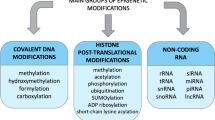
Abstract
Mammalian spermatogenesis is characterized by disproportionate germ cell apoptosis. The high frequency of apoptosis is considered a safety mechanism that serves to avoid unfavorable transmission of paternal aberrant genetic information to the offspring as well as elimination mechanism for removal of overproduced immature or damaged spermatogenic cells. The molecular mechanisms involved in the induction of germ cell apoptosis include both intrinsic mitochondrial Bcl-2/Bax and extrinsic Fas/FasL pathways. However, little is known about the nuclear trigger of those systems. Recent studies indicate that epigenomes are essential in the regulation of gene expression through remodeling of the chromatin structure, and are genome-like transmission materials that reflect the effects of various environmental factors. In spermatogenesis, epigenetic errors can act as the trigger for elimination of germ cells with abnormal chromatin structure, abnormal gene expression and/or morphological defects (disordered differentiation). In this review, we focus on the relationship between global changes in epigenetic parameters and germ cell apoptosis in mice and other mammals.




Similar content being viewed by others
References
Allard P, Colaiácovo MP (2010) Bisphenol a impairs the double-strand break repair machinery in the germline and causes chromosome abnormalities. Proc Natl Acad Sci USA 107:20405–20410. https://doi.org/10.1073/pnas.1010386107
Balhorn R (1982) A model for the structure of chromatin in mammalian sperm. J Cell Biol 93:298–305. https://doi.org/10.1083/jcb.93.2.298
Balhorn R (2007) The protamine family of sperm nuclear proteins. Genome Biol 8:227. https://doi.org/10.1186/gb-2007-8-9-227
Balhorn R, Gledhill BL, Wyrobek AJ (1977) Mouse sperm chromatin proteins: quantitative isolation and partial characterization. Biochemistry 16:4074–4080. https://doi.org/10.1021/bi00637a021
Bannister AJ, Kouzarides T (2011) Regulation of chromatin by histone modifications. Cell Res 21:381–395. https://doi.org/10.1038/cr.2011.22
Bellgrau D, Gold D, Selawry H, Moore J, Franzusoff A, Duke RC (1995) A role for CD95 ligand in preventing graft rejection. Nature 377:630–632. https://doi.org/10.1038/377630a0
Berger SL (2002) Histone modifications in transcriptional regulation. Curr Opin Genet Dev 12:142–148. https://doi.org/10.1016/s0959-437x(02)00279-4
Bernardino-Sgherri J, Flagiello D, Dutrillaux B (2002) Overall DNA methylation and chromatin structure of normal and abnormal X chromosomes. Cytogenet Genome Res 99:85–91. https://doi.org/10.1159/000071578
Bernstein BE, Kamal M, Lindblad-Toh K, Bekiranov S, Bailey DK, Huebert DJ, McMahon S, Karlsson EK, Kulbokas EJ, Gingeras TR, Schreiber SL, Lander ES (2005) Genomic maps and comparative analysis of histone modifications in human and mouse. Cell 120:169–181. https://doi.org/10.1016/j.cell.2005.01.001
Bhan A, Hussain I, Ansari KI, Bobzean SA, Perrotti LI, Mandal SS (2014) Histone methyltransferase EZH2 is transcriptionally induced by estradiol as well as estrogenic endocrine disruptors bisphenol-A and diethylstilbestrol. J Mol Biol 426:3426–3441. https://doi.org/10.1016/j.jmb.2014.07.025
Boivin AJ, Momparler LF, Hurtubise A, Momparler RL (2002) Antineoplastic action of 5-aza-2′-deoxycytidine and phenylbutyrate on human lung carcinoma cells. Anticancer Drugs 13:869–874. https://doi.org/10.1097/00001813-200209000-00013
Bourc’his D, Bestor TH (2004) Meiotic catastrophe and retrotransposon reactivation in male germ cells lacking Dnmt3L. Nature 431:96–99. https://doi.org/10.1038/nature02886
