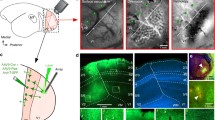
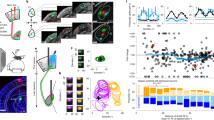
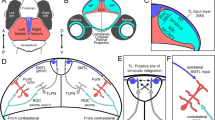
Abstract
In spite of their anatomical robustness, it has been difficult to establish the functional role of corticogeniculate circuits connecting primary visual cortex with the lateral geniculate nucleus of the thalamus (LGN) in the feedback direction. Growing evidence suggests that corticogeniculate feedback does not directly shape the spatial receptive field properties of LGN neurons, but rather regulates the timing and precision of LGN responses and the information coding capacity of LGN neurons. We propose that corticogeniculate feedback specifically stabilizes the response gain of LGN neurons, thereby increasing their information coding capacity. Inspired by early work by McClurkin et al. (1994), we manipulated the activity of corticogeniculate neurons to test this hypothesis. We used optogenetic methods to selectively and reversibly enhance the activity of corticogeniculate neurons in anesthetized ferrets while recording responses of LGN neurons to drifting gratings and white noise stimuli. We found that optogenetic activation of corticogeniculate feedback systematically reduced LGN gain variability and increased information coding capacity among LGN neurons. Optogenetic activation of corticogeniculate neurons generated similar increases in information encoded in LGN responses to drifting gratings and white noise stimuli. Together, these findings suggest that the influence of corticogeniculate feedback on LGN response precision and information coding capacity could be mediated through reductions in gain variability.




Similar content being viewed by others
References
Acker, L., Pino, E. N., & Desimone, R. (2016). FEF inactivation with improved optogenetic methods. Procedings of the National Academy of Science USA, 113, E7297–E7306.
Andolina, I. M., Jones, H. E., Wang, W., & Sillito, A. M. (2007). Corticothalamic feedback enhances stimulus response precision in the visual system. PNAS, 104(5), 1685–1690.
Carrasco, M., Ling, S., & Read, S. (2004). Attention alters appearance. Nature Neuroscience, 7(3), 308–313.
Cohen, M. R., & Maunsell, J. H. (2009). Attention improves performance primarily by reducing interneuronal correlations. Nature Neuroscience, 12(12), 1594–1600.
Denman, D. J., & Contreras, D. (2015). Complex effects on in vivo visual responses by specific projections from mouse cortical layer 6 to dorsal lateral geniculate nucleus. Journal of Neuroscience, 35(25), 9265–9280.
Derrington, A. M., & Fuchs, A. F. (1979). Spatial and temporal properties of X and Y cells in the cat lateral geniculate nucleus. Journal of Physiology, 293, 347–364.
Erisir, A., Van Horn, S. C., Bickford, M. E., & Sherman, S. M. (1997a). Immunocytochemistry and distribution of parabrachial terminals in the lateral geniculate nucleus of the cat: A comparison with corticogeniculate terminals. The Journal of Comparative Neurology, 377, 535–549.
Erisir, A., Van Horn, S. C., & Sherman, S. M. (1997b). Relative numbers of cortical and brainstem inputs to the lateral geniculate nucleus. PNAS, 94, 1517–1520.
Funke, K., Nelle, E., Li, B., & Worgotter, F. (1996). Corticofugal feedback improves the timing of retino-geniculate signal transmission. NeuroReports, 7(13), 2130–2134.
Geisert, E. E., Langsetmo, A., & Spear, P. D. (1981). Influence of the cortico-geniculate pathway on response properties of cat lateral geniulate nucleus. Brain Research, 208, 409–415.
Goris, R. L. T., Movshon, J. A., & Simoncelli, E. P. (2014). Partitioning neuronal variability. Nature Neuroscience, 17(6), 858–865.
Goris, R. L. T., Ziemba, C. M., Movshon, J. A., & Simoncelli, E. P. (2018). Slow gain fluctuations limit benefits of temporal integration in visual cortex. Journal of Vision, 18(8), 1–13.
Gu, Y., Fetsch, C. R., Adeyemo, B., DeAngelis, G. C., & Angelaki, D. E. (2010). Decoding of MSTd population activity accounts for variations in the precision of heading perception. Neuron, 66, 596–609.
Gulyas, B., Lagae, L., Eysel, U. T., & Orban, G. A. (1990). Corticofugal feedback influences the responses of geniculate neurons to moving stimuli. Experimental Brain Research, 79(2), 441–446.
