Abstract
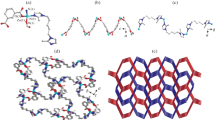
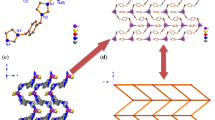
Three Co(II) metal–organic frameworks (Co-MOFs) build by 4,4′-bis(imidazolyl)diphenyl ether (bidpe), m-Phthalic acid (H2mpa), 5-Nitroisophthalic acid (H2nmpa) and 5-sulfonatobenzene-1,3-dicarboxylate (H2smpa), namely: [Co(mpa)(bidpe)]n (1), [Co(nmpa)(bidpe)]n (2), {[Co1.5(smpa)(bidpe)2]·1.5H2O·0.25DMA}n (3), were synthesized under same solvothermal condition. The compounds have been characterized by elemental analysis, IR spectroscopy, single-crystal X-ray diffraction, thermogravimetric (TG) analysis and powder X-ray diffraction (PXRD). The single-crystal X-ray diffraction analysis reveals that compound 1 shows a 4-connected sql net with Schläfli point symbol (44·62). Compound 2 displays a 3, 5-connected net with Schläfli point symbol (42·67·8)(42·6). Compound 3 possesses a 3-connected net with Schläfli point symbol (42·6). The photocatalytic investigations indicate that compounds 1–3 afford good photocatalytic capabilities in degradation of methylene blue (MB) and methylene violet (MV). Furthermore, the possible photocatalytic mechanisms have been discussed.







Similar content being viewed by others
References
C. Belpaire, T. Reyns, C. Geeraerts, J.V. Loco, Chemosphere 138, 784 (2015)
Y. Shao, X. Wang, Y. Kang, Y. Shu, Q. Sun, L. Li, J. Colloid Interface Sci. 25, 429 (2014)
L.S. Cui, C.H. Li, D.P. Qin, F. Liu, J.Q. Long, J. Mol. Struct. 269, 1177 (2019)
Z.A. Zong, C.B. Fan, X. Zhang, X.M. Meng, F. Jin, Y.H. Fan, CrystEngComm 21, 673 (2019)
J.M. Sylvia, J.A. Janni, J.D. Klein, K.M. Spencer, Anal. Chem. 72, 5834 (2000)
R.D. Luggar, M.J. Farquharson, J.A. Horrocks, R.J. Lacey, X-Ray Spectrom. 27, 87 (1998)
Y. Wu, G.P. Yang, Y. Zhao et al., Dalton Trans. 44, 3271 (2015)
Y.J. Cui, Y.F. Yue, G.D. Qian, B.L. Chen, Chem. Rev. 2, 703 (2012)
H. Yang, F. Wang, Y.X. Tan, T.H. Li, J. Zhang, Chem. Asian J. 7, 1069 (2012)
M.J. Sie, Y.J. Chang, P.W. Cheng, P.T. Kuo, C.W. Yeh, C.F. Cheng, J.D. Chen, J.C. Wang, CrystEngComm 14, 5505 (2012)
Y.W. Li, H. Ma, Y.Q. Chen, K.H. He, Z.X. Li, X.H. Bu, Cryst. Growth Des. 12, 189 (2012)
Z.Y. Du, H.B. Xu, J.G. Mao, Inorg. Chem. 45, 6424 (2006)
P. Kar, P. Jain, V. Kumar, R.K. Gupta, J. Environ. Chem. Eng. 7, 102843 (2019)
N. Singh, J. Prakash, M. Misra, A. Sharma, R.K. Gupta, ACS Appl. Mater. Interfaces 9, 28495 (2017)
P. Jain, A. Kumar, N. Verma, R.K. Gupta, Sol. Energy 189, 35–44 (2019)
N. Singh, R. Chakraborty, R.K. Gupta, J. Environ. Chem. Eng. 6, 459 (2018)
S.R. Venna, M.A. Carreon, J. Am. Chem. Soc. 132, 76 (2010)
P. Horcajada, C. Serre, G. Maurin, N.A. Ramsahye, F. Balas, M. Vallet-Regi, G. Ferey, J. Am. Chem. Soc. 30, 6774 (2008)
J.W. Canary, S. Mortezaei, J. Liang, Coord. Chem. Rev. 254, 2249 (2010)
H. Furukawa, K.E. Cordova, M. O’Keeffe, O.M. Yaghi, Science 341, 1230444 (2013)