Branco MR, Ficz G, Reik W (2011) Uncovering the role of 5-hydroxymethylcytosine in the epigenome. Nat Rev Genet 13:7–13. https://doi.org/10.1038/nrg3080
Brykczynska U, Hisano M, Erkek S, Ramos L, Oakeley EJ, Roloff TC, Beisel C, Schübeler D, Stadler MB, Peters AH (2010) Repressive and active histone methylation mark distinct promoters in human and mouse spermatozoa. Nat Struct Mol Biol 17:679–687. https://doi.org/10.1038/nsmb.1821
Burlibaşa L, Ionescu AC, Dragusanu DM (2019) Histone hyperacetylation and DNA methylation interplay during murine spermatogenesis. Zygote 27:305–314. https://doi.org/10.1017/S0967199419000303
Calvaruso G, Carabillò M, Giuliano M, Lauricella M, D’Anneo A, Vento R, Tesoriere G (2001) Sodium phenylbutyrate induces apoptosis in human retinoblastoma Y79 cells: the effect of combined treatment with the topoisomerase I-inhibitor topotecan. Int J Oncol 18:1233–1237. https://doi.org/10.3892/ijo.18.6.1233
Chen X, Song N, Matsumoto K, Nanashima A, Nagayasu T, Hayashi T, Ying M, Endo D, Wu Z, Koji T (2013) High expression of trimethylated histone H3 at lysine 27 predicts better prognosis in non-small cell lung cancer. Int J Oncol 43:1467–1480. https://doi.org/10.3892/ijo.2013.2062
Chipuk JE, Kuwana T, Bouchier-Hayes L, Droin NM, Newmeyer DD, Schuler M, Green DR (2004) Direct activation of Bax by p53 mediates mitochondrial membrane permeabilization and apoptosis. Science 303:1010–1014. https://doi.org/10.1126/science.1092734
Dai L, Endo D, Akiyama N, Yamamoto-Fukuda T, Koji T (2015) Aberrant levels of histone H3 acetylation induce spermatid anomaly in mouse testis. Histochem Cell Biol 143:209–224. https://doi.org/10.1007/s00418-014-1283-1
Damavandi E, Hishikawa Y, Izumi S-I, Shin M, Koji T (2002) Involvement of bax redistribution in the induction of germ cell apoptosis in neonatal mouse testes. Acta Histochem Cytochem 35:449–459. https://doi.org/10.1679/aohc.66.1
De Rooij DG, Lok D (1987) Regulation of the density of spermatogonia in the seminiferous epithelium of the Chinese hamster: II, differentiating spermatogonia. Anat Rec 217:131–136. https://doi.org/10.1002/ar.1092170204
de Ruijter AJ, van Gennip AH, Caron HN, Kemp S, van Kuilenburg AB (2003) Histone deacetylases (HDACs): characterization of the classical HDAC family. Biochem J 370:737–749. https://doi.org/10.1042/BJ20021321
Deininger PL, Moran JV, Batzer MA, Kazazian HH (2003) Mobile elements and mammalian genome evolution. Curr Opin Genet Dev 13:651–658. https://doi.org/10.1016/j.gde.2003.10.013
D’Souza R, Gill-Sharma MK, Pathak S, Kedia N, Kumar R, Balasinor N (2005) Effect of high intratesticular estrogen on the seminiferous epithelium in adult male rats. Mol Cell Endocrinol 241:41–48. https://doi.org/10.1016/j.mce.2005.04.011
Dumasia K, Kumar A, Deshpande S, Balasinor NH (2017) Estrogen, through estrogen receptor 1, regulates histone modifications and chromatin remodeling during spermatogenesis in adult rats. Epigenetics 12:953–963. https://doi.org/10.1080/15592294.2017.1382786
Feinman R, Clarke KO, Harrison LE (2002) Phenylbutyrate-induced apoptosis is associated with inactivation of NF-kappaB IN HT-29 colon cancer cells. Cancer Chemother Pharmacol 49:27–34. https://doi.org/10.1007/s00280-001-0390-6
Gabbara S, Bhagwat AS (1995) The mechanism of inhibition of DNA (cytosine-5-)-methyltransferases by 5-azacytosine is likely to involve methyl transfer to the inhibitor. Biochem J 307:87–92. https://doi.org/10.1042/bj3070087
Gatewood JM, Cook GR, Balhorn R, Bradbury EM, Schmid CW (1987) Sequence-specific packaging of DNA in human sperm chromatin. Science 236:962–964. https://doi.org/10.1126/science.3576213
