Abstract
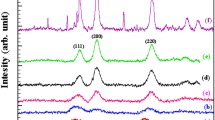
Ni nanopowder was synthesized from NiO through mechanically induced magnesiothermic NiO–Mg reaction that can also be regarded as a kind of progressive synthesis, or Mechanically induced Self-sustaining Reaction (MSR). The obtained powder was characterized by XRD, FESEM, and TEM. After 8 min of milling, the mixture ignited in the mill vial to yield Ni and MgO. The latter was removed by leaching with 18% HCl. The mean crystallite size of resultant Ni was found to have a value of 45–50 nm.






Similar content being viewed by others
REFERENCES
Chaturvedi, S., Dave, P.N., and Shah, N.K., Applications of nano-catalysts in new era, J. Saudi Chem. Soc., 2012, vol. 16, no. 3, pp. 307–325. https://doi.org/10.1016/j.jscs.2011.01.015
Mary, A.R., Sandeep, C.S., Narayanan, T.N., Philip, R., Moloney, P., Ajayan, P.M., and Anantharaman, M.R., Nonlinear and magneto-optical transmission studies on magnetic nanofluids of non-interacting metallic nickel nanoparticles, Nanotechnology, 2011, vol. 22, no. 37, 375702. https://doi.org/10.1088/0957-4484/22/37/375702
Pascu, O., Caicedo, J.M., Fontcuberta, J., Herranz, G., and Roig, A., Magneto-optical characterization of colloidal dispersions: Application to nickel nanoparticles, Langmuir, 2010, vol. 26, no. 15, pp. 12548–12552. https://doi.org/10.1021/la1011617
Xu, C., Hu, Y., Rong, J., Jiang, SP., and Liu, Y., Ni hollow spheres as catalysts for methanol and ethanol electrooxidation, Electrochem. Commun., 2007, vol. 9, no. 8, pp. 2009–2012. https://doi.org/10.1016/j.elecom.2007.05.028
Metin, O., Mazumder, V., Ozkar, S., and Sun, S., Monodisperse nickel nanoparticles and their catalysis in hydrolytic dehydrogenation of ammonia borane, J. Am. Chem. Soc., 2010, vol. 132, no. 5, pp. 1468–1469. https://doi.org/10.1021/ja909243z
Aguilhon, J., Boissière, C., Durupthy, O., Thomazeau, C., and Sanchez, C., Nickel nanoparticles with controlled morphologies: Application in selective hydrogenation catalysis, Stud. Surf. Sci. Catal., 2010, vol. 175, pp. 521–524. https://doi.org/10.1016/S0167-2991(10)75099-0
Mai, Y.J., Tu, J.P., Gu, C.D., and Wang, X.L., Graphene anchored with nickel nanoparticles as a high-performance anode material for lithium ion batteries, J. Power Sources, 2012, vol. 209, pp. 1–6. https://doi.org/10.1016/j.jpowsour.2012.02.073
Dung, N.Q., Patil. D., Jung, H., Kim, J., and Kim, D., NiO-decorated single-walled carbon nanotubes for high-performance non-enzymatic glucose sensing, Sens. Actuators B: Chem., 2013, vol. 183, pp. 381–387. https://doi.org/10.1016/j.snb.2013.04.018
Hernández, P.T., Kuznetsov, M.V., Morozov, Y.G., and Parkin, I.P., Application of levitation-jet synthesized nickel-based nanoparticles for gas sensing, Mater. Sci. Eng. B, 2019, vol. 244, pp. 81–92. https://doi.org/10.1016/j.mseb.2019.05.003
Neiva, E.G., Oliveira, M.M., Marcolino Jr, L.H., and Zarbin, A.J., Nickel nanoparticles with hcp structure: Preparation, deposition as thin films and application as electrochemical sensor, J. Colloid Interf. Sci., 2016, vol. 468, pp. 34–41. https://doi.org/10.1016/j.jcis.2016.01.036
Tehrani, R.M. and Ab Ghani, S., The nanocrystalline nickel with catalytic properties for methanol oxidation in alkaline medium, Fuel Cells, 2009, vol. 9, no. 5, pp. 579–587. https://doi.org/10.1002/fuce.200800122
Ewing, S.J., Lan, R., Xu, X.X., and Tao, S.W., Synthesis of dendritic nano-sized nickel for use as anode material in an alkaline membrane fuel cell, Fuel Cells, 2010, vol. 10, no. 1, pp. 72–76. https://doi.org/10.1002/fuce.200900102
Sada, T., Izawa, K., Fujikawa, N., and Fujioka, Y., Mechanism of pre-breakdown process in Ni–BaTiO3 multilayer ceramic capacitors, Jpn. J. Appl. Phys., 2018, vol. 57, no. 11S. https://doi.org/10.7567/JJAP.57.11UC02
Wu, Z.G., Munoz, M., and Montero, O., The synthesis of nickel nanoparticles by hydrazine reduction, Adv. Powder Technol., 2010, vol. 21, no. 2, pp. 165–168. https://doi.org/10.1016/j.apt.2009.10.012
