Abstract
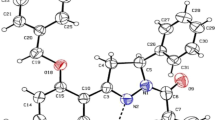
Mapping molecular electrostatic potential (MESP) maps for chemical structure has become of great concern. Since it may explain valuable information about the interaction, active sites as well as determining the nature of chemical addition, through which a molecule is most probable to undergo; either electrophilic or nucleophilic addition. Calculations at a DFT high theoretical level and MESP maps for fulleropyrrolidine and its derivatives were carried out. The relation between total dipole moment (TDM) and the constructed MESPs were also investigated. Three different series of fulleropyrrolidine derivatives; ortho, meta and para, were presented. Results show that positioning the same element at different sites may change significantly TDM of the structure. The electron density of the fullerene structure appears as a red ellipse shape indicating an extremely negative region that is most likely to undergo nucleophilic reactions. The addition of electronegative substitutes; O and N, creates negative potentials at the terminal of the structure referring that electrophilic reactions are most probable there. The negative potential of fullerene core has the main role in determining the TDM direction.




Similar content being viewed by others
References
Abdelsalam, H., Elhaes, H., Ibrahim, M.A.: First principles study of edge carboxylated graphene quantum dots. Physica B 537, 77–86 (2018a)
Abdelsalam, H., Elhaes, H., Ibrahim, M.A.: Tuning electronic properties in graphene quantum dots by chemical functionalization: density functional theory calculations. Chem. Phys. Lett. 695, 138–148 (2018b)
Abdelsalam, H., Saroka, V.A., Ali, M., Teleb, N.H., Elhaes, H., Ibrahim, M.A.: Stability and electronic properties of edge functionalized silicene quantum dots: a first principles study. Physica E 108, 339–346 (2019a)
Abdelsalam, H., Teleb, N.H., Yahia, I.S., Zahran, H.Y., Elhaes, H., Ibrahim, M.A.: First principles study of the adsorption of hydrated heavy metals on graphene quantum dots. J. Phys. Chem. Solids 130, 32–40 (2019b)
Arteca, G.A., Jammal, V.B., Mezey, P.G., Yadav, J.S., Hermsmeier, M.A., Gund, T.M.: Shape group studies of molecular similarity: relative shapes of Van der Waals and electrostatic potential surfaces of nicotinic agonists. J. Mol. Gr. 6(1), 45–53 (1988)
Bader, R.F.W., Carroll, M.T., Cheeseman, J.R., Chang, C.: Properties of atoms in molecules: atomic volumes. J. Am. Chem. Soc. 109(26), 7968–7979 (1987)
Bader, R.F.W., Henneker, W.H., Cade, P.E.: Molecular charge distributions and chemical binding. J. Chem. Phys. 46(9), 3341–3363 (1967)
Becke, A.D.: Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 98, 5648–5652 (1993)
Bondi, A.: Van der Waals volumes and radii. J. Phys. Chem. 68(3), 441–451 (1964)
Bulat, F.A., Toro-Labbé, A., Brinck, T., Murray, J.S., Politzer, P.: Quantitative analysis of molecular surfaces: areas, volumes, electrostatic potentials and average local ionization energies. J. Mol. Model. 16(11), 1679–1691 (2010)
Connolly, M.L.: Solvent-accessible surfaces of proteins and nucleic acids. Science 221(4612), 709–713 (1983)
Diao, Y., Liu, L., Xia, S.: Comparative study on electronic and optical properties for composition-tunable GaAlAs and InGaAs nanowires from first-principles calculation. Physica E 117, 113843 (2020)
Dunitz, J.D., Filippini, G., Gavezzotti, A.: A statistical study of density and packing variations among crystalline isomers. Tetrahedron 56(36), 6595–6601 (2000)
ElHaes, H., Saleh, N.A., Omar, A., Ibrahim, M.: Molecular spectroscopic study of fulleropyrrolidine carbodithioic acid. J. Comput. Theor. Nanosci. 11, 2136–2140 (2014)
Ergürhan, O., Parlak, C., Alver, Ö., Şenyel, M.: Conformational and electronic properties of hydroquinone adsorption on C60 fullerenes: doping atom, solvent and basis set effects. J. Mol. Struct. 1167, 227–231 (2018)
