Abstract
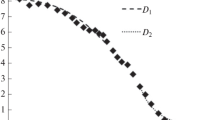
Electron probe microanalysis (EPMA) has been used to obtain concentration curves and calculate the bulk diffusion coefficients of Sn in solid solutions of the copper–tin system in a tin concentration range of less than 13.9% weight (7.96 at %) and temperature range from 500 to 650°C. Diffusion couples are made from pure Cu (99.995%) and a two-component alloy of Cu with chemically pure Sn by direct alloying metallic Cu with Sn in an Ar–H2 atmosphere at 1100°C for 2 h. Diffusion coefficients were calculated using the Matano–Boltzmann and Grube methods from the upper part of the concentration curve (from 6 to 8 at % (D1)) and the lower part (from 2 at % to zero (D2)). It is shown that Sn diffusion coefficients in a concentrated solution were several times greater than Sn diffusion coefficients in dilute solution. Both values of diffusion activation energy, especially the second, coincide with isotope data on Sn diffusion in a pure copper (187 kJ/mol). A qualitative interpretation is proposed of the effect of accelerating the diffusion of tin in a concentrated solid solution of the copper–tin system.





Similar content being viewed by others
REFERENCES
Kostrzhitskii, A.I. and Gusareva, O.F., Patterns of formation of the structure of iron-chromium coatings during ion deposition, Izv. Akad. Nauk SSSR. Met., 1987, no. 1, pp. 169–172.
Wang, J., Leinbach, C., Liu, H.S., Roth, M., and Jin, Z.P., Re-assessment of diffusion mobilities in the face-centered cubic Cu–Sn alloys, CALPHAD, 2009, no. 33, pp. 704–710.
Minho, O., Vakanas, G., Moelans, N., Kajihara, M., and Zhang, W., Formation of compounds and Kirkendall vacancy in the Cu–Sn system, Microelectron. Eng., 2014, vol. 120, pp. 133–137.
Baheti, V.A., Kashuap, S., Kumar, P., Chattopadhya, K., and Paul, A.J., Solid-state diffusion-controlled growth of the intermediate phases from room temperature to an elevated temperature in the Cu–Sn and the Ni–Sn systems, J. Alloys Compd., 2017, vol. 727, pp. 832–840.
Labie, R., Ruythooren, W., and Van Humbeeck, J., Solid state diffusion in Cu–Sn and Ni–Sn diffusion couples with flip-chip scale dimensions, Intermetallics, 2007, no. 15, pp. 396–403.
Chao, B., Chae, S.-H., Zhang, X., Lu, K.-H., Im, J., and Ho, P.S., Investigation of diffusion and electromigration parameters for Cu–Sn intermetallic compounds in Pb-free solders using simulated annealing, Acta Mater., 2007, no. 55, pp. 2805–2814.
Gorbachev, V.A., Klotsman, S.M., Rabovskii, Y.A., Talinskii, V.K., and Timofeev, A.N., Diffusion of impurities in copper. IV. Diffusion of tin and antimony in single crystals of copper, Fiz. Met. Metalloved., 1973, no. 35, pp. 889–893.
Fogelson, R.L., Ugay, Y.A., and Akimova, I.A., Diffusion of tin in copper, Phys. Met. Metallogr., 1974, no. 37, pp. 1107–1108.
Oikawa, H. and Hosoi, A., Interdiffusion in Cu–Sn solid solutions confirmation of anomalously large Kirkendall effect, Scr. Metall., 1975, no. 9, pp. 823–828.
Hoshino, K., Iijima, A.Y., and Hirano, K.I., Inter-diffusion and Kirkendall effect in Cu–Sn alloys, Trans. Jpn. Inst. Met., 1980, vol. 21, no. 10, pp. 674–682.
Santra, S. and Paul, A., Vacancy wind effect on interdiffusion in a dilute Cu (Sn) solid solution, Philos. Mag. Lett., 2012, vol. 92, no. 8, pp. 1–11.
Onishi, M. and Fujibuchi, M., Reaction-diffusion in the Cu–Sn system, Trans. Jpn. Inst. Met., 1975, no. 16, pp. 539–547.
Paul, A., Ghoshi, C., and Boetinger, W.J., Diffusion parameters and growth mechanism of phases in the Cu‒Sn system, Metall. Mater. Trans. A, 2011, vol. 42, no. 4, pp. 952–963.
Kumar, S., Handwerker, C., Dayamanda, M., and Phase, J., Intrinsic and interdiffusion in Cu–Sn system, J. Phase Equilib. Diffus., 2011, vol. 32, no. 4, pp. 309–319.
Massalski, B., Binary Alloy Phase Diagrams, Metals Park, OH: American Society for Metals, 1986.
Predel, B. and Schallner, U., Thermodynamic investigation of the Cu–Ga, Cu–In, Cu–Ge, and Cu–Sn systems, Mater. Sci. Eng., 1972, no. 10, pp. 249–258.
Wallbrecht, R.C., Blachnik, R., and Mills, R.C., The heat capacity and enthalpy of some hume-rothery phases formed by copper, silver and gold. Part II. Cu + Ge, Cu + Sn, Ag + Sn, Au + Sn, Au + Pb systems, Thermochim. Acta, 1981, no. 46, pp. 167–174.
Matano, C., On the relation between the diffusion coefficient and concentration of solid metals, Jpn. J. Phys., 1933, no. 8, pp. 109–113.
den Broeder, E.J.A., A general simplification and improvement of the Matano–Boltzmann method in the determination of the interdiffusion coefficients in binary systems, Scr. Metall., 1969, vol. 3, no. 5, pp. 321–325.
Darken, L., Diffusion, mobility and their interrelation through free energy in binary metallic systems, Trans. AIME, 1948, no. 175, pp. 184–201.
Adda, Y. and Philibert, J., La Diffusion dans les solides, Paris: Press Universitaires de France, 1966.
Grube, G. and Jedele, A., Die Diffusion der Metalle im esten Zustand, Z. Elektrochem., 1932, no. 38, pp. 799–807.
Crank, J., Mathematics of Diffusion, Oxford: Clarendon Press, 1956.
Bardeen, J. and Herring, C., Atom Movements, Cleveland, OH: A.S.M., 1951, pp. 87–288.
Bardeen, J. and Herring, C., Imperfections in Nearly Perfect Crystals, New York: John Wiley and Sons, 1952.
Bokstein, B.S., Mendelev, M.I., and Srolovitz, D.J., Thermodynamics and Kinetics in Materials Science, Oxford: Oxford Univ. Press, 2005.
ACKNOWLEDGMENTS
We thank V. Yesin (Center des materiaux, CNRS UMR 7633, Paris, France) for help in conducting the experiments.
Funding
This study was financed by the Russian Science Foundation (project no. 16-12-10478).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare that they have no conflict of interest.
About this article
Cite this article
Nikulkina, V.P., Rodin, A.O. & Bokshtein, B.S. Diffusion of Tin in a Solid Solution of the Copper–Tin System. Russ. J. Non-ferrous Metals 61, 291–296 (2020). https://doi.org/10.3103/S1067821220030116
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3103/S1067821220030116