Abstract
Purpose
To explore the alterations of spontaneous neuronal activity and functional connectivity pattern using fractional amplitude of low-frequency fluctuation (fALFF) and functional connectivity (FC) in patients with primary angle-closure glaucoma (PACG) and fALFF relationship with the glaucoma clinical indices.
Materials and methods
Forty-two PACG patients and 21 normal controls were enrolled in this study. Resting-state functional magnetic resonance imaging was firstly analyzed by fALFF and brain regions with altered fALFF between groups were selected as seeds for the further FC analysis. The relationships between fALFF/FC values of abnormal regions and ophthalmological measures, including mean deviation of visual field (MDVF) and retinal nerve fiber layer (RNFL) thickness, were also analyzed.
Results
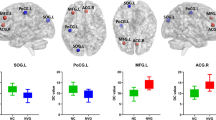
Compared with NC, PACG had significant lower fALFF values in the left cuneus, left middle temporal gyrus, right middle temporal gyrus, and right precentral gyrus, while higher fALFF values in the bilateral superior frontal gyrus (P < 0.05 after correction). Furthermore, PACG showed increased FC between left cuneus and bilateral superior frontal gyrus/bilateral posterior cingulate gyrus; between left middle temporal gyrus and bilateral superior frontal gyrus; and between right middle temporal gyrus and bilateral insular (P < 0.05 after correction). In addition, in the PACG group, the mean fALFF values of the left cuneus were positively correlated with MDVF (R = 0.419, P = 0.005) and RNFL thickness (R = 0.322, P = 0.038). Meanwhile, the mean fALFF values of bilateral superior frontal gyrus were negatively correlated with MDVF (R = − 0.454, P = 0.003) and RNFL thickness (R = − 0.556, P < 0.001).
Conclusions
PACG exhibited abnormal spontaneous neural activity and connectivity in several brain regions mainly associated with visual and visual-related functions. In addition, the fALFF values of the left cuneus and bilateral superior frontal gyrus may be complementary biomarkers for assessing the disease severity.




Similar content being viewed by others
References
Quigley HA, Broman AT (2006) The number of people with glaucoma worldwide in 2010 and 2020. Br J Ophthalmol 90(3):262–267. https://doi.org/10.1136/bjo.2005.081224
Chen S, Lv J, Fan S, Zhang H, Xie L, Xu L, Jiang B, Yuan H, Liang Y, Li S, Chen P, Zhang X, Wang N (2017) Laser peripheral iridotomy versus laser peripheral iridotomy plus laser peripheral iridoplasty in the treatment of multi-mechanism angle closure: study protocol for a randomized controlled trial. Trials 18(1):130. https://doi.org/10.1186/s13063-017-1860-4
Lei Y, Garrahan N, Hermann B, Fautsch MP, Johnson DH, Hernandez MR, Boulton M, Morgan JE (2009) Topography of neuron loss in the retinal ganglion cell layer in human glaucoma. Br J Ophthalmol 93(12):1676–1679. https://doi.org/10.1136/bjo.2009.159210
Yucel Y, Gupta N (2008) Glaucoma of the brain: a disease model for the study of transsynaptic neural degeneration. Prog Brain Res 173:465–478. https://doi.org/10.1016/s0079-6123(08)01132-1
Gupta N, Yucel YH (2006) Glaucoma in the brain: a piece of the puzzle. Can J Ophthalmo 41(5):541–542. https://doi.org/10.1016/s0008-4182(06)80022-0
Nuzzi R, Dallorto L, Rolle T (2018) Changes of visual pathway and brain connectivity in glaucoma: a systematic review. Front Neurosci 12:363–363. https://doi.org/10.3389/fnins.2018.00363
Biswal B, Yetkin FZ, Haughton VM, Hyde JS (1995) Functional connectivity in the motor cortex of resting human brain using echo-planar MRI. Magn Reson Med 34(4):537–541. https://doi.org/10.1002/mrm.1910340409
Chen W, Zhang L, Xu Y-G, Zhu K, Luo M (2017) Primary angle-closure glaucomas disturb regional spontaneous brain activity in the visual pathway: an fMRI study. Neuropsychiatr Dis Treat 13:1409–1417. https://doi.org/10.2147/NDT.S134258