Ezzat, H., Badry, R., Yahia, I.S., Zahran, H.Y., Elhaes, H., Ibrahim, M.A.: Mapping the molecular electrostatic potential of carbon nanotubes. Biointerface Res. Appl. Chem. 8, 3539–3542 (2018)
Ezzat, H., Badry, R., Yahia, I.S., Zahran, H.Y., Ibrahim, A., Elhaes, H., Ibrahim, M.A.: Mapping the molecular electrostatic potential of fullerene. Egypt. J. Chem. 6, 1391–1402 (2019)
Francl, M.M., Hout, R.F., Hehre, W.J.: Representation of electron densities. 1. Sphere fits to total electron density surfaces. J. Am. Chem. Soc. 106(3), 563–570 (1984)
Frisch, M.J., et al.: Gaussian, Inc., Wallingford CT (2010)
Fuks-Janczarek, I., Dabos-Seignan, S., Sahraoui, B., Kityk, I.V., Berdowski, J., Allard, E., Cousseau, J.: Experimental study of third-order nonlinear optical properties in C60-TTF dyads with saturated (–C–C–) chemical bonds. Opt. Commun. 211(1–6), 303–308 (2002)
Hameed, A.J., Ibrahim, M., ElHaes, H.: Computational notes on structural, electronic and QSAR properties of [C60] fulleropyrrolidine-1-carbodithioic acid 2; 3 and 4-substituted-benzyl esters. J. Mol. Struct. Theochem. 809, 131–136 (2007)
Ibrahim, M.A., Elhaes, H., El-Khodary, S.A., Morsy, M., Refaat, A., Yahia, I.S., Zahran, H.Y.: Molecular modeling analyses for the effect of alkali metal oxides on graphene. Biointerface Res. Appl. Chem. 8, 3522–3525 (2018)
Lee, B., Richards, F.M.: The interpretation of protein structures: estimation of static accessibility. J. Mol. Biol. 55(3), 379–400 (1971)
Lee, C., Yang, W., Parr, R.G.: Development of the Colic–Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 37, 785 (1988)
Lu, X., Cui, M., Pan, X., Wang, P., Sun, L.: Investigation of the structural and electronic properties of pristine and Au-embedded MoS2/C60 and WSe2/C60 van der Waals heterostructures: a first-principles study. Appl. Surf. Sci. 503, 144328 (2020)
Rostami, Z., Hosseinian, A., Monfared, A.: DFT results against experimental data for electronic properties of C60 and C70 fullerene derivatives. J. Mol. Gr. Modell. 81, 60–67 (2018)
Saleh, N.A., ElHaes, H., Osman, O., Mahmoud, A.-A., Ibrahim, M.: Spectroscopic analyses of modified fulleropyrrolidine derivatives. Open Spectrosc. J. 9, 1–6 (2015)
Sahraoui, B., Kityk, I.V., Bielieninik, J., Hudhomme, P., Gorgues, A.: Optical limiting processes in C60-2-thioxo-1,3-dithiole cycloadduct. Opt. Mater. 13(3), 349–353 (1999)
Taherpour, A.A., Jamshidi, M., Rezaei, O.: DFT and TD-DFT theoretical studies on photo-induced electron transfer process on [Cefamandole]. C60 nano-complex. J. Mol. Gr. Modell. 75, 42–48 (2017)
Tian, C., Lin, K., Lu, J., Feng, W., Song, S., Xie, L., Wei, Z.: Interfacial Bridge using a cis-fulleropyrrolidine for efficient planar perovskite solar cells with enhanced stability. Small Methods 4(5), 1900476 (2020)
Villagómez, C.J., Garzón, I.L., Paz-Borbón, L.O.: A first-principles DFT dispersion-corrected C60/Au (111) Raman study. Comput. Mater. Sci. 171, 109208 (2020)
Vosko, S.H., Wilk, L., Nusair, M.: Accurate spin-dependent electron liquid correlation energies for local spin density calculations: a critical analysis. J. Phys. 58, 1200–1211 (1980)
Weiner, P.K., Langridge, R., Blaney, J.M., Schaefer, R., Kollman, P.A.: Electrostatic potential molecular surfaces. Proc. Nat. Acad. Sci. 79(12), 3754–3758 (1982)
Zhang, F., Shi, W., Luo, J., Pellet, N., Yi, C., Li, X., Zhao, X., Dennis, T.J.S., Li, X., Wang, S., Xiao, Y., Zakeeruddin, S.M., Bi, D., Grätzel, M.: Isomer-pure bis-PCBM-assisted crystal engineering of perovskite solar cells showing excellent efficiency and stability. Adv. Mater. 29(17), 1606806 (2017)
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bayoumy, A.M., Ibrahim, M. & Omar, A. Mapping molecular electrostatic potential (MESP) for fulleropyrrolidine and its derivatives. Opt Quant Electron 52, 346 (2020). https://doi.org/10.1007/s11082-020-02467-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11082-020-02467-6