Gaysinskaya V, Miller BF, De Luca C, van der Heijden GW, Hansen KD, Bortvin A (2018) Transient reduction of DNA methylation at the onset of meiosis in male mice. Epigenetics Chromatin 11(1):15. https://doi.org/10.1186/s13072-018-0186-0
Giles RH, Peters DJ, Breuning MH (1998) Conjunction dysfunction: CBP/p300 in human disease. Trends Genet 14:178–183. https://doi.org/10.1016/s0168-9525(98)01438-3
Godmann M, Auger V, Ferraroni-Aguiar V, Di Sauro A, Sette C, Behr R, Kimmins S (2007) Dynamic regulation of histone H3 methylation at lysine 4 in mammalian spermatogenesis. Biol Reprod 77:754–764. https://doi.org/10.1095/biolreprod.107.062265
Goldberg AD, Allis CD, Bernstein E (2007) Epigenetics: a landscape takes shape. Cell 128:635–638. https://doi.org/10.1016/j.cell.2007.02.006
Govin J, Caron C, Lestrat C, Rousseaux S, Khochbin S (2004) The role of histones in chromatin remodelling during mammalian spermiogenesis. Eur J Biochem 271:3459–3469. https://doi.org/10.1111/j.1432-1033.2004.04266.x
Guerrero-Bosagna C, Skinner MK (2012) Environmentally induced epigenetic transgenerational inheritance of phenotype and disease. Mol Cell Endocrinol 354:3–8. https://doi.org/10.1016/j.mce.2011.10.004
Hata K, Kusumi M, Yokomine T, Li E, Sasaki H (2006) Meiotic and epigenetic aberrations in Dnmt3L-deficient male germ cells. Mol Reprod Dev 73:116–122. https://doi.org/10.1002/mrd.20387
Hazzouri M, Pivot-Pajot C, Faure AK, Usson Y, Pelletier R, Sèle B, Khochbin S, Rousseaux S (2000) Regulated hyperacetylation of core histones during mouse spermatogenesis: involvement of histone deacetylases. Eur J Cell Biol 79:950–960. https://doi.org/10.1078/0171-9335-00123
Hill PW, Amouroux R, Hajkova P (2014) DNA demethylation, Tet proteins and 5-hydroxymethylcytosine in epigenetic reprogramming: an emerging complex story. Genomics 104:324–333. https://doi.org/10.1016/j.ygeno.2014.08.012
Hishikawa Y, An S, Yamamoto-Fukuda T, Shibata Y, Koji T (2009) Improvement of in situ PCR by optimization of PCR cycle number and proteinase k concentration: localization of x chromosome-linked phosphoglycerate kinase-1 gene in mouse reproductive organs. Acta Histochem Cytochem 42:15–21. https://doi.org/10.1267/ahc.09011
Iannitti T, Palmieri B (2011) Clinical and experimental applications of sodium phenylbutyrate. Drugs R D 11:227–249. https://doi.org/10.2165/11591280-000000000-00000
Jones PA (2012) Functions of DNA methylation: islands, start sites, gene bodies and beyond. Nat Rev Genet 13:484–492. https://doi.org/10.1038/nrg3230
Jue K, Bestor TH, Trasler JM (1995) Regulated synthesis and localization of DNA methyltransferase during spermatogenesis. Biol Reprod 53:561–569. https://doi.org/10.1095/biolreprod53.3.561
Jüttermann R, Li E, Jaenisch R (1994) Toxicity of 5-aza-2′-deoxycytidine to mammalian cells is mediated primarily by covalent trapping of DNA methyltransferase rather than DNA demethylation. Proc Natl Acad Sci USA 91:11797–11801. https://doi.org/10.1073/pnas.91.25.11797
Kaneda M, Okano M, Hata K, Sado T, Tsujimoto N, Li E, Sasaki H (2004) Essential role for de novo DNA methyltransferase Dnmt3a in paternal and maternal imprinting. Nature 429:900–903. https://doi.org/10.1038/nature02633
Kato Y, Kaneda M, Hata K, Kumaki K, Hisano M, Kohara Y, Okano M, Li E, Nozaki M, Sasaki H (2007) Role of the Dnmt3 family in de novo methylation of imprinted and repetitive sequences during male germ cell development in the mouse. Hum Mol Genet 16:2272–2280. https://doi.org/10.1093/hmg/ddm179
