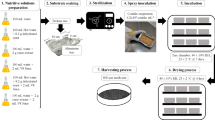
Abstract
Commercial products based on Trichoderma are obtained mainly from solid-state fermentation. Submerged liquid fermentation is the most appropriate method compared to the solid medium for large-scale production of Trichoderma spp. The present study aimed to optimize the combination of key variables that influence the liquid fermentation process of Trichoderma asperelloides LQC-96 for conidial production coupled with its efficiency in the control of Sclerotinia sclerotiorum. In addition, we verified whether the optimized culture conditions can be used for the conidial production of Trichoderma erinaceum T-12 and T-18 and Trichoderma harzianum T-15. Fermentation studies were performed in shake flasks following a planned experimental design to reduce the number of tests and consumable costs. The effect of temperature, pH, photoperiod, carbon:nitrogen ratio and water activity on conidial production were assessed, which of pH was the only meaningful factor contributing to increased conidial production of T. asperelloides LQC-96. From the five variables studied initially, pH and C:N ratio were further used in the second design (rotational central composite design—RCCD). Hence, the best conditions for the production of T. asperelloides LQC-96 conidia by liquid fermentation consisted of initial pH of 3.5, C:N ratio of 200:1 at 30 °C, without glycerol, and under 24 h photoperiod. The highest conidial concentration was observed after seven days of fermentation. Under these optimal conditions, T. erinaceum T-12 and T-18, and T. harzianum T-15 were also cultivated, but only LQC-96 efficiently parasitized S. sclerotiorum, precluding sclerotium myceliogenic germination. Our findings propose optimal fermentation conditions that maximize conidial production of T. asperelloides as a potential biofungicide against S. sclerotiorum.




Similar content being viewed by others
References
Bettiol W, Pinto ZV, Silva JC, Forner C, Faria MR, Pacífico MG, Costa LSAS (2019) Produtos comerciais à base de Trichoderma [Commercial Trichoderma-based products]. In: Meyer MC, Mazaro SM, Silva JC (eds) Trichoderma: uso na agricultura [Trichoderma: use in agriculture]. Brasília, Embrapa, pp 45–160
Bettiol W, Maffia LA, Castro MLMP (2014) Control biológico de enfermedades de plantas en Brasil. In: Bettiol W, Rivera MC, Mondino P, Montealegre JR, Colmenárez YC (eds) Control biológico de enfermedades de plantas en América Latina y el Caribe {Biological controlo f plant diseases in Latin America and the Caribbean]. [Montevidéu] Faculdad de Agronomia, Universidad de la Republica, pp 91–137. https://www.fagro.edu.uy/index.php/documentos/category/46-libros-fagro.html.
Buchs J (2001) Introduction to advantages and problems of shaken cultures. Biochem Eng J 7:91–98. https://doi.org/10.1016/S1369-703X(00)00106-6
Bueno VHP, Parra JRP, Bettiol W, van Lenteren JC (2020) Biological control in Brazil. In: van Lenteren JC, Bueno VHP, Luna MG, Colmenarez YC (eds) Biological control in Latin America and the Caribbean. CABI, Oxfordshire, pp 78–107
Brotman Y, Lisec J, Méret M, Chet I, Willmitzer L, Viterbo A (2012) Transcript and metabolite analysis of the Trichoderma-induced systemic resistance response to Pseudomonas syringae in Arabidopsis thaliana. Microbiol 158:139–146. https://doi.org/10.1099/mic.0.052621-0
Carvalho ALA, Rezende LC, Costa LB, Halfeld-Vieira BA, Pinto ZV, Morandi MAB, Medeiros FHV, Bettiol W (2018) Optimizing the mass production of Clonostachys rosea by liquid-state fermentation. Biol Control 118:16–25. https://doi.org/10.1016/j.biocontrol.2017.11.014
Couto SR, Sanromán MA (2006) Application of solid-state fermentation to food industry: a review. J Food Eng 76:291–302. https://doi.org/10.1016/j.jfoodeng.2005.05.022
Domínguez S, Rubio MR, Cardoza RE, Gutiérrez S, Nicolás C, Bettiol W, Hermosa R, Monte E (2016) Nitrogen metabolism and growth enhancement in tomato plants challenged with Trichoderma harzianum expressing the Aspergillus nidulans acetamidase amdS gene. Front Microbiol 7:1182. https://doi.org/10.3389/fmicb.2016.01182
Druzhinina IS, Seidl-Seiboth V, Herrera-Estrella A, Horwitz BA, Kenerley CM, Monte E, Mukherjee PK, Zeillinger S, Grigoriev IV, Kubicek CP (2011) Trichoderma: the genomics of opportunistic success. Nat Rev Microbiol 9:749–759. https://doi.org/10.1038/nrmicro2637
