Abstract
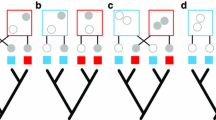
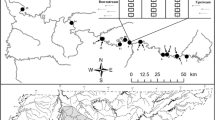
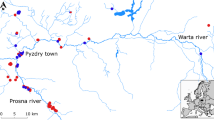
Understanding “why species are where they are” at different scales is one of the main focuses of ecological and biogeographical studies. Although ecological features, such as trophic group and species abundance, are thought to be more important for driving co-occurrence patterns at the habitat scale, it is not yet known if phylogenetic constraints can also exert some influence. Here, we studied global co-occurrence patterns of butterflyfishes in relation to species abundance, trophic characteristics, and evolutionary histories, specifically examining two questions—Question (1): does phylogenetic affinity and/or abundance explain co-occurrence at the habitat scale? To answer this, we used abundance data from 23 global localities to evaluate whether phylogenetic affinity alone as well as after accounting for differences in diet among sympatric species explains co-occurrences at the habitat scale. Question (2): are the diets of sister species from the Atlantic Ocean and the Eastern Pacific phylogenetically conserved? To examine this, we used a more detailed diet classification of species present within these realms. We found that phylogenetic distance per se fails to explain the co-occurrence of butterflyfish species pairs. Instead, species abundance exerted a major influence on interspecific co-occurrences. We also found no correlation between phylogenetic distance and diet similarities for Atlantic and East Pacific butterflyfishes; thus, in these regions, species' diets do not seem to be phylogenetically conserved. This suggests that evolutionary processes are not the main drivers of butterflyfish co-occurrence highlighting species' abundance and niche-related processes as the most important factors in determining whether species co-occur at the habitat scale.




Similar content being viewed by others
Data accessibility
All the data and scripts will be available at the Zenodo platform (https://zenodo.org/record/3895195#.XufO-NrQjIV).
References
Abrams P (1983) The theory of limiting similarity. Annu Rev Ecol Syst 14:359–376
Anderson GRV, Ehrlich AH, Ehrlich PR, Roughgarden JD, Russelland BC, Talbot FH (1981) The community structure of coral reef fishes. Am Nat 117:476–494
Auster PL, Semmens BX, Barber K (2005) Pattern in the co-occurrence of fishes inhabiting the coral reefs of Bonaire, Netherlands Antilles. Environ Biol Fishes 74:187–194. https://doi.org/10.1007/s10641-005-8078-0
Barneche DR, Rezende EL, Parravicini V, Maire E, Edgar GJ, Stuart-Smith RD, Arias-González JE, Ferreira CEL, Friedlander AM, Green AL, Luiz OJ, Rodríguez-Zaragoza FA, Vigliola L, Kulbicki M, Floeter SR (2019) Body size, reef area, and temperature predict global reef-fish species richness across spatial scales. Glob Ecol Biogeogr 28:315–327. https://doi.org/10.1111/geb.12851
Bellwood DR, Pratchett MS (2013) The origins and diversification of coral reef butterflyfishes. In: Pratchett MS, Berumen ML, Kapoor BG (eds) Biology of butterflyfishes. CRC Press, Cambridge, pp 1–18
Bellwood DR, Klanten S, Cowman PF, Pratchett MS, Konow N, Herwerden LV (2010) Evolutionary history of the butterflyfishes (f: Chaetodontidae) and the rise of coral feeding fishes. J Evol Biol 23:335–349. https://doi.org/10.1111/j.1420-9101.2009.01904.x