Kebede AF, Schneider R, Daujat S (2015) Novel types and sites of histone modifications emerge as players in the transcriptional regulation contest. FEBS J 282:1658–1674. https://doi.org/10.1111/febs.13047
Kelly TL, Li E, Trasler JM (2003) 5-aza-2′-deoxycytidine induces alterations in murine spermatogenesis and pregnancy outcome. J Androl 24:822–830. https://doi.org/10.1002/j.1939-4640.2003.tb03133.x
Khalil AM, Wahlestedt C (2008) Epigenetic mechanisms of gene regulation during mammalian spermatogenesis. Epigenetics 3:21–28. https://doi.org/10.4161/epi.3.1.5555
Kitazawa S, Haraguchi R, Kitazawa R (2018) Morphology-oriented epigenetic research. Histochem Cell Biol 150:3–12. https://doi.org/10.1007/s00418-018-1675-8
Koji T (2000) Molecular histochemical techniques. Springer-Verlag, Tokyo. https://doi.org/10.1007/978-4-431-67915-8
Koji T (2001) Male germ cell death in mouse testes: possible involvement of Fas and Fas ligand. Med Electron Microsc 34:213–222. https://doi.org/10.1007/s007950100018
Koji T, Hishikawa Y (2003) Germ cell apoptosis and its molecular trigger in mouse testes. Arch Histol Cytol 66:1–16. https://doi.org/10.1679/aohc.66.1
Koji T, Kondo S, Hishikawa Y, An S, Sato Y (2008) In situ detection of methylated DNA by histo endonuclease-linked detection of methylated DNA sites: a new principle of analysis of DNA methylation. Histochem Cell Biol 130:917–925. https://doi.org/10.1007/s00418-008-0487-7
Kondo T, Goto S, Ihara Y, Urata Y, Ikeda S, Hishikawa Y, Izumi S, Shin M, Koji T (2002) Diethylstilbestrol attenuates antioxidant activities in testis from male mice. Free Radic Res 36:957–966. https://doi.org/10.1080/107157621000006617
Kouzarides T (2007) Chromatin modifications and their function. Cell 128:693–705. https://doi.org/10.1016/j.cell.2007.02.005
Kuo MH, Allis CD (1998) Roles of histone acetyltransferases and deacetylases in gene regulation. BioEssays 20:615–626. https://doi.org/10.1002/(SICI)1521-1878(199808)20:8%3c615:AID-BIES4%3e3.0.CO;2-H
La Salle S, Trasler JM (2006) Dynamic expression of DNMT3a and DNMT3b isoforms during male germ cell development in the mouse. Dev Biol 296:71–82. https://doi.org/10.1016/j.ydbio.2006.04.436
Lee B, Rhead W, Diaz GA, Scharschmidt BF, Mian A, Shchelochkov O, Marier JF, Beliveau M, Mauney J, Dickinson K, Martinez A, Gargosky S, Mokhtarani M, Berry SA (2010) Phase 2 comparison of a novel ammonia scavenging agent with sodium phenylbutyrate in patients with urea cycle disorders: safety, pharmacokinetics and ammonia control. Mol Genet Metab 100:221–228. https://doi.org/10.1016/j.ymgme.2010.03.014
Li M, Liu C, Yang L, Zhang L, Chen C, He M, Lu Y, Feng W, Pi H, Zhang Y, Zhong M, Yu Z, Zhou Z (2016) G9a-mediated histone methylation regulates cadmium-induced male fertility damage in pubertal mice. Toxicol Lett 252:11–21. https://doi.org/10.1016/j.toxlet.2016.04.004
Li H, Zhang P, Zhao Y, Zhang H (2020) Low doses of carbendazim and chlorothalonil synergized to impair mouse spermatogenesis through epigenetic pathways. Ecotoxicol Environ Saf 188:109908. https://doi.org/10.1016/j.ecoenv.2019.109908
Liu Y, Lu C, Yang Y, Fan Y, Yang R, Liu CF, Korolev N, Nordenskiöld L (2011) Influence of histone tails and H4 tail acetylations on nucleosome-nucleosome interactions. J Mol Biol 414:749–764. https://doi.org/10.1016/j.jmb.2011.10.031
Lv Y, Zhang P, Guo J, Zhu Z, Li X, Xu D, Zeng W (2018) Melatonin protects mouse spermatogonial stem cells against hexavalent chromium-induced apoptosis and epigenetic histone modification. Toxicol Appl Pharmacol 340:30–38. https://doi.org/10.1016/j.taap.2017.12.017
