Abstract
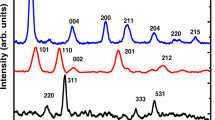
Nanostructured iron–zirconium oxides (NIZOs), with Fe-to-Zr molar ratio of 1:1 and 1:2, have been found to show high adsorption efficiency toward Pb2+ from aqueous solution. The NIZO was synthesized by the co-precipitation method and characterized in terms of XRD, TEM, SEM–EDX, BET surface area analysis, TGA, FT-IR spectroscopy and zeta potential measurement. The data of adsorption of Pb2+ on the NIZO, under optimized condition of pH, Pb2+ concentration, adsorbent amount, time and temperature, were fitted in Langmuir, Freundlich and Temkin adsorption model, and the data showed the best agreement with Langmuir model with R2 = 0.990 and 0.994 for Fe/Zr molar ratio = 1:1 and 1:2, respectively. The thermodynamic studies showed that the adsorption of Pb2+ on the NIZO proceeds spontaneously and exothermically through the involvement of weak van der Waals forces. Further, the adsorption of Pb2+ on the NIZO was found to follow pseudo-second-order kinetics.













Similar content being viewed by others
References
Ahuja, S.: Lessons learned from water disasters of the world. Separ. Sci. Tech. 11, 417–427 (2019)
Kabeer, A.; Mailafiya, M.M.; Danmaigoro, A.; Rahim, E.A.; bu Bakar, M.Z.A.: Therapeutic potential of curcumin against lead-induced toxicity: a review. Biomed. Res. Therap. 6, 3053–3066 (2019)
Carocci, A.; Catalano, A.; Lauria, G.; Sinicropi, M.S.; Genchi, G.: Lead toxicity, antioxidant defense and environment. Rev. Environ. Contam. Toxicol. 238, 45–67 (2016)
El Henafy, H.M.; Ibrahim, M.A.; El Aziz, S.A.A.; Gouda, E.M.: Oxidative stress and DNA methylation in male rat pups provoked by the transplacental and translactational exposure to bisphenol A. Environ. Sci. Pollut. Res. 27, 1–7 (2019)
Ahmed, M.O.; Ibrahim, M.N.; Mohammed, S.M.: Physiological and histological effects of broccoli on lead acetate induced hepatotoxicity in young male albino rats. IIJVS 33, 21–26 (2019)
Mohiuddin, A.K.: Heavy metals in cosmetics: the notorious daredevils and burning health issues. Am. J. Biomed. Sci. Res. 4, 332–337 (2019)
Ensafi, A.M.; Moghadam, P.N.; Baradarani, M.M.: Synthesis of a new nanocomposite based-on graphene-oxide for selective removal of Pb2+ ions from aqueous solutions. Polym. Compos. 40, 730–737 (2019)
Sharma, D.; Chaudhari, P.K.; Prajapati, A.K.: Removal of chromium (VI) and lead from electroplating effluent using electrocoagulation. Sep. Sci. Technol. 55, 1–11 (2019)
Bora, A.J.; Dutta, R.K.: Removal of metals (Pb, Cd, Cu, Cr, Ni and Co) from drinking water by oxidation-coagulation-absorption at optimized pH. J. Water Process. Eng. 31, 100839 (2019)
Ali, S.; Rehman, S.A.U.; Luan, H.Y.; Farid, M.U.; Huang, H.: Challenges and opportunities in functional carbon nanotubes for membrane-based water treatment and desalination. Sci. Total Environ. 646, 1126–1139 (2019)
Qu, J.; Song, T.; Liang, J.; Bai, X.; Li, Y.; Wei, Y.; Huang, S.; Dong, L.; Jin, Y.U.: Adsorption of lead (II) from aqueous solution by modified Auricularia matrix waste: a fixed-bed column study. Ecotoxicol. Environ. Saf. 169, 722–729 (2019)
Xu, H.; Yuan, H.; Yu, J.; Lin, S.: Study on the competitive adsorption and correlational mechanism for heavy metal ions using the carboxylated magnetic iron oxide nanoparticles (MNPs-COOH) as efficient adsorbents. Appl. Surf. Sci. 473, 960–966 (2019)
Egirani, D.; Latif, M.T.; Wessey, N.; Poyi, N.R.; Acharjee, S.: Synthesis and characterization of kaolinite coated with copper oxide and its effect on the removal of aqueous Lead (II) ions. Appl. Water Sci. 9, 109 (2019)
Tsade, H.; Abebe, B.; Murthy, H.A.: Nano sized Fe–Al oxide mixed with natural maize cob sorbent for lead remediation. Mater. Res. Express 6, 085043 (2019)