Huang X, Zhong YL, Zeng XJ, Zhou F, Liu XH, Hu PH, Pei CG, Shao Y, Dai XJ (2015) Disturbed spontaneous brain activity pattern in patients with primary angle-closure glaucoma using amplitude of low-frequency fluctuation: a fMRI study. Neuropsychiatr Dis Treat 11:1877–1883. https://doi.org/10.2147/ndt.S87596
Zang Y, Jiang T, Lu Y, He Y, Tian L (2004) Regional homogeneity approach to fMRI data analysis. NeuroImage 22(1):394–400. https://doi.org/10.1016/j.neuroimage.2003.12.030
Zang YF, He Y, Zhu CZ, Cao QJ, Sui MQ, Liang M, Tian LX, Jiang TZ, Wang YF (2007) Altered baseline brain activity in children with ADHD revealed by resting-state functional MRI. Brain Dev 29(2):83–91. https://doi.org/10.1016/j.braindev.2006.07.002
Zou QH, Zhu CZ, Yang Y, Zuo XN, Long XY, Cao QJ, Wang YF, Zang YF (2008) An improved approach to detection of amplitude of low-frequency fluctuation (ALFF) for resting-state fMRI: fractional ALFF. J Neurosci Methods 172(1):137–141. https://doi.org/10.1016/j.jneumeth.2008.04.012
Ramaekers JG, Evers EA, Theunissen EL, Kuypers KP, Goulas A, Stiers P (2013) Methylphenidate reduces functional connectivity of nucleus accumbens in brain reward circuit. Psychopharmacology 229(2):219–226. https://doi.org/10.1007/s00213-013-3105-x
Li S, Li P, Gong H, Jiang F, Liu D, Cai F, Pei C, Zhou F, Zeng X (2017) Intrinsic functional connectivity alterations of the primary visual cortex in primary angle-closure glaucoma patients before and after surgery: a resting-state fMRI study. PLoS One 12(1):e0170598. https://doi.org/10.1371/journal.pone.0170598
Dai H, Morelli JN, Ai F, Yin D, Hu C, Xu D, Li Y (2013) Resting-state functional MRI: functional connectivity analysis of the visual cortex in primary open-angle glaucoma patients. Hum Brain Mapp 34(10):2455–2463. https://doi.org/10.1002/hbm.22079
Yan CG, Wang XD, Zuo XN, Zang YF (2016) DPABI: data processing & analysis for (resting-state) brain imaging. Neuroinformatics 14(3):339–351. https://doi.org/10.1007/s12021-016-9299-4
Power JD, Mitra A, Laumann TO, Snyder AZ, Schlaggar BL, Petersen SE (2014) Methods to detect, characterize, and remove motion artifact in resting state fMRI. NeuroImage 84:320–341. https://doi.org/10.1016/j.neuroimage.2013.08.048
Yan CG, Wang XD, Zuo XN, Zang YFJN (2016) DPABI: data processing & analysis for (resting-state) brain imaging. Neuroinformatics 14(3):339–351. https://doi.org/10.1007/s12021-016-9299-4
Tillikainen L, Salli E, Korvenoja A, Aronen HJ (2006) A cluster mass permutation test with contextual enhancement for fMRI activation detection. NeuroImage 32(2):654–664. https://doi.org/10.1016/j.neuroimage.2006.03.058
Keitel C, Maess B, Schroger E, Muller MM (2013) Early visual and auditory processing rely on modality-specific attentional resources. NeuroImage 70:240–249. https://doi.org/10.1016/j.neuroimage.2012.12.046
Toth LJ, Rao SC, Kim DS, Somers D, Sur M (1996) Subthreshold facilitation and suppression in primary visual cortex revealed by intrinsic signal imaging. Proc Natl Acad Sci U S A 93(18):9869–9874. https://doi.org/10.1073/pnas.93.18.9869
Li T, Liu Z, Li J, Liu Z, Tang Z, Xie X, Yang D, Wang N, Tian J, Xian J (2014) Altered amplitude of low-frequency fluctuation in primary open-angle glaucoma: a resting-state FMRI study. Invest Ophthalmol Vis Sci 56(1):322–329. https://doi.org/10.1167/iovs.14-14974
Song Y, Mu K, Wang J, Lin F, Chen Z, Yan X, Hao Y, Zhu W, Zhang H (2014) Altered spontaneous brain activity in primary open angle glaucoma: a resting-state functional magnetic resonance imaging study. PLoS One 9(2):e89493. https://doi.org/10.1371/journal.pone.0089493