Maatouk DM, Kellam LD, Mann MR, Lei H, Li E, Bartolomei MS, Resnick JL (2006) DNA methylation is a primary mechanism for silencing postmigratory primordial germ cell genes in both germ cell and somatic cell lineages. Development 133:3411–3418. https://doi.org/10.1242/dev.02500
Marques CJ, João Pinho M, Carvalho F, Bièche I, Barros A, Sousa M (2011) DNA methylation imprinting marks and DNA methyltransferase expression in human spermatogenic cell stages. Epigenetics 6:1354–1361. https://doi.org/10.4161/epi.6.11.17993
Ménézo Y, Entezami F, Lichtblau I, Belloc S, Cohen M, Dale B (2014) Oxidative stress and fertility: incorrect assumptions and ineffective solutions? Zygote 22:80–90. https://doi.org/10.1017/S0967199412000263
Meyer RG, Ketchum CC, Meyer-Ficca ML (2017) Heritable sperm chromatin epigenetics: a break to remember. Biol Reprod 97:784–797. https://doi.org/10.1093/biolre/iox137
Milkevitch M, Shim H, Pilatus U, Pickup S, Wehrle JP, Samid D, Poptani H, Glickson JD, Delikatny EJ (2005) Increases in NMR-visible lipid and glycerophosphocholine during phenylbutyrate-induced apoptosis in human prostate cancer cells. Biochim Biophys Acta 1734:1–12. https://doi.org/10.1016/j.bbalip.2005.01.008
Mínguez-Alarcón L, Hauser R, Gaskins AJ (2016) Effects of bisphenol a on male and couple reproductive health: a review. Fertil Steril 106:864–870. https://doi.org/10.1016/j.fertnstert.2016.07.1118
Miyashita T, Krajewski S, Krajewska M, Wang HG, Lin HK, Liebermann DA, Hoffman B, Reed JC (1994) Tumor suppressor p53 is a regulator of bcl-2 and bax gene expression in vitro and in vivo. Oncogene 9:1799–1805
Mizzen CA, Yang XJ, Kokubo T, Brownell JE, Bannister AJ, Owen-Hughes T, Workman J, Wang L, Berger SL, Kouzarides T, Nakatani Y, Allis CD (1996) The TAF(II)250 subunit of TFIID has histone acetyltransferase activity. Cell 87:1261–1270. https://doi.org/10.1016/s0092-8674(00)81821-8
Monesi V (1967) Ribonucleic acid and protein synthesis during differentiation of male germ cells in the mouse. Arch Anat Microsc Morphol Exp 56:61–74
Mu W, Starmer J, Fedoriw AM, Yee D, Magnuson T (2014) Repression of the soma-specific transcriptome by Polycomb-repressive complex 2 promotes male germ cell development. Genes Dev 28:2056–2069. https://doi.org/10.1101/gad.246124.114
Natsume-Kitatani Y, Shiga M, Mamitsuka H (2011) Genome-wide integration on transcription factors, histone acetylation and gene expression reveals genes co-regulated by histone modification patterns. PLoS ONE 6:e22281. https://doi.org/10.1371/journal.pone.0022281
Neal KC, Pannuti A, Smith ER, Lucchesi JC (2000) A new human member of the MYST family of histone acetyl transferases with high sequence similarity to Drosophila MOF. Biochim Biophys Acta 1490:170–174. https://doi.org/10.1016/s0167-4781(99)00211-0
Neuwald AF, Landsman D (1997) GCN5-related histone N-acetyltransferases belong to a diverse superfamily that includes the yeast SPT10 protein. Trends Biochem Sci 22:154–155. https://doi.org/10.1016/s0968-0004(97)01034-7
Ni K, Dansranjavin T, Rogenhofer N, Oeztuerk N, Deuker J, Bergmann M, Schuppe HC, Wagenlehner F, Weidner W, Steger K, Schagdarsurengin U (2016) TET enzymes are successively expressed during human spermatogenesis and their expression level is pivotal for male fertility. Hum Reprod 31:1411–1424. https://doi.org/10.1093/humrep/dew096
Oakes CC, La Salle S, Smiraglia DJ, Robaire B, Trasler JM (2007) Developmental acquisition of genome-wide DNA methylation occurs prior to meiosis in male germ cells. Dev Biol 307:368–379. https://doi.org/10.1016/j.ydbio.2007.05.002
