Abstract
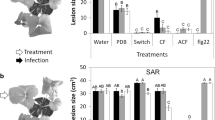
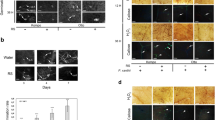
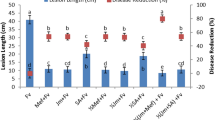
Development of a sustainable agriculture is a current trend searching for alternative and more environmentally friendly strategies to control crop diseases. The necrotrophic fungus Botrytis cinerea, the causal agent of grey mould, affects crops pre- and post-harvest and cause huge economical losses. Microorganisms and microbially-derived elicitors are known to induce plant defences. Here, we study two fungal extracts derived from Colletotrichum acutatum (isolate M11). The crude extract, called CF (culture filtrate) and the semi-purified fraction ACF (axenic culture filtrate) consist in a blend of fungal-derived elicitors. Their activities were evaluated in the plant-pathogen system Arabidopsis thaliana-B. cinerea. Both CF and ACF induced local and systemic resistance, rendering A. thaliana resistant against grey mould disease. The extracts showed no antifungal activity since no inhibition of mycelium growth, changes in hyphal ultrastructure or damage in cell membrane permeability were observed. These findings would indicate that the protective effect was indirect and it was found that CF and ACF induced up-regulation of the defence-related genes PR1, WRKY70 and ERF6, as well as giving local callose deposition in treated leaves. Both fungal extracts have the potential to be used as a main component of a biologically based disease control product with activity against grey mould, with the advantage that the purification process of CF is simpler than for ACF.







Similar content being viewed by others
References
AbuQamar, S., Chen, X., Dhawan, R., Bluhm, B., Salmeron, J., Lam, S., Dietrich, S. A., & Mengiste, T. (2006). Expression profiling and mutant analysis reveals complex