K. Kim, D. Whang, H. Lee, S.I. Jun, J. Oh, Y.J. Jeon, J.S. Seo, Nature 404, 982 (2000)
P. Horcajada, T. Chalati, C. Serre, B. Gillet, C. Sebrie, T. Baati, J.S. Chang et al., Nat. Mater. 9, 172 (2010)
X.Y. Dong, M. Zhang, R.B. Pei, Q. Wang, D.H. Wei, S.Q. Zang, T.C. Mak et al., Angew. Chem. Int. Ed. 55, 2073 (2016)
X. Yang, D. Yan, Chem. Sci. 7, 4519 (2010)
G. Fan, D. Yan, Sci. Rep. 4, 4933 (2015)
D. Yan, G.O. Lloyd, A. Delori, W. Jones, X. Duan, ChemPlusChem 77, 1112 (2012)
M.F. Wang, Y. Mi, F.L. Hu, Z. Niu, X.H. Yin, Q. Huang, H.F. Wang, J.P. Lang, J. Am. Chem. Soc. 142, 700 (2020)
F.L. Li, P.T. Wang, X.Q. Huang, D.J. Young, H.F. Wang, P. Braunstein, J.P. Lang, Angew. Chem. Int. Ed. 58, 7051 (2019)
T. Zhang, F.J. Song, W.B. Lin, Chem. Commun. 48, 8766 (2012)
J.W. Liu, L.F. Chen, H. Cui, J.Y. Zhang, L. Zhang, C.Y. Su, Chem. Soc. Rev. 43, 6011 (2014)
G.L. Fan, F. Li, D.G. Evans, X. Duan, Chem. Soc. Rev. 43, 7040 (2014)
F.L. Li, O. Shao, X.Q. Huang, J.P. Lang, Angew. Chem. Int. Ed. 57, 1888 (2018)
M. Dai, X.R. Su, X. Wang, B. Wu, Z.G. Gen, X. Zhou, J.P. Lang, Cryst. Growth Des. 14, 240 (2014)
D.X. Li, C.Y. Ni, M.M. Chen, M. Dai, W.H. Zhang, W.Y. Yan, H.X. Qi, Z.G. Ren, J.P. Lang, CrystEngComm 16, 2158 (2014)
X.Y. Zhang, X. Zhang, C.B. Fan, Z.A. Zong, D.M. Zhang, Q.D. Luo, C.F. Bi, Y.H. Fan, Polyhedron 168, 21 (2019)
V.A. Blatov, IUCr CompComm Newslett. 7, 4 (2006)
V.A. Blatov, A.P. Shevchenko, V.N. Serezhkin, J. Appl. Crystallogr. 33, 1193 (2000)
V.A. Blatov, M. O’Keeffe, D.M. Proserpio, CrystEngComm 12, 44 (2010)
O.D. Friedrichs, Program SYSTRE 1.14 beta https://gavrog.sourceforge.net/, 2007.
A.L. Spek, PLATON, A Multipurpose Crystallographic Tool (Utrecht University, Utrecht, 2002)
X.M. Meng, X. Zhang, P.F. Qi, Z.A. Zong, F. Jin, Y.H. Fan, RSC Adv. 7, 4855 (2017)
Z.A. Zong, C.B. Fan, X. Zhang, X.M. Meng, F. Jin, Y.H. Fan, Microporous Mesoporous Mat. 282, 82 (2019)
C.-B. Liu, Y. Cong, H.-Y. Sun, G.-B. Che, Inorg. Chem. Commun. 47, 71–74 (2014)
Y. Qiao, Y.-F. Zhou, W.-S. Guan, L.-H. Liu, B. Liu, G.-B. Che, C.-B. Liu, X. Lin, E.-W. Zhu, Inorg. Chim. Acta 66, 291–297 (2017)
Acknowledgements
This work was supported by the Fundamental Research Funds for the Central Universities (Grant No. 201822012), the National Natural Science Foundation of China (Grant Nos. 21476218 and 21601171). Guangxi Natural Science Foundation Program (Grant No. 2019GXNSFBA245086).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, D., Bi, C., Zong, Z. et al. Three Different Co(II) Metal–Organic Frameworks Based on 4,4′-Bis(imidazolyl)diphenyl Ether: Syntheses, Crystal Structure and Photocatalytic Properties. J Inorg Organomet Polym 30, 5148–5156 (2020). https://doi.org/10.1007/s10904-020-01657-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-020-01657-9