Oka M, Meacham AM, Hamazaki T, Rodić N, Chang LJ, Terada N (2005) De novo DNA methyltransferases Dnmt3a and Dnmt3b primarily mediate the cytotoxic effect of 5-aza-2′-deoxycytidine. Oncogene 24:3091–3099. https://doi.org/10.1038/sj.onc.1208540
Okamoto A, Tainaka K, Kamei T (2006) Sequence-selective osmium oxidation of DNA: efficient distinction between 5-methylcytosine and cytosine. Org Biomol Chem 4:1638–1640. https://doi.org/10.1039/b600401f
Okano M, Bell DW, Haber DA, Li E (1999) DNA methyltransferases Dnmt3a and Dnmt3b are essential for de novo methylation and mammalian development. Cell 99:247–257. https://doi.org/10.1016/s0092-8674(00)81656-6
Oliva R (2006) Protamines and male infertility. Hum Reprod Update 12:417–435. https://doi.org/10.1093/humupd/dml009
Peters AH, O’Carroll D, Scherthan H, Mechtler K, Sauer S, Schöfer C, Weipoltshammer K, Pagani M, Lachner M, Kohlmaier A, Opravil S, Doyle M, Sibilia M, Jenuwein T (2001) Loss of the Suv39h histone methyltransferases impairs mammalian heterochromatin and genome stability. Cell 107:323–337. https://doi.org/10.1016/s0092-8674(01)00542-6
Powers NR, Parvanov ED, Baker CL, Walker M, Petkov PM, Paigen K (2016) The meiotic recombination activator PRDM9 trimethylates both H3K36 and H3K4 at recombination hotspots in vivo. PLoS Genet 12:e1006146. https://doi.org/10.1371/journal.pgen.1006146
Pradhan S, Chin HG, Estève PO, Jacobsen SE (2009) SET7/9 mediated methylation of non-histone proteins in mammalian cells. Epigenetics 4:383–387. https://doi.org/10.4161/epi.4.6.9450
Rathke C, Baarends WM, Awe S, Renkawitz-Pohl R (2014) Chromatin dynamics during spermiogenesis. Biochim Biophys Acta 1839:155–168. https://doi.org/10.1016/j.bbagrm.2013.08.004
Rodriguez I, Ody C, Araki K, Garcia I, Vassalli P (1997) An early and massive wave of germinal cell apoptosis is required for the development of functional spermatogenesis. EMBO J 16:2262–2270. https://doi.org/10.1093/emboj/16.9.2262
Rousseaux S, Reynoird N, Escoffier E, Thevenon J, Caron C, Khochbin S (2008) Epigenetic reprogramming of the male genome during gametogenesis and in the zygote. Reprod Biomed Online 16:492–503. https://doi.org/10.1016/s1472-6483(10)60456-7
Russell LD, Chiarini-Garcia H, Korsmeyer SJ, Knudson CM (2002) Bax-dependent spermatogonia apoptosis is required for testicular development and spermatogenesis. Biol Reprod 66:950–958. https://doi.org/10.1095/biolreprod66.4.950
Russo VEA, Martienssen RA, Riggs AD (1996) Epigenetic mechanisms of gene regulation. Cold Spring Harbor Laboratory Press, New York. https://doi.org/10.1017/S0016672397229320
Saitou M, Kurimoto K (2014) Paternal nucleosomes: are they retained in developmental promoters or gene deserts? Dev Cell 30:6–8. https://doi.org/10.1016/j.devcel.2014.06.025
Sakai Y, Suetake I, Itoh K, Mizugaki M, Tajima S, Yamashina S (2001) Expression of DNA methyltransferase (Dnmt1) in testicular germ cells during development of mouse embryo. Cell Struct Funct 26:685–691. https://doi.org/10.1247/csf.26.685
Schulze C (1988) Response of the human testis to long-term estrogen treatment: morphology of Sertoli cells, Leydig cells and spermatogonial stem cells. Cell Tissue Res 251:31–43. https://doi.org/10.1007/BF00215444
Seisenberger S, Andrews S, Krueger F, Arand J, Walter J, Santos F, Popp C, Thienpont B, Dean W, Reik W (2012) The dynamics of genome-wide DNA methylation reprogramming in mouse primordial germ cells. Mol Cell 48:849–862. https://doi.org/10.1016/j.molcel.2012.11.001
Shi M, Sekulovski N, MacLean JA, Hayashi K (2018) Prenatal exposure to bisphenol a analogues on male reproductive functions in mice. Toxicol Sci 163:620–631. https://doi.org/10.1093/toxsci/kfy061