Hartveit, E., & Heggelund, P. (1994). Response variability of single cells in the dorsal lateral geniculate nucleus of the cat. Comparison with retinal input and effect of brain stem stimulation. Journal of Neurophysiology, 72, 1278–1289.
Hasse, J. M., Bragg, E. M., Murphy, A. J., & Briggs, F. (2019). Morphological heterogeneity among corticogeniculate neurons in ferrets: Quantification and comparison with a previous report in macaque monkeys. Journal of Comparative Neurology, 527, 546–557.
Hasse, J. M., & Briggs, F. (2017). Corticogeniculate feedback sharpens the temporal precision and spatial resolution of visual signals in the ferret. Procedings of the National Academy of Science USA, 114(30), E6222–E6230.
Henaff, O. J., Boundy-Singer, Z. M., Meding, K., Ziemba, C. M., & Goris, R. L. T. (2020). Representation of visual uncertainty through neural gain variability. Nature Communications, 11(2513), 1–12.
Kara, P., Reinagel, P., & Reid, R. C. (2000). Low response variability in simultaneously recorded retinal, thalamic, and cortical neurons. Neuron, 27, 635–646.
Kumbhani, R. D., Nolt, M. J., & Palmer, L. A. (2007). Precision, reliability, and information-theoretic analysis of visual thalamocortical neurons. Journal of Neurophysiology, 98, 2647–2663.
Levine, M. W., Cleland, B. G., Mukherjee, P., & Kaplan, E. (1996). Tailoring of variability in the lateral geniculate nucleus of the cat. Biological Cybernetics, 75, 219–227.
Li, G., Ye, X., Song, T., Yang, Y., & Zhou, Y. (2011). Contrast adaptation in cat lateral geniculate nucleus and influence of corticothalamic feedback. European Journal of Neuroscience, 34, 622–631.
Ling, S., Pratte, M., & Tong, F. (2015). Attention alters orientation processing in the human lateral geniculate nucleus. Nature Neuroscience, 18(4), 496–498.
Liu, R. C., Tzonev, S., Rebrik, S., & Miller, K. D. (2001). Variability and information in a neural code of the cat lateral geniculate nucleus. Journal of Neurophysiology, 86, 2789–2806.
Marrocco, R. T., McClurkin, J. W., & Alkire, M. T. (1996). The influence of the visual cortex on the spatiotemporal response properties of lateral geniculate nucleus cells. Brain Research, 737(1–2), 110–118.
Maunsell, J. H. R., & Cook, E. P. (2002). The role of attention in visual processing. Phil. Trans. Royal Society London, 357, 1063–1072.
McAlonan, K., Cavanaugh, J. R., & Wurtz, R. H. (2008). Guarding the gateway to cortex with attention in visual thalamus. Nature, 456, 391–394.
McClurkin, J. W., Gawne, T. J., Optican, L. M., & Richmond, B. J. (1991). Lateral geniculate neurons in behaving primates. II. Encoding of visual information in the temporal shape of the response. J. Neurophysiology, 66(3), 794–808.
McClurkin, J. W., Optican, L. M., & Richmond, B. J. (1994). Cortical feedback increases visual information transmitted by monkey parvlcellular lateral geniculate nucleus neurons. Visual Neuroscience, 11(3), 601–617.
Mitchell, J. F., Sundberg, K. A., & Reynolds, J. H. (2009). Spatial attention decorrelates intrinsic activity fluctuations in macaque area V4. Neuron, 63, 879–888.
Mock, V. L., Luke, K. L., Hembrook-Short, J. R., & Briggs, F. (2018). Dynamic communication of attention signals between the LGN and V1. Journal of Neurophysiology, 120(4), 1625–1639.
Nover, H., Anderson, C. H., & DeAngelis, G. C. (2005). A logarithmic, scale-invariant representation of speed in macaque middle temporal area accounts for speed discrimination performance. The Journal of Neuroscience, 25(43), 10049–10060.
O'Connor, D. H., Fukui, M. M., Pinsk, M. A., & Kastner, S. (2002). Attention modulates responses in the human lateral geniculate nucleus. Nature Neuroscience, 5(11), 1203–1209.
Pouget, A., Zhang, K., Deneve, S., & Latham, P. E. (1998). Sstatistically efficient estimation using population coding. Neural Computation, 10, 373–401.
Pryluk, R., Kfir, Y., Gelbard-Sagiv, H., Fried, I., & Paz, R. (2019). A tradeoff in the neural code across regions and species. Cell, 176, 597–609.