Zhang, H.T., Wu, G., Chen, X.H., and Qiu, X.G., Synthesis and magnetic properties of nickel nanocrystals, Mater. Res. Bull., 2006, vol. 4, no. 3, pp. 495–501. https://doi.org/10.1016/j.materresbull.2005.09.019
Li, D. and Komarneni, S., Microwave-assisted polyol process for synthesis of Ni nanoparticles, J. Am. Ceram. Soc., 2006, vol. 89, no. 5, pp. 1510–1517. https://doi.org/10.1111/j.1551-2916.2006.00925.x
Neiva, E.G., Bergamini, M.F., Oliveira, M.M., Marcolino Jr, L.H., and Zarbin, A.J., PVP-capped nickel nanoparticles: Synthesis, characterization and utilization as a glycerol electrosensor, Sens. Actuators B: Chem., 2014, vol. 196, pp. 574–581. https://doi.org/10.1016/j.snb.2014.02.041
Liu, Y., Li, X., Li, Y., Chen, J., Zhao, W., and Bai, F., The formation mechanism and morphology of the nickel particles by the ultrasound-aided spark discharge in different liquid media, Adv. Powder Technol., 2016, vol. 27, no. 6, pp. 2399–2408. https://doi.org/10.1016/j.apt.2016.08.016
Hou, Y., Kondoh, H., Ohta, T., and Gao, S., Size-controlled synthesis of nickel nanoparticles, Appl. Surf. Sci., 2005, vol. 241, nos. 1–2, pp. 218–222. https://doi.org/10.1016/j.apsusc.2004.09.045
Wang, D.P., Sun, D.B., Yu, H.Y., and Meng, H.M., Morphology controllable synthesis of nickel nanopowders by chemical reduction process, J. Cryst. Growth, 2008, vol. 310, no. 6, pp. 1195–1201. https://doi.org/10.1016/j.jcrysgro.2007.12.052
Bai, L., Fan, J., Hu, P., Yuan, F., Li, J., and Tang, Q., RF plasma synthesis of nickel nanopowders via hydrogen reduction of nickel hydroxide/carbonate, J. Alloys Comp., 2009, vol. 481, nos. 1–2, pp. 563–567. https://doi.org/10.1016/j.jallcom.2009.03.054
Davoodi, D., Miri, R., Emami, A.H., Tayebi, M., and Salahshour, S., The effect of NiO catalyst on reduction, synthesis and binder content of TiC–Ni nanocomposite, Int. J. Refract. Met. Hard Mater., 2019, vol. 88. https://doi.org/10.1016/j.ijrmhm.2019.105175
Davoodi, D., Tayebi, M., Miri, R., Emami, A.H., and Salahshour, S., An investigation of the reduction mechanisms and magnesiothermic reactions in ZrC–Ni nanocomposite synthesis, Mater. Chem. Phys., vol. 222, pp. 351–360. https://doi.org/10.1016/j.matchemphys.2018.10.014
Davoodi, D., Emami, A.H., Tayebi, M., and Hosseini, S.K., Rapid mechanochemical synthesis of nickel–vanadium carbide nanocomposite powder by magnesiothermic reaction, Ceram. Int., vol. 44, no. 5, pp. 5411–5419. https://doi.org/10.1016/j.ceramint.2017.12.170
Sharifi, H., Tayebi, M., and Honarmand, M., Production of a nanocrystalline composite of Al–4% Cu/SiC by mechanical milling method, Mater. Res. Express, vol. 3, no. 10. https://doi.org/10.1088/2053-1591/3/10/105050
Wu, Z.G., Munoz, M., and Montero, O., The synthesis of nickel nanoparticles by hydrazine reduction, Adv. Powder Technol., 2010, vol. 21, no. 2, pp. 165–168. https://doi.org/10.1016/j.apt.2009.10.012
Baláž, P., Mechanochemistry in minerals engineering, in Mechanochemistry in Nanoscience and Minerals Engineering, Berlin–Heidelberg: Springer, 2008, pp. 257–296. https://doi.org/10.1007/978-3-540-74855-7
Sharifi, H., Faradonbeh, S.R., and Tayebi, M., Production and characterization of cobalt/vanadium boride nanocomposite powder by mechanochemical method, Mater. Chem. Phys., 2017, vol. 202, pp. 251–257. https://doi.org/10.1016/j.matchemphys.2017.09.015
Suryanarayana, C., Mechanical alloying and milling. Prog. Mater. Sci., 2001, vol. 46, nos. 1–2, pp. 1–184. https://doi.org/10.1016/S0079-6425(99)00010-9
Takacs, L., Self-sustaining reactions induced by ball milling, Prog. Mater. Sci., 2002, vol. 47, no. 4, pp. 355–414. https://doi.org/10.1016/S0079-6425(01)00002-0
Moore, J.J. and Feng, H.J., Combustion synthesis of advanced materials: I. Reaction parameters, Prog. Mater. Sci., 1995, vol. 39, nos. 4–5, pp. 243–273. https://doi.org/10.1016/0079-6425(94)00011-5
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Gharibi, M., Sharifi, H., Nilforoushan, M.R. et al. Nickel Nanoparticles by Mechanical Synthesis in the NiO–Mg System. Int. J Self-Propag. High-Temp. Synth. 29, 108–114 (2020). https://doi.org/10.3103/S1061386220020065
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3103/S1061386220020065