Shima JE, McLean DJ, McCarrey JR, Griswold MD (2004) The murine testicular transcriptome: characterizing gene expression in the testis during the progression of spermatogenesis. Biol Reprod 71:319–330. https://doi.org/10.1095/biolreprod.103.026880
Söderström KO, Parvinen M (1976) RNA synthesis in different stages of rat seminiferous epithelial cycle. Mol Cell Endocrinol 5:181–199. https://doi.org/10.1016/0303-7207(76)90082-4
Song N, Liu J, An S, Nishino T, Hishikawa Y, Koji T (2011) Immunohistochemical analysis of histone H3 modifications in germ cells during mouse spermatogenesis. Acta Histochem Cytochem 44:183–190. https://doi.org/10.1267/ahc.11027
Song N, Endo D, Koji T (2014) Roles of epigenome in mammalian spermatogenesis. Reprod Med Biol 13:59–69. https://doi.org/10.1007/s12522-013-0167-8
Song N, Endo D, Song B, Shibata Y, Koji T (2016) 5-aza-2′-deoxycytidine impairs mouse spermatogenesis at multiple stages through different usage of DNA methyltransferases. Toxicology 361–362:62–72. https://doi.org/10.1016/j.tox.2016.07.005
Sorm F, Pískala A, Cihák A, Veselý J (1964) 5-Azacytidine, a new, highly effective cancerostatic. Experientia 20:202–203. https://doi.org/10.1007/BF02135399
Spencer TE, Jenster G, Burcin MM, Allis CD, Zhou J, Mizzen CA, McKenna NJ, Onate SA, Tsai SY, Tsai MJ, O’Malley BW (1997) Steroid receptor coactivator-1 is a histone acetyltransferase. Nature 389:194–198. https://doi.org/10.1038/38304
Stewart KR, Veselovska L, Kelsey G (2016) Establishment and functions of DNA methylation in the germline. Epigenomics 8:1399–1413. https://doi.org/10.2217/epi-2016-0056
Tachibana M, Nozaki M, Takeda N, Shinkai Y (2007) Functional dynamics of H3K9 methylation during meiotic prophase progression. EMBO J 26:3346–3359. https://doi.org/10.1038/sj.emboj.7601767
Takada Y, Naruse C, Costa Y, Shirakawa T, Tachibana M, Sharif J, Kezuka-Shiotani F, Kakiuchi D, Masumoto H, Shinkai Y, Ohbo K, Peters AH, Turner JM, Asano M, Koseki H (2011) HP1γ links histone methylation marks to meiotic synapsis in mice. Development 138:4207–4217. https://doi.org/10.1242/dev.064444
Takashima S, Takehashi M, Lee J, Chuma S, Okano M, Hata K, Suetake I, Nakatsuji N, Miyoshi H, Tajima S, Tanaka Y, Toyokuni S, Sasaki H, Kanatsu-Shinohara M, Shinohara T (2009) Abnormal DNA methyltransferase expression in mouse germline stem cells results in spermatogenic defects. Biol Reprod 81:155–164. https://doi.org/10.1095/biolreprod.108.074708
Tun N, Shibata Y, Soe MT, Htun MW, Koji T (2019) Histone deacetylase inhibitors suppress transdifferentiation of gonadotrophs to prolactin cells and proliferation of prolactin cells induced by diethylstilbestrol in male mouse pituitary. Histochem Cell Biol 151:291–303. https://doi.org/10.1007/s00418-018-1760-z
Uetake Y, Sluder G (2018) Activation of the apoptotic pathway during prolonged prometaphase blocks daughter cell proliferation. Mol Biol Cell 29:2632–2643. https://doi.org/10.1091/mbc.E18-01-0026
Urdinguio RG, Bayón GF, Dmitrijeva M, Toraño EG, Bravo C, Fraga MF, Bassas L, Larriba S, Fernández AF (2015) Aberrant DNA methylation patterns of spermatozoa in men with unexplained infertility. Hum Reprod 30:1014–1028. https://doi.org/10.1093/humrep/dev053
Waddington C (1942) The epigenotype. Endeavour 1:18–20
Wang RA, Nakane PK, Koji T (1998) Autonomous cell death of mouse male germ cells during fetal and postnatal period. Biol Reprod 58:1250–1256. https://doi.org/10.1095/biolreprod58.5.1250
Wang L, Xu Z, Khawar MB, Liu C, Li W (2017) The histone codes for meiosis. Reproduction 154:R65–R79. https://doi.org/10.1530/REP-17-0153
