Abstract
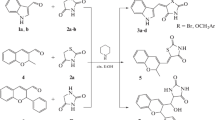
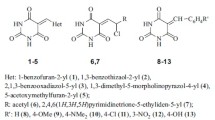
Bioisosteric replacements are often tried goaling to affect the lipophilicity, polarity, and aqueous solubility of the substances, as a way to obtain therapeutically improved medicines. Also, hydrazonyl compounds are described with a wide range of pharmacological activities, having also recognized activities in antimycobacterial field. In this study, twenty-seven pyrimidinyl and pyrazinyl derivatives have been synthesized and evaluated for their antimycobacterial activity against M. tuberculosis ATTC 27294. The componds were obtained by the reaction of 2-hydrazinylpyridine, 4-hydrazinylpyridine, or 2-hydrazinylpyrazine with appropriated aromatic or heteroaromatic aldehydes. Antimycobacterial activity of the compounds was determined according to MTT assay. The most active compound, a 2-hydroxyl-5-nitrophenyl-4-pyridinylhydrazone derivative, showed good biodisponibility and nonmutagenic or tumorigenic profiles in preliminaries in silico studies, and exceptional in vitro activity, being compared with the reference drug ethambutol. This study supports that pyrimidinyl and pyrazinyl derivatives may be used for the development of new tuberculostatic agents.

Similar content being viewed by others
References
Alptüzün V, Parlar S, Taşlı H, Erciyas E (2009) Synthesis and antimicrobial activity of some pyridinium salts. Molecules 14:5203–5215
Amim RS, Firmino GSS, Rego ACPD, Nery AL, Da Silva SAG, Figueroa-Villar JD, Resende JALC, De Souza MVN, Pessoa CO, Lessa JA (2016) Cytotoxicity and leishmanicidal activity of isoniazid-derived hydrazones and 2-pyrazineformamide thiosemicarbazones. J Braz Chem Soc 27:769–777
Berge DG (1983) Synthesis of new 2-pyridylhydrazones and 2-quinolylhydrazones containing 2-thiophene or 2-furan groups. J Chem Eng Data 28:431–432
Brahmachari G (2018) Synthesis of biologically relevant heterocycles in aqueous media. Asian J Org Chem 7:1982–2004
Candéa ALP, Ferreira ML, Pais KC, Cardoso LNF, Kaiser CR, Henriques MGMO, Lourenço MCS, Bezerra FAFM, De Souza MVN (2009) Synthesis and antitubercular activity of 7-chloro-4-quinolinylhydrazones derivatives. Bioorg Med Chem Lett 19:6272–6274
Cardoso LNF, Nogueira TCM, Rodrigues FAR, Oliveira ACA, Luciano MCS, Pessoa CO, De Souza MVN (2017) N-acylhydrazones containing thiophene nucleus: a new anticancer class. Med Chem Res 26:1605–1608
Cardoso LNF, Bispo MLF, Kaiser CR, Wardell JL, Wardell SMSV, Lourenço MCS, Bezerra FAFM, Soares RPP, Rocha MN, De Souza MVN (2014) Anti-tuberculosis evaluation and conformational study of N -acylhydrazones containing the thiophene nucleus. Arch Pharm 347:432–448
Case FH (1976) Preparation of new 2-pyridyl and pyrazinylhydrazones containing ferroin group. J Chem Eng Data 21:124–125
Coimbra ES, Antinarelli LMR, Da Silva AD, Bispo MLF, Kaiser CR, De Souza MVN (2013) 7-chloro-4-quinolinylhydrazones: a promising and potent class of antileishmanial compounds. Chem Biol Drug Des 81:658–665
Cushman M, Nagarathnam D, Gopal D, Geahlen RL (1991) Synthesis and evaluation of new protein-tyrosine kinase inhibitors. Part 2. Phenylhydrazones. Bioorg Med Chem Lett 1:215–218
De Souza MVN, Coimbra ES, Antinarelli LMR, Crispi MA, Nogueira TCM, Pinheiro AC (2018) Synthesis, biological activity, and mechanism of action of 2-pyrazyl and pyridylhydrazone derivatives, new classes of antileishmanial agents. ChemMedChem 13:1387–1394
Dua R, Shrivastava S, Sonwane SK, Srivastava SK (2011) Pharmacological significance of synthetic heterocycles scaffold: a review. Adv Biol Res 5:120–144
El-Sayed TH, Aboelnaga A, El-Atawy MA, Hagar M (2018) Ball milling promoted N-heterocycles synthesis. Molecules 23:1348
Lions F, Martin KV (1958) Sexadentate chelate compounds.1X. JACS 80:3858–3865
