis diversicolor. Int J Biol Macromol 129:538–551
Lee SY, Lee BL, Söderhäll K (2003) Processing of an antimicrobial peptide from hemocyanin of the freshwater crayfish Pacifastacus leniusculus. J Biol Chem 278:7927–7933
Zhang X, Huang C, Qin Q (2004) Antiviral properties of hemocyanin isolated from shrimp Penaeus monodon. Antiviral Res 61:93–99
Yan F, Zhang Y, Jiang R, Zhong M, Hu Z, Du H, Lun J, Chen J, Li Y (2011) Identification and agglutination properties of hemocyanin from the mud crab (Scylla serrata). Fish Shellfish Immunol 30:354–360
Zhang Y, Yan F, Hu Z, Zhao X, Min S, Du Z, Zhao S, Ye X, Li Y (2009) Hemocyanin from shrimp Litopenaeus vannamei shows hemolytic activity. Fish Shellfish Immunol 27:330–335
Ramasamy SM, Denis M, Sivakumar S, Munusami A (2017) Phenoloxidase activity in humoral plasma, hemocyanin, and hemocyanin separated proteins of the giant freshwater prawn Macrobrachium rosenbergii. Int J Biol Macromol 102:977–985
Srisuk C, Senapin S, Bendena WG, Longyant S, Sithigorngul P, Chaivisuthangkura P (2018) Molecular isolation and characterization of a haemocyanin of Macrobrachium rosenbergii reveal its antibacterial activities. Aquac Res 49:505–516
Acknowledgements
The authors thank the Director, ICAR-Central Institute of Freshwater Aquaculture, Bhubaneswar for providing necessary facilities during the study. The financial support (Grant No. BT/PR12710/AAQ/3/713/2015) received from Department of Biotechnology, Govt. of India, New Delhi, India is also duly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mohanty, J., Sahoo, S., Badhe, M.R. et al. Lectin-Like Activity of Hemocyanin in Freshwater Prawn, Macrobrachium rosenbergii. Protein J 39, 358–365 (2020). https://doi.org/10.1007/s10930-020-09912-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10930-020-09912-1