Berumen ML, Pratchett MS (2008) Trade-offs associated with dietary specialization in corallivorous butterflyfishes (Chaetodontidae: Chaetodon). Behav Ecol Sociobiol 62:989–994. https://doi.org/10.1007/s00265-007-0526-8
Birkeland C, Neudecker S (1981) Foraging behaviour of the two Caribbean Chaetodontids: Chaetodon capistratus and C. aculeatus. Copeia 1:169–178
Bjornstad ON (2018) NCF: spatial covariance functions. R package version 1.2–5. https://CRAN.R-project.org/package=ncf. Accessed Dec 2019
Blowes SA, Pratchett MS, Connolly SR (2013) Heterospecific aggression and dominance in a guild of coral-feeding fishes: the roles of dietary ecology and phylogeny. Am Nat 182:157–168. https://doi.org/10.1086/670821
Bouchon-Navaro Y (1986) Partitioning of food and space resources by chaetodontid fishes on coral reefs. J Exp Mar Biol Ecol 103:21–40. https://doi.org/10.1016/0022-0981(86)90130-9
Brown JH (1984) On the relationship between abundance and distribution of species. Am Nat 124:255–279. https://doi.org/10.1086/284267
Brown WL, Wilson EO (1956) Character displacement. Syst Zool 5:49–64. https://doi.org/10.2307/2411924
Burns JH, Strauss SY (2011) More closely related species are more ecologically similar in an experimental test. Proc Natl Acad Sci USA 108:5302–5307. https://doi.org/10.1073/pnas.1013003108
Copus JM, Pyle RL, Greene BD, Randall JE (2019) Prognathodes geminus, a new species of butterflyfish (Teleostei, Chaetodontidae) from Palau. Zookeys 835:125–137. https://doi.org/10.3897/zookeys.835.32562
Cowman PF, Bellwood DR (2011) Coral reefs as drivers of cladogenesis: expanding coral reefs, cryptic extinction events, and the development of biodiversity hotspots. J Evol Biol 24:2543–2562. https://doi.org/10.1111/j.1420-9101.2011.02391.x
Delmas E, Besson M, Brice MH, Burkle LA, Riva GVD, Fortin MJ, Gravel D, Guimarães PR, Hembry DH, Newman EA, Olesen JM, Pires MM, Yeakel JD, Poisot T (2019) Analysing ecological networks of species interactions. Biol Rev 94:16–36. https://doi.org/10.1111/brv.12433
Diez JM, Sullivan JJ, Hulme PE, Edwards G, Duncan RP (2008) Darwin’s naturalization conundrum: dissecting taxonomic patterns of species invasions. Ecol Lett 11:674–681. https://doi.org/10.1111/j.1461-0248.2008.01178.x
Early R, Keith SA (2019) Geographically variable biotic interactions and implications for species ranges. Glob Ecol Biogeogr 28:42–53. https://doi.org/10.1111/geb.12861
Ekman EP (1953) Zoogeography of the Sea. Sidgwick & Jackson, London, p 417
Eurich JG, Matley JK, Baker R, McCormick MI, Jones GP (2019) Stable isotope analysis reveals trophic diversity and partitioning in territorial damselfishes on a low-latitude coral reef. Mar Biol 166:17. https://doi.org/10.1007/s00227-018-3463-3
Fessler JL, Westneat MW (2007) Molecular phylogenetics of the butterflyfishes (Chaetodontidae): taxonomy and biogeography of a global coral reef fish family. Mol Phylogenet Evol 45:50–68. https://doi.org/10.1016/j.ympev.2007.05.018
Findley JS, Findley MT (1989) Circumtropical patterns in butterflyfish communities. Environ Biol Fishes 25:33–46. https://doi.org/10.1007/BF00002199