Sharma, M.; Singh, J.; Hazra, S.; Basu, S.: Adsorption of heavy metal ions by mesoporous ZnO and TiO2@ZnO monoliths: adsorption and kinetic studies. Microchem. J. 145, 105–112 (2019)
Ibupoto, A.S.; Qureshi, U.A.; Arain, M.; Ahmed, F.; Khatri, Z.; Brohi, R.Z.; Kim, I.S.; Ibupoto, Z.: Zno/Carbon nanofibers for efficient adsorption of lead from aqueous solutions. Environ. Technol. 12, 1–11 (2019)
Xie, X.; Huang, J.; Zhang, Y.; Tong, Z.; Liao, A.; Guo, X.; Qin, Z.; Guo, Z.: Aminated cassava residue-based magnetic microspheres for Pb(II) adsorption from wastewater. Kor. J. Chem. Eng. 36, 226–235 (2019)
Mousavi, S.V.; Bozorgian, A.; Mokhtari, N.; Gabris, M.A.; Nodeh, H.R.; Ibrahim, W.A.W.: A novel cyanopropylsilane-functionalized titanium oxide magnetic nanoparticle for the adsorption of nickel and lead ions from industrial wastewater: equilibrium, kinetic and thermodynamic studies. Microchem. J. 145, 914–920 (2019)
Shi, Q.; Terracciano, A.; Zhao, Y.; Wei, C.; Christodoulatos, C.; Meng, X.: Evaluation of metal oxides and activated carbon for lead removal: kinetics, isotherms, column tests, and the role of co-existing ions. Sci. Total Environ. 648, 176–183 (2019)
Sarma, G.K.; Gupta, S.S.; Bhattacharyya, K.G.: Nanomaterials as versatile adsorbents for heavy metal ions in water: a review. Environ. Sci. Pollut. Res. 26, 6245–6278 (2019)
Huang, T.; Liu, L.; Zhang, S.; Xu, J.: Evaluation of electrokinetics coupled with a reactive barrier of activated carbon loaded with a nanoscale zero-valent iron for selenite removal from contaminated soils. J. Hazard. Mater. 368, 104–114 (2019)
Irawan, C.; Nata, I.F.; Lee, C.K.: Removal of Pb(II) and As (V) using magnetic nanoparticles coated montmorillonite via one-pot solvothermal reaction as adsorbent. J. Environ. Chem. Eng. 7, 103000 (2019)
Lounsbury, A.W.; Wang, R.; Plata, D.L.; Billmyer, N.; Muhich, C.; Kanie, K.; Sugimoto, T.; Peak, D.; Zimmerman, J.B.: Preferential adsorption of selenium oxyanions onto 1 1 0 and 0 1 2 nano-hematite facets. J. Colloid Interface Sci. 537, 465–474 (2019)
Chen, W.; Lu, Z.; Xiao, B.; Gu, P.; Yao, W.; Xing, J.; Asiri, A.M.; Alamry, K.A.; Wang, X.; Wang, S.: Enhanced removal of lead ions from aqueous solution by iron oxide nanomaterials with cobalt and nickel doping. J. Clean. Prod. 211, 1250–1258 (2019)
Lingamdinne, L.P.; Koduru, J.R.; Rao, K.R.: Green synthesis of iron oxide nanoparticles for lead removal from aqueous solutions. Key Eng. Mater. 805, 122–127 (2019)
Gupta, K.; Biswas, K.; Ghosh, U.C.: Nanostructure Fe(III)–Zr(IV) binary mixed oxide: synthesis, characterization, and physicochemical aspects of As(III) sorption from the aqueous solution. Ind. Eng. Chem. Res. 4, 9903–9912 (2008)
Gupta, K.; Basu, T.; Ghosh, U.C.: Sorption characteristics of arsenic(V) for removal from water using agglomerated nanostructure Fe(III)–Zr(IV) bimetal mixed oxide. J. Chem. Eng. 54, 2222–2228 (2009)
Ren, Z.; Zhang, G.; Chen, J.P.: Adsorptive removal of arsenic from water by an iron–zirconium binary oxide adsorbent. J. Colloid Interface Sci. 358, 230–237 (2011)
Ali, S.R.; Arya, M.C.; Kalam, A.; Al-Sehemi, A.G.; Khan, Z.; Ansari, S.; Kumar, R.: Adsorption potential of zirconium-ferrite nanoparticles for phenol, 2-chlorophenol and 2-nitrophenol: thermodynamic and kinetic studies. Desalin. Water Treat. 179, 183–196 (2020)
Ali, S.R.; Kumar, R.; Kadabinakatti, S.K.; Arya, M.C.: Enhanced UV and visible light-driven photocatalytic degradation of tartrazine by nickel-doped cerium oxide nanoparticles. Mater. Res. Express 6, 025513 (2018)
Shahid, S.A.; Nafady, A.; Ullah, I.; Yun, H.; Yap, T.; Shakir, I.; Anwar, F.; Rashid, U.: Characterization of newly synthesized ZrFe2O5 nanomaterial and investigations of its tremendous photocatalytic properties under visible light irradiation. J. Nanomater. 2013, 1–6 (2013)