Gao L, Sun MH, Liu XZ, Che YS (2007) Effects of carbon concentration and carbon to nitrogen ratio on the growth and sporulation of several biocontrol fungi. Mycol Res 111:87–92. https://doi.org/10.1016/j.mycres.2006.07.019
Gao L, Liu XZ, Gao L, Sun MH, Li SD, Wang JL (2009) Use of a novel two-stage method to determine the effects of environmental factors on the growth and sporulation of several biocontrol fungi. Mycoscience 50:317–321. https://doi.org/10.1007/S10267-009-0483-3
Garcia RA, Juliatti FC, Cassemiro TA (2012) Produção de escleródios de Sclerotinia sclerotiorum (LIB.) de Bary em meio de cultura [Production of sclerotia on Sclerotinia sclerotiorum (Lib.) de Bary in culture media]. Biosci J 28:1–7
Harman GE (2011) Multifunctional fungal plant symbionts: new tools to enhance plant growth and productivity. New Phytol 189:647–649. https://doi.org/10.1111/j.1469-8137.2010.03614.x
Harman GE, Jin X, Stasz TE, Peruzzotti G, Leopold AC (1991) Production of conidial biomass of Trichoderma harzianum for biological control. Biol Control 1:23–28. https://doi.org/10.1016/1049-9644(91)90097-J
Hermosa R, Viterbo A, Chet I, Monte E (2012) Plant-beneficial effects of Trichoderma and of its genes. Microbiology 158:17–25. https://doi.org/10.1099/mic.0.052274-0
Jakubíková L, Farkas V, Kolarova N, Nemcovic M (2006) Conidiation of Trichoderma atroviride isolate during submerged cultivation in a laboratory stirred-tank fermenter. Folia Microbiol 51:209–213. https://doi.org/10.1007/BF02932124
Jaronski ST (2014) Mass production of entomopathogenic fungi: state of the art. In: Morales-Ramos JA, Rojas MG, Shapiro-Ilan D (eds) Mass production of beneficial organisms. Elsevier Inc, Amsterdan, pp 357–413
Jaronski ST, Mascarin GM (2016) Mass production of fungal entomopathogens. In: Lacey LA (ed) Microbial control of insect and mite pests: from theory to practice. Academic Press, New York
Jin X, Taylor AG, Harman GE (1996) Development of media and automated liquid fermentation methods to produce desiccation-tolerant propagules of Trichoderma harzianum. Biol Control 7:267–274. https://doi.org/10.1006/bcon.1996.0093
Kobori NN, Mascarin GM, Jackson MA, Schisler DA (2015) Liquid culture production of microesclerotia and submerged conidia by Trichoderma harzianum active against damping-off disease caused by Rhizoctonia solani. Fungal Biol 11:179–190. https://doi.org/10.1016/j.funbio.2014.12.005
Kolombet LV, Zhigletsova SK, Kosareva NI, Bystrova EV, Derbyshev VV, Krasnova SP, Schisler D (2008) Development of an extended shelf-life, liquid formulation of the biofungicide Trichoderma asperellum. World J Microbiol Biotechnol 24:123–131. https://doi.org/10.1007/s11274-007-9449-9
Lorito M, Woo SL, Harman GE, Monte E (2010) Translational research on Trichoderma: from ‘omics to the field. Annu Rev Phytopathol 48:395–417. https://doi.org/10.1146/annurev-phyto-073009-114314
Lopes ARO, Locatelli GO, Barbosa RM, Lobo Junior M, Mascarin GM, Finkler CLL (2020) Preparation, characterization and cell viability of encapsulated Trichoderma asperellum in alginate beads. J Microencaps 37:270–282. https://doi.org/10.1080/02652048.2020.1729884
Markets and Markets (2019) Biopesticides market by type (bioinseticides, biofungicides, and bionematicides), source (microbials, plant extracts, and beneficial insects), mode of application, formulation, crop application and region—global forecast to 2023. https://www.marketsandmarkets.com/Market-Reports/biopesticides-267.html. Accessed 04 May 2020
Mascarin GM, Jackson MA, Kobori NN, Behle RW, Dunlap CA, Delaliber Júnior I (2015) Glucose concentration alters dissolved oxygen levels in liquid cultures of Beauveria bassiana and affects formation and bioefficacy of blastospores. Appl Microbiol Biotechnol 99:6653–6665. https://doi.org/10.1007/s00253-015-6620-3
Mascarin GM, Lopes RB, Delalibera I Jr, Fernandes EKK, Luz C, Faria M (2019) Current status and perspectives of fungal entomopathogens used for microbial control of arthropod pest in Brazil. J Invert Path 165:46–52. https://doi.org/10.1016/j.jip.2018.01.001
Meyer MC, Campos HD, Godoy CV, Utiamada CM, Nunes Junior J, Goussain Junior MM, Jaccoud Filho DS, Juliatti FC, Martins MC, Venacio WS, Carneiro LC, Silva LHCP, Dias AR, Borges EP, Ito MF (2020) Mofo-granco em soja – ensaios cooperativos [White mould in soybean—cooperatives assays]. In: Meyer MC, Mazaro SM, Silva JC (eds) Trichoderma: uso na agricultura [Trichoderma: use in agriculture]. Embrapa, Brasília, pp 417–432