Floeter SR, Vázquez DP, Grutter AS (2007) The macroecology of marine cleaning mutualisms. J Anim Ecol 76:105–111. https://doi.org/10.1111/j.1365-2656.2006.01178.x
Floeter SR, Rocha LA, Robertson DR, Joyeux JC, Smith-Vaniz WF, Wirtz P, Edwards AJ, Barreiros JP, Ferreira CEL, Gasparini JL, Brito A, Falcón JM, Bowen BW, Bernardi G (2008) Atlantic reef fish biogeography and evolution. J Biogeogr 35:22–47. https://doi.org/10.1111/j.1365-2699.2007.01790.x
Floeter SR, Bender MG, Siqueira AC, Cowman PF (2018) Phylogenetic perspectives on reef fish functional traits. Biol Rev 93:131–151. https://doi.org/10.1111/brv.12336
Fontoura L, Cantor M, Longo GO, Bender MG, Bonaldo RM, Floeter SR (2020) The macroecology of reef fish agonistic behaviour. Ecography 43:1–13. https://doi.org/10.1111/ecog.05079
Gaboriau T, Leprieur F, Mouillot D, Hubert N (2018) Influence of the geography of speciation on current patterns of coral reef fish biodiversity across the Indo-Pacific. Ecography 41:1295–1306. https://doi.org/10.1111/ecog.02589
Germain RM, Weir JT, Gilbert B (2016) Species coexistence: macroevolutionary relationships and the contingency of historical interactions. Proc Biol Sci 283:20160047. https://doi.org/10.1098/rspb.2016.0047
Harper JL, Clatworthy JN, McNaughton IH, Sagar GR (1961) The evolution and ecology of closely related species living in the same area. Evolution 15:209–227. https://doi.org/10.2307/2406081
Hodge JR, Bellwood DR (2016) The geography of speciation in coral reef fishes: the relative importance of biogeographical barriers in separating sister-species. J Biogeogr 43:1324–1335. https://doi.org/10.1111/jbi.12729
Hodge JR, Herwerden LV, Bellwood DR (2014) Temporal evolution of coral reef fishes: global patterns and disparity in isolated locations. J Biogeogr 41:2115–2127. https://doi.org/10.1111/jbi.12356
Hynes HBN (1950) The food of fresh-water sticklebacks (Gasterosteus aculeatus and Pygosteus pungitius), with a review of methods used in studies of the food of fishes. J Anim Ecol 19:36–58. https://doi.org/10.2307/1570
Hyslop EJ (1980) Stomach contents analysis—a review of methods and their application. J Fish Biol 17:411–429. https://doi.org/10.1111/j.1095-8649.1980.tb02775.x
Kawakami E, Vazzoler G (1980) Método gráfico e estimativa de índice alimentar aplicado no estudo de alimentação de peixes. Braz J Oceanogr 29:205–207. https://doi.org/10.1590/S0373-55241980000200043
Keith SA, Baird AH, Hobbs JPA, Woolsey ES, Hoey AS, Fadli N, Sanders NJ (2018) Synchronous behavioural shifts in reef fishes linked to mass coral bleaching. Nat Clim Chang 8:986–991. https://doi.org/10.1038/s41558-018-0314-7
Konow N, Ferry LA (2013) Functional morphology of butterflyfishes. In: Pratchett MS, Berumen ML, Kapoor BG (eds) Biology of butterflyfishes. CRC Press, Cambridge, pp 19–47
Kuiter RH (2002) Butterflyfishes, bannerfishes and their relatives: a comprehensive guide to chaetodontidae and microcanthidae. TMC Publishing, London, p 208
Kulbicki M, Vigliola L, Wantiez L, Hubert N, Floeter SR, Myers RF (2013) Biogeography of butterflyfishes: a global model for reef fishes? In: Pratchett MS, Berumen ML, Kapoor BG (eds) Biology of butterflyfishes. CRC Press, Cambridge, pp 70–106