Atkins, P.; De Paula, J.: Physical Chemistry, 9th edn. W. H. Freeman and Company, New York (2010)
Chang, R.; Thoman Jr., J.W.: Physical Chemistry for the Chemical Sciences. University Science Books, Mill Valley (2014)
Lima, E.C.; Hosseini-Bandegharaei, A.; Moreno-Pirajan, J.C.; Anastopoulos, I.: A critical review of the estimation of the thermodynamic parameters on adsorption equilibria. Wrong use of equilibrium constant in the Van’t Hoff equation for calculation of thermodynamic parameters of adsorption. J. Mol. Liq. 273, 425–434 (2019)
Liu, Y.; Xu, H.: Equilibrium, thermodynamics and mechanisms of Ni2+ biosorption by aerobic granules. Biochem. Eng. J. 35, 174–182 (2007)
Prola, L.D.T.; Machado, F.M.; Bergmann, C.P.; de Souza, F.E.; Gally, C.R.; Lima, E.C.; Adebayo, M.A.; Dias, S.L.P.; Calvete, T.: Adsorption of Direct Blue 53 dye from aqueous solutions by multi-walled carbon nanotubes and activated carbon. J. Environ. Manag. 130, 166–175 (2013)
Saucier, C.; Karthickeyan, P.; Ranjithkumar, V.; Lima, E.C.; dos Reis, G.S.; de Brum, I.A.S.: Efficient removal of amoxicillin and paracetamol from aqueous solutions using magnetic activated carbon. Environ. Sci. Pollut. Res. 24, 5918–5932 (2017)
Long, F.; Gong, J.L.; Zeng, G.M.; Chen, L.; Wang, X.Y.; Deng, J.H.; Niu, Q.Y.; Zhang, H.Y.; Zhang, X.R.: Removal of phosphate from aqueous solution by magnetic Fe–Zr binary oxide. Chem. Eng. J. 171, 448–455 (2011)
Namduri, H.; Nasrazadani, S.: Quantitative analysis of iron oxides using Fourier transform infrared spectrophotometry. Corros. Sci. 50, 2493–2497 (2008)
Le Toullec, M.; Simmons, C.J.; Simmons, J.H.: Infrared spectroscopic studies of the hydrolysis reaction during leaching of heavy-metal fluoride glasses. J. Am. Cream. Soc. 71, 219–224 (1988)
Dou, X.; Mohan, D.; Pittman Jr., C.U.; Yang, S.: Remediating fluoride from water using hydrous zirconium oxide. Chem. Eng. J. 198, 236–245 (2012)
Dong, L.; Jiao, F.; Qin, W.; Zhu, H.; Jia, W.: Activation effect of lead ions on scheelite flotation: adsorption mechanism, AFM imaging and adsorption model. Sep. Purif. Technol. 209, 955–963 (2019)
Zhang, Q.; Zhu, H.; Yang, B.; Jia, F.; Yan, H.; Zeng, M.; Qu, H.: Effect of Pb2+ on the flotation of molybdenite in the presence of sulfide ion. Results Phys. 14, 102361 (2019)
Nguyen, T.T.; Nguyen, T.N.T.; Bach, L.G.; Nguyen, D.T.; Bui, T.P.Q.: Adsorptive removal of Pb(II) using exfoliated graphite adsorbent: influence of experimental conditions and magnetic CoFe2O4 decoration. IIUM Eng. J. 20, 202–215 (2019)
Sag, Y.; Kutsal, T.: Determination of the biosorption heats of heavy metal ions on Zoogloea ramigera and Rhizopus arrhizus. Biochem. Eng. J. 6, 145–151 (2000)
Hayward, D.O.; Trapnell, B.M.W.: Chemisorption, 2nd edn. Butterworth, London (1964)
Liu, Yu: Is the free energy change of adsorption correctly calculated? J. Chem. Eng. Data 54, 1981–1985 (2009)
Acknowledgements
The corresponding author is highly thankful to the ‘University Grant Commission, New Delhi’, for ‘Rajiv Gandhi National Fellowship (RGNF)’ (Letter Number F1-17.1/2016-17/RGNF-2015-17-SC-UTT-15953). The authors (SRA, AK and AGS) extend their gratitude to the Deanship of Scientific Research at King Khalid University for funding the present research work through the Research groups program under Grant Number R.G.P. 2/36/40.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ali, S.R., Kalam, A., Al-Sehemi, A.G. et al. Comparative Adsorption of Pb2+ on Nanostructured Iron–Zirconium Oxide with Fe-to-Zr Molar Ratio of 1:1 and 1:2: Thermodynamic and Kinetic Studies. Arab J Sci Eng 46, 287–300 (2021). https://doi.org/10.1007/s13369-020-04715-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13369-020-04715-z