Monte E, Bettiol W, Hermosa R (2019) Trichoderma e seus mecanismos de ação para o controle de doenças de plantas [Trichoderma and its mechanisms of action for the control of plant diseases]. In: Meyer MC, Mazaro SM, Silva JC (eds) Trichoderma: uso na agricultura [Trichoderma: use in agriculture]. Embrapa, Brasília, pp 181–199
Montgomey DC (2005) Design and analysis of experiments, 6th edn. Wiley, New York
Rodrigues MI, Iemma AF (2009) Planejamento de experimentos e otimização de processos [Experiment planning and process optimization], 2nd edn. Casa do Espírito Amigo Fraternidade Fé e Amor, Campinas, p 358
Rubio MB, Hermosa R, Vicente R, Gómez-Acosta FA, Morcuende R, Monte E, Bettiol W (2017) The combination of Trichoderma harzianum and chemical fertilization leads to the deregulation of phytohormone networking, preventing the adaptive responses of tomato plants to salt stress. Front Plant Sci 8:294. https://doi.org/10.3389/fpls.2017.00294
Soper RS, Ward MG (1981) Production, formulation and application of fungi for insect control. In: Papavizas CG (ed) Biological control in crop production. Allanheld & Osmun, New York, pp 161–180
Sriram S, Roopa KP, Savitha MJ (2011) Extended shelf life of liquid fermentation derived talc formulation of Trichoderma harzianum with the addition of glycerol in the production medium. Crop Prot 30:1334–1339. https://doi.org/10.1016/j.cropro.2011.06.003
Steyaert JM, Weld RJ, Stewart A (2010) Ambient pH intrinsically influences Trichoderma conidiation and colony morphology. Fungal Biol 114:198–208. https://doi.org/10.1016/j.funbio.2009.12.004
Trushina N, Levin M, Mukherjee PK, Horwitz BA (2013) PacC and pH–dependent transcriptome of the mycotrophic fungus Trichoderma virens. BMC Genomics 14:138. https://doi.org/10.1186/1471-2164-14-138
van Lenteren JC, Bolckmans K, Köhl J, Ravensberg WJ, Urbaneja A (2018) Biological control using invertebrates and microorganisms: plenty of new opportunities. Biocontrol 63:39–59. https://doi.org/10.1007/s10526-017-9801-4
Verma M, Brar SK, Tyagi RD, Surampalli RY, Valéro JR (2007) Antagonistic fungi, Trichoderma spp.: panoply of biological control. Bioch Eng J 37:1–20. https://doi.org/10.1016/j.bej.2007.05.012
Watanabe S, Kato H, Kumakura K, Ishibashi E, Nagayama K (2006) Properties and biological control activities of aerial and submerged spores in Trichoderma asperellum SKT-1. J Pest Sci 31:375–379. https://doi.org/10.1584/jpestics.G06-09
Woo SL, Ruocco M, Vinale F, Nigro M, Marra R, Lombardi N, Pascale A, Lanzuise S, Manganiello G, Lorito M (2014) Trichoderma-based products and their widespread use in agriculture. The Open Mycol J 8:71–126. https://doi.org/10.2174/1874437001408010071
Zhang Y, Gao X, Liu J, Ge Y (2015) Pilot production of Clonostachys rosea conidia in a solid-state fermentor optimized using response surface methodology. Eng Life Sci 15:772–778. https://doi.org/10.1002/elsc.201400260
Zhang YY, Liu JH, Zhou YM, Zhang YY, Liu Y, Gong TY, Wang J (2013) A new two-phase kinet model of sporulation of Clonostachys rosea in a new solid-state fermentation reactor. Process Bioch 48:1119–1125. https://doi.org/10.1016/j.procbio.2013.03.009
Acknowledgments
This work was financed by Embrapa Macroprograma 3 grant number 03.10.06.005.00.00. Wagner Bettiol (CNPq 303899/2015-8), Flavio HV Medeiros (CNPq 309307/2017-1), and Bernardo A. Halfeld-Vieira (CNPq 303396/2018-0) acknowledge Conselho Nacional de Desenvolvimento Científico e Tecnológico – CNPq for the productivity fellowship CNPq. ALAC, LBC, LCR, LGS, and ZVP acknowledge Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – CAPES for the scholarship. Special thanks go to Dr. Ricardo Harakava, Instituto Biológico de São Paulo, for the molecular identification of Trichoderma strains.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by ALAC, LBC, LCR, LGS, and ZVP. Reagents/materials and analysis tools were obtained by BAHV, MABM, and WB. ALAC, LBC, LCR, LGS, FHVM, GMM, ZVP, and WB analyzed the data. The first draft of the manuscript was written by LCR and WB and all authors commented on previous versions of the manuscript. WB, GMM, BAHV, and FHVM, prepared the final version of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
de Rezende, L.C., de Andrade Carvalho, A.L., Costa, L.B. et al. Optimizing mass production of Trichoderma asperelloides by submerged liquid fermentation and its antagonism against Sclerotinia sclerotiorum. World J Microbiol Biotechnol 36, 113 (2020). https://doi.org/10.1007/s11274-020-02882-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-020-02882-7