Przybyszewski, A. W., Gaska, J. P., Foote, W., & Pollen, D. A. (2000). Striate cortex increases contrast gain of macaque LGN neurons. Visual Neuroscience, 17, 485–494.
Rabinowitz, N. C., Goris, R. L. T., Cohen, M. R., & Simoncelli, E. P. (2015). Attention stabilizes the shared gain of V4 populations. eLife, 4, 1–24, doi:https://doi.org/10.7554/eLife.08998.
Rathbun, D. L., Warland, D. K., & Usrey, W. M. (2010). Spike timing and information transmission at retinogeniculate synapses. Journal of Neuroscience, 30(41), 13558–13566.
Reinagel, P., & Reid, R. C. (2000). Temporal coding of visual information in the thalamus. The Journal of Neuroscience, 20(14), 5392–5400.
Seung, H. S., & Sompolinsky, H. (1993). Simple models for reading neuronal population codes. Procedings of the National Academy of Science USA, 90, 10749–10753.
Sherman, S. M., & Guillery, R. W. (1998). On the actions that one nerve cell can have on another: Distinguishing "drivers" from "modulators". PNAS, 95, 7121–7126.
Sherman, S. M., & Guillery, R. W. (2006). Exploring the thalamus and its role in cortical function (2nd ed.). Boston: MIT Press.
Sherman, S. M., & Spear, P. D. (1982). Organization of visual pathways in normal and visually deprived cats. Physiological Reviews, 62(2), 738–850.
Steriade, M. (2003). The corticothalamic system in sleep. Frontiers in Bioscience, 8, d878–d899.
Strong, S. P., Koberle, R., de Ruyter van Steveninck, R. R., & Bialek, W. (1998). Entropy and information in neural spike trains. Physical Review Letters, 80(1), 197–200.
Tsumoto, T., Creutzfeldt, O. D., & Legendy, C. R. (1978). Functional organization of the corticofugal system from visual cortex to lateral geniculate nucleus in the cat. Experimental Brain Research, 32, 345–364.
Usrey, W. M., Reppas, J. B., & Reid, R. C. (1999). Specificity and strength of retiongeniculate connections. Journal of Neurophysiology, 82, 3527–3540.
Vanduffel, W., Tootell, R. B. H., & Orban, G. (2000). Attention-dependent suppression of metabolic activity in the early stages of the macaque visual system. Cerebral Cortex, 10, 109–126.
Acknowledgements
We thank Brianna Carr, Marc Mancarella, and Elise Bragg for expert technical assistance and Drs. Dana LeMoine, Wendy Bates, Diane Moorman-White, Jeff Wyatt, Karen Moodie, and Kirk Maurer for veterinary assistance. We thank Dr. Daniel Rathbun, Uday Chockanathan, and Dr. Krishnan Padmanabhan for helpful discussions of data analysis methods and comments on this manuscript.
Funding
This work was funded by the National Institutes of Health (National Eye Institute: EY018683 and EY025219 to F.B. and T32EY007125 to A.J.M.) and the Whitehall Foundation (2013-05-06). J.M.H. was supported by a Graduate Fellowship from the Albert J. Ryan Foundation.
Author information
Authors and Affiliations
Contributions
A.J.M, R.L.T.G., and F.B. designed the experiments. A.J.M. and J.M.H. collected the data. A.J.M., L.S., and F.B. analyzed the data. A.J.M., R.L.T.G., and F.B. wrote the manuscript.
Corresponding author
Ethics declarations
Conflicts of interest/competing interests
All of the authors declare no financial conflicts of interest or competing interests related to this study.
Availability of data and material
Data generated for this study will be made available upon reasonable written request to the corresponding author.
Code availability
Variance code is available here: https://gorislab.github.io/resources/ Information theory analysis code is available here: https://github.com/BriggsNeuro/InfoVarianceCode.
Additional information
Action Editor: Aasef G. Shaikh
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article belongs to the Topical Collection: Vision and Action
Guest Editors: Aasef Shaikh and Jeffrey Shall
This article belongs to the Topical Collection: Vision and Action Guest Editors: Aasef Shaikh and Jeffrey Shall.
Rights and permissions
About this article
Cite this article
Murphy, A.J., Shaw, L., Hasse, J.M. et al. Optogenetic activation of corticogeniculate feedback stabilizes response gain and increases information coding in LGN neurons. J Comput Neurosci 49, 259–271 (2021). https://doi.org/10.1007/s10827-020-00754-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10827-020-00754-5