Cai F, Gao L, Gong H, Jiang F, Pei C, Zhang X, Zeng X, Huang R (2015) Network centrality of resting-state fMRI in primary angle-closure glaucoma before and after surgery. PLoS One 10(10):e0141389. https://doi.org/10.1371/journal.pone.0141389
Lalezary M, Medeiros FA, Weinreb RN, Bowd C, Sample PA, Tavares IM, Tafreshi A, Zangwill LM (2006) Baseline optical coherence tomography predicts the development of glaucomatous change in glaucoma suspects. Am J Ophthalmol 142(4):576–582. https://doi.org/10.1016/j.ajo.2006.05.004
Vartanian O, Goel V, Lam E, Fisher M, Granic JJPoAC, Arts t (2013) Middle temporal gyrus encodes individual differences in perceived facial attractiveness. Psychology of Aesthetics Creativity & the Arts 7(1):38–47
Iaria G, Fox CJ, Waite CT, Aharon I, Barton JJ (2008) The contribution of the fusiform gyrus and superior temporal sulcus in processing facial attractiveness: neuropsychological and neuroimaging evidence. Neuroscience 155(2):409–422. https://doi.org/10.1016/j.neuroscience.2008.05.046
Porro CA, Francescato MP, Cettolo V, Diamond ME, Baraldi P, Zuiani C, Bazzocchi M, di Prampero PE (1996) Primary motor and sensory cortex activation during motor performance and motor imagery: a functional magnetic resonance imaging study. J Neurosci 16(23):7688–7698
Ugur HC, Kahilogullari G, Coscarella E, Unlu A, Tekdemir I, Morcos JJ, Elhan A, Baskaya MK (2005) Arterial vascularization of primary motor cortex (precentral gyrus). Surg Neurol 64(Suppl 2):S48–S52. https://doi.org/10.1016/j.surneu.2005.07.049
Wu J, Coffey M, Reidy A, Wormald R (1998) Impaired motion sensitivity as a predictor of subsequent field loss in glaucoma suspects: the Roscommon Glaucoma Study. Br J Ophthalmol 82(5):534–537
Choi W, Desai RH, Henderson JM (2014) The neural substrates of natural reading: a comparison of normal and nonword text using eyetracking and fMRI. Front Hum Neurosci 8:1024. https://doi.org/10.3389/fnhum.2014.01024
Bodis-Wollner I, Bucher SF, Seelos KC, Paulus W, Reiser M, Oertel WH (1997) Functional MRI mapping of occipital and frontal cortical activity during voluntary and imagined saccades. Neurology 49(2):416–420. https://doi.org/10.1212/wnl.49.2.416
Martino J, Gabarros A, Deus J, Juncadella M, Acebes JJ, Torres A, Pujol J (2011) Intrasurgical mapping of complex motor function in the superior frontal gyrus. Neuroscience 179:131–142. https://doi.org/10.1016/j.neuroscience.2011.01.047
Picado M, Carmona S, Hoekzema E, Pailhez G, Berge D, Mane A, Fauquet J, Hilferty J, Moreno A, Cortizo R, Vilarroya O, Bulbena A (2015) The neuroanatomical basis of panic disorder and social phobia in schizophrenia: a voxel based morphometric study. PLoS One 10(3):e0119847. https://doi.org/10.1371/journal.pone.0119847
du Boisgueheneuc F, Levy R, Volle E, Seassau M, Duffau H, Kinkingnehun S, Samson Y, Zhang S, Dubois B (2006) Functions of the left superior frontal gyrus in humans: a lesion study. Brain 129(Pt 12):3315–3328. https://doi.org/10.1093/brain/awl244
Smith ND, Glen FC, Crabb DP (2012) Eye movements during visual search in patients with glaucoma. BMC Ophthalmol 12:45. https://doi.org/10.1186/1471-2415-12-45
Olson IR, Rao H, Moore KS, Wang J, Detre JA, Aguirre GK (2006) Using perfusion fMRI to measure continuous changes in neural activity with learning. Brain Cogn 60(3):262–271. https://doi.org/10.1016/j.bandc.2005.11.010
Wu Y-Y, Wang S-F, Zhu P-W, Yuan Q, Shi W-Q, Lin Q, Li B, Min Y-L, Zhou Q, Shao Y (2020) Altered intrinsic functional connectivity of the primary visual cortex in patients with neovascular glaucoma: a resting-state functional magnetic resonance imaging study. Neuropsychiatr Dis Treat 16:25–33. https://doi.org/10.2147/NDT.S228606
Wang J, Li T, Zhou P, Wang N, Xian J, He H (2017) Altered functional connectivity within and between the default model network and the visual network in primary open-angle glaucoma: a resting-state fMRI study. Brain Imaging Behav 11(4):1154–1163. https://doi.org/10.1007/s11682-016-9597-3