Webster KE, O’Bryan MK, Fletcher S, Crewther PE, Aapola U, Craig J, Harrison DK, Aung H, Phutikanit N, Lyle R, Meachem SJ, Antonarakis SE, de Kretser DM, Hedger MP, Peterson P, Carroll BJ, Scott HS (2005) Meiotic and epigenetic defects in Dnmt3L-knockout mouse spermatogenesis. Proc Natl Acad Sci USA 102:4068–4073. https://doi.org/10.1073/pnas.0500702102
Wei Y, Schatten H, Sun QY (2015) Environmental epigenetic inheritance through gametes and implications for human reproduction. Hum Reprod Update 21:194–208. https://doi.org/10.1093/humupd/dmu061
Western PS, Miles DC, van den Bergen JA, Burton M, Sinclair AH (2008) Dynamic regulation of mitotic arrest in fetal male germ cells. Stem Cells 26:339–347. https://doi.org/10.1634/stemcells.2007-0622
Wittschieben BO, Otero G, de Bizemont T, Fellows J, Erdjument-Bromage H, Ohba R, Li Y, Allis CD, Tempst P, Svejstrup JQ (1999) A novel histone acetyltransferase is an integral subunit of elongating RNA polymerase II holoenzyme. Mol Cell 4:123–128. https://doi.org/10.1016/s1097-2765(00)80194-x
Xie M, Bu P, Li F, Lan S, Wu H, Yuan L, Wang Y (2016) Neonatal bisphenol a exposure induces meiotic arrest and apoptosis of spermatogenic cells. Oncotarget 7:10606–10615. https://doi.org/10.18632/oncotarget.7218
Yamamoto-Fukuda T, Hishikawa Y, Shibata Y, Kobayashi T, Takahashi H, Koji T (2010) Pathogenesis of middle ear cholesteatoma: a new model of experimentally induced cholesteatoma in Mongolian gerbils. Am J Pathol 176:2602–2606. https://doi.org/10.2353/ajpath.2010.091182
Yin Y, Stahl BC, DeWolf WC, Morgentaler A (2002) P53 and fas are sequential mechanisms of testicular germ cell apoptosis. J Androl 23:64–70. https://doi.org/10.1002/jand.2002.23.1.64
Yu KH, Weng LJ, Fu S, Piantadosi S, Gore SD (1999) Augmentation of phenylbutyrate-induced differentiation of myeloid leukemia cells using all-trans retinoic acid. Leukemia 13:1258–1265. https://doi.org/10.1038/sj.leu.2401468
Zamudio N, Barau J, Teissandier A, Walter M, Borsos M, Servant N, Bourc’his D (2015) DNA methylation restrains transposons from adopting a chromatin signature permissive for meiotic recombination. Genes Dev 29(12):1256–1270. https://doi.org/10.1101/gad.257840.114
Zhang X, Ho SM (2011) Epigenetics meets endocrinology. J Mol Endocrinol 46:R11–32. https://doi.org/10.1677/jme-10-0053
Zheng J, Xia X, Ding H, Yan A, Hu S, Gong X, Zong S, Zhang Y, Sheng HZ (2008) Erasure of the paternal transcription program during spermiogenesis: the first step in the reprogramming of sperm chromatin for zygotic development. Dev Dyn 237:1463–1476. https://doi.org/10.1002/dvdy.21499
Zheng H, Zhou X, Li DK, Yang F, Pan H, Li T, Miao M, Li R, Yuan W (2017) Genome-wide alteration in DNA hydroxymethylation in the sperm from bisphenol A-exposed men. PLoS ONE 12:e0178535. https://doi.org/10.1371/journal.pone.0178535
Acknowledgements
This work was partially supported by a Grant-in-Aid for Scientific Research from the Japanese Ministry of Education, Science, Sports, and Culture (No. 20K07246 to T. Koji).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
We declare that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Histochemistry and Cell Biology Lecture to be presented at the 16th International Congress of Histochemistry and Cytochemistry in Prague, Czech Republic
Rights and permissions
About this article
Cite this article
Koji, T., Shibata, Y. Global changes in epigenomes during mouse spermatogenesis: possible relation to germ cell apoptosis. Histochem Cell Biol 154, 123–134 (2020). https://doi.org/10.1007/s00418-020-01900-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-020-01900-x