Liedke AMR, Barneche DR, Ferreira CEL, Segal B, Nunes LT, Burigo AP, Carvalho JA, Buck S, Bonaldo RM, Floeter SR (2016) Abundance, diet, foraging and nutritional condition of the banded butterflyfish (Chaetodon striatus) along the western Atlantic. Mar Biol 163:6. https://doi.org/10.1007/s00227-015-2788-4
Liedke AMR, Bonaldo RM, Segal B, Ferreira CEL, Nunes LT, Burigo AP, Buck S, Oliveira-Santos LGR, Floeter SR (2018) Resource partitioning by two syntopic sister-species of butterflyfish (Chaetodontidae). J Mar Biol Assoc 98:1767–1773. https://doi.org/10.1017/S0025315417001321
Lobato FL, Barneche DR, Siqueira AC, Liedke AMR, Lindner A, Pie MR, Bellwood DR, Floeter SR (2014) Diet and diversification in the evolution of coral reef fishes. PLoS ONE 9:e102094. https://doi.org/10.1371/journal.pone.0102094
Mariani S, Piscitelli MP, Uriz MJ (2001) Temporal and spatial co-occurrence in spawning and larval release of Cliona viridis (Porifera: Hadromerida). J Mar Biol Assoc 81:565–567. https://doi.org/10.1017/S0025315401004246
McGill B, Collins C (2003) A unified theory for macroecology based on spatial patterns of abundance. Evol Ecol Res 5:469–492
Morais RA, Ferreira CEL, Floeter SR (2017) Spatial patterns of fish standing biomass across Brazilian reefs, Southwestern Atlantic. J Fish Biol 91:1642–1667. https://doi.org/10.1111/jfb.13482
Munday PL (2004) Habitat loss, resource specialization, and extinction on coral reefs. Glob Change Biol 10:1642–1647. https://doi.org/10.1111/j.1365-2486.2004.00839.x
Nagelkerken I, Van der Velde G, Wartenbergh SL, Nugues MM, Pratchett MS (2009) Cryptic dietary components reduce dietary overlap among sympatric butterflyfishes (Chaetodontidae). J Fish Biol 75:1123–1143. https://doi.org/10.1111/j.1095-8649.2009.02303.x
Narwani A, Alexandrou MA, Oakley TH, Carroll IT, Cardinale BJ (2013) Experimental evidence that evolutionary relatedness does not affect the ecological mechanisms of coexistence in freshwater green algae. Ecol Lett 16:1373–1381. https://doi.org/10.1111/ele.12182
Nickell LA, Sayer MDJ (1998) Occurrence and activity of mobile macrofauna on a sublittoral reef: diel and seasonal variation. J Mar Biol Assoc 78:1061–1082. https://doi.org/10.1017/S0025315400044325
Nunes LT, Cord I, Francini-Filho RB, Stampar SN, Pinheiro HT, Rocha LA, Floeter SR, Ferreira CEL (2019) Ecology of Prognathodes obliquus, a butterflyfish endemic to mesophotic ecosystems of St. Peter and St. Paul’s Archipelago. Coral Reefs. https://doi.org/10.1007/s00338-019-01822-8
Paradis E, Claude J, Strimmer K (2004) APE: analyses of phylogenetics and evolution in R language. Bioinformatics 20:289–290
Pfennig DW, Rice AM, Martin RA (2006) Ecological opportunity and phenotypic plasticity interact to promote character displacement and species coexistence. Ecology 87:769–779. https://doi.org/10.1890/05-0787
Pratchett MS (2005) Dietary overlap among coral-feeding butterflyfishes (Chaetodontidae) at Lizard Island, northern Great Barrier Reef. Mar Biol 148:373–382. https://doi.org/10.1007/s00227-005-0084-4
Pratchett MS, Wilson SK, Berumen ML, McCormick MI (2004) Sublethal effects of coral bleaching on an obligate coral feeding butterflyfish. Coral Reefs 23:352–356. https://doi.org/10.1007/s00338-004-0394-x
Pratchett MS, Berumen ML, Kapoor BG (2014) Biology of Butterflyfishes. CRC Press, Cambridge
R Core Team (2019) R: a language and environment for statistical computing.