Hegarty AF, Moroney PJ, Scott FL (1973) A change from rate-determining bromination to geometric isomerisation of pyridylhydrazones. J Chem Soc Perkin Trans 2:1466–1471
Kakemi K, Uno T, Arita T, Nakano H, Shimada I, Ikegami Y, Kitazawa S (1961) Synthesis of pyrazinoic acid derivatives. I. Derivatives of pyrazinoic acid, aminopyrazine, pyrazinohydrazide, and pyrazinecarboxaldehyde. Yakugaku Zasshi 81:1609–1614
Khanna P, Khanna L, Thomas SJ, Asiri AM, Panda SS (2018) Microwave assisted synthesis of spiro heterocyclic systems: a review. Curr Org Chem 22:67–84
Mahmud NP, Garrett SW, Threadgill MD (1998) The 5-nitrofuran-2-ylmethylidene group as a potential bioreductively activated prodrug system for diol-containing drugs. Anticancer Drug Des 13:655–662
Martins P, Jesus J, Santos S, Raposo LR, Roma-Rodrigues C, Baptista PV, Fernandes AR (2015) Heterocyclic anticancer compounds: recent advances and the paradigm shift towards the use of nanomedicine’s tool box. Molecules 20:16852–16891
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65:55–63
Nogueira TCM, Cruz LS, Lourenço MCS, De Souza MVN (2019) Design, synthesis and anti-tuberculosis activity of hydrazones and N-acylhydrazones containing vitamin B6 and different heteroaromatic nucleus. Lett Drug Des Disco 16:792–798
Przybylski P, Pyta-Klich K, Pyta K, Janas A (2018) Cascade reactions as efficient and universal tools for construction and modification of 6-, 5-, 4- and 3-membered sulfur heterocycles of biological relevance. Tetrahedron 74:6335–6365
Rakesh, Bruhn DF, Scherman MS, Woolhiser LK, Madhura DB, Maddox MM, Singh AP, Lee RB, Hurdle JG, McNeil MR, Lenaerts AJ, Meibohm B, Lee RE (2014) Pentacyclic nitrofurans with in vivo efficacy and activity against nonreplicating mycobacterium tuberculosis. PLoS ONE 9:e8790
Rodrigues FAR, Bomfim IS, Cavalcanti BC, Pessoa CO, Wardell JL, Solange SMSV, Pinheiro AC, Kaiser CR, Nogueira TCM, Low JN, Gomes LR, de Souza MVN (2014) Design, synthesis and biological evaluation of (E)-2-(2-arylhydrazinyl)quinoxalines, a promising and potent new class of anticancer agents. Bioorg Med Chem Lett 24(3):934–939
Rollas S, Küçükgüzel ŞG (2007) Biological activities of hydrazone derivatives. Molecules 12:1910–1939
Sandbhor U, Padhye S, Billington D, Rathbone D, Franzblau S, Anson CE, Powell AK (2002) Metal complexes of carboxamidrazone analogs as antitubercular agents. 1. Synthesis, X-ray crystal-structures, spectroscopic properties and antimycobacterial activity against Mycobacterium tuberculosis H(37)Rv. J Inorg Biochem 90:127–136
Sondhi SM, Jain S, Dinodia M, Kumar A (2008) Synthesis of some thiophene, imidazole and pyridine derivatives exhibiting good anti-inflammatory and analgesic activities. Med Chem 4:146–154
Squella JA, Bollo S, Núñez-Vergara LJ (2005) Recent Developments in the Electrochemistry of Some Nitro Compounds of Biological Significance. Curr Org Chem 9:565–581
Syeda N, Srinivasan VR (1965) Some fused S-triazoles derived from aza-heterocyclic hydrazones. Indian J Chem 3:162–167
Taylor AP, Robinson RP, Fobian YM, Blakemore DC, Jones LH, Fadeyi O (2016) Modern advances in heterocyclic chemistry in drug discovery. Org Biomol Chem 14:6611–6637
Verma G, Marella A, Shaquiquzzaman M, Akhtar M, Rahmat Ali M, Alam MM (2014) A review exploring biological activities of hydrazones. J Pharm Bioallied Sci 6:69–80
Acknowledgements
We are grateful to the Brazilian agency CNPq (Conselho Nacional de Desenvolvimento Cientifico e Tecnológico) for fellowships and financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Pinheiro, A.C., Nogueira, T.C.M., Pereira, G.E. et al. Synthesis and antimycobacterial evaluation of pyridinyl- and pyrazinylhydrazone derivatives. Med Chem Res 29, 1662–1668 (2020). https://doi.org/10.1007/s00044-020-02592-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-020-02592-7