Ye L, Wei R, Huang X, Shi W-Q, Yang Q-C, Yuan Q, Zhu P-W, Jiang N, Li B, Zhou Q, Zhou F-Q, Shao Y (2018) Reduction in interhemispheric functional connectivity in the dorsal visual pathway in unilateral acute open globe injury patients: a resting-state fMRI study. Int J Ophthalmol 11(6):1056–1060. https://doi.org/10.18240/ijo.2018.06.26
Shao Y, Bao J, Huang X, Zhou F-Q, Ye L, Min Y-L, Yang L, Sethi Z, Yuan Q, Zhou Q (2018) Comparative study of interhemispheric functional connectivity in left eye monocular blindness versus right eye monocular blindness: a resting-state functional MRI study. Oncotarget 9(18):14285–14295. https://doi.org/10.18632/oncotarget.24487
Leech R, Sharp DJ (2014) The role of the posterior cingulate cortex in cognition and disease. Brain 137(Pt 1):12–32. https://doi.org/10.1093/brain/awt162
Mattingley JB, Husain M, Rorden C, Kennard C, Driver J (1998) Motor role of human inferior parietal lobe revealed in unilateral neglect patients. Nature 392(6672):179–182. https://doi.org/10.1038/32413
Desmurget M, Sirigu A (2012) Conscious motor intention emerges in the inferior parietal lobule. Curr Opin Neurobiol 22(6):1004–1011. https://doi.org/10.1016/j.conb.2012.06.006
Raichle ME (2015) The brain’s default mode network. Annu Rev Neurosci 38:433–447. https://doi.org/10.1146/annurev-neuro-071013-014030
Goulden N, Khusnulina A, Davis NJ, Bracewell RM, Bokde AL, McNulty JP, Mullins PG (2014) The salience network is responsible for switching between the default mode network and the central executive network: replication from DCM. NeuroImage 99:180–190. https://doi.org/10.1016/j.neuroimage.2014.05.052
Frezzotti P, Giorgio A, Toto F, De Leucio A, De Stefano N (2016) Early changes of brain connectivity in primary open angle glaucoma. Hum Brain Mapp 37(12):4581–4596. https://doi.org/10.1002/hbm.23330
Giorgio A, Zhang J, Costantino F, De Stefano N, Frezzotti P (2020) Altered large-scale brain functional connectivity in ocular hypertension. Front Neurosci 14:146. https://doi.org/10.3389/fnins.2020.00146
Yoo JH, Kim D, Choi J, Jeong B (2018) Treatment effect of methylphenidate on intrinsic functional brain network in medication-naïve ADHD children: a multivariate analysis. Brain Imaging Behav 12(2):518–531. https://doi.org/10.1007/s11682-017-9713-z
Sporns O (2011) The human connectome: a complex network. Ann N Y Acad Sci 1224:109–125. https://doi.org/10.1111/j.1749-6632.2010.05888.x
van den Heuvel MP, Kahn RS, Goñi J, Sporns O (2012) High-cost, high-capacity backbone for global brain communication. Proc Natl Acad Sci U S A 109(28):11372–11377. https://doi.org/10.1073/pnas.1203593109
Funding
This work was financially supported by the National Natural Science Foundation of China (Grant No. 81871341), the State Key Program of National Natural Science Foundation of China (Grant No. 81430007), and the Grant of Science and Technology Commission of Shanghai Municipality (No. 14411962000).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
The protocols of this study were approved by the Local Ethics Committee on Human Research of Eye & ENT Hospital of Fudan University.
Informed consent
Written informed consent was collected from each participant prior to his or her participation in this study.
Disclaimer
The sponsor or funding organization had no role in the design or conduct of this research.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, R., Tang, Z., Liu, T. et al. Altered spontaneous neuronal activity and functional connectivity pattern in primary angle-closure glaucoma: a resting-state fMRI study. Neurol Sci 42, 243–251 (2021). https://doi.org/10.1007/s10072-020-04577-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-020-04577-1