Reaka-Kudla ML (1997) The global biodiversity of coral reefs: a comparison with rain forests. In: Reaka-Kudla ML, Wilson DE, Wilson EO (eds) Biodiversity II: understanding and protecting our biological resources. Joseph Henry Press, Washington, pp 83–108
Reese ES (1981) Predation on corals by fishes of the family Chaetodontidae: implications for conservation and management of coral reef ecosystems. Bull Mar Sci 31:594–604
Roberts CM, McClean CJ, Veron JE, Hawkins JP, Allen GR, McAllister DE, Mittermeier CG, Schueler FW, Spalding M, Wells F, Vynne C, Werner TB (2002) Marine biodiversity hotspots and conservation priorities for tropical reefs. Science 295:1280–1284. https://doi.org/10.1126/science.1067728
Schoener TW (1971) Theory of feeding strategies. Annu Rev Ecol Evol Syst 2:369–404. https://doi.org/10.1146/annurev.es.02.110171.002101
Siqueira A, Morais R, Bellwood D, Cowman P (2020) Trophic innovations fuel reef fish diversification. Nat Commun 11:2669. https://doi.org/10.1038/s41467-020-16498-w
Slatkin M (1980) Ecological character displacement. Ecology 61:163–177. https://doi.org/10.2307/1937166
Thuiller W, Gallien L, Boulangeat I, Bello FD, Münkemüller T, Roquet C, Lavergne S (2010) Resolving Darwin’s naturalization conundrum: a quest for evidence. Divers Distrib 16:461–475. https://doi.org/10.1111/j.1472-4642.2010.00645.x
Vázquez DP, Melian CJ, Williams NM, Bluthgen N, Krasnov BR, Poulin R (2007) Species abundance and asymmetric interaction strength in ecological networks. Oikos 116:1120–1127. https://doi.org/10.1111/j.0030-1299.2007.15828.x
Vázquez DP, Bluthgen N, Cagnolo L, Chacoff NP (2009) Uniting pattern and process in plant–animal mutualistic networks: a review. Ann Bot 103:1445–1457. https://doi.org/10.1093/aob/mcp057
Vine NG (1998) Aspects of the biology of the doublesash butterflyfish, Chaetodon marleyi (Pisces: Chaetodontidae). MSc. Thesis, Rhodes University, South Africa, p 115
Whittaker RH (1972) Evolution and measurement of species diversity. Taxon 21:213–251
Acknowledgements
We thank Mauricio Cantor and Luiz Gustavo Rodrigues Oliveira Santos for statistical help and insights. We thank Alana Fraga for assistance with the stomach content analyses. We thank A Bernardi, G Bernardi, E Faria, R Freitas, A Jackson, AM Millán, R Morais, L Rocha and P Vasconcelos for sampling and ield support. We thank A Lindner, B Segal, S Stampar and T Silveira for helping with the identiication of food items, insights and laboratory equipment. We thank P Cowman for providing the phylogeny. We also thank the three anonymous reviewers who helped to improve the manuscript.
Funding
This study was funded by Conselho Nacional de Desenvolvimento Cientifico e Tecnologico (Grant numbers 483682/2010-1, 563276/2010-0, 403740/2012-6), Fundação de Amparo à Pesquisa e Inovação do Estado de Santa Catarina (Grant number 6308/2011-8). The sampling was supported by Chaetodon project [CNPq #483682/2010-1 to SRF], SISBIOTA-Mar [CNPq #563276/2010-0 and FAPESC #6308/2011-8; to SRF], Long Term Ecological Research of Brazilians Oceanic Islands—PELD/ILOC [CNPq #403740/2012-6 to CELF], PROSPEC-Mar [CNPq #458548/2013-8 to Dr. Leticia Veras Costa Lotufo]. LTN and IC received a scholarship from the Brazilian Ministry Educational Council—[CAPES Finance Code 001].
Author information
Authors and Affiliations
Contributions
LTN and SRF conceived the ideas and designed the study; LTN, ACS and BMF collected the data and/or performed statistical analyses; LTN and IC performed the stomach content analyses. LTN, IC, AMRL and CELF contributed to sampling effort; LTN led the writing; all the authors contributed to the writing and discussions. All authors gave final approval for publication.
Corresponding author
Ethics declarations
Conflict of interest
We declare that there is no conflict of interest.
Ethical approval
We also declare that all individuals were sampled according to the international and national guidelines for sampling. Permission for sampling in Brazil was given by the ICMBio Institute (SISBio #55911-3 and #29953-10 to CEFL; and SISBio #41327-9 to SRF); In Ascension island permission was given by the Conservation Centre of the Ascension Government (ERP-2015-09 to Dr. Renato Morais). Chaetodon robustus individuals were obtained from the University of Cabo Verde (reference collection voucher: UCV 2020/00018-00024).
Additional information
Responsible Editor: D. Goulet.
Reviewed by undisclosed experts.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.



Rights and permissions
About this article
Cite this article
Nunes, L.T., Siqueira, A.C., Cord, I. et al. The influence of species abundance, diet and phylogenetic affinity on the co-occurrence of butterflyfishes. Mar Biol 167, 107 (2020). https://doi.org/10.1007/s00227-020-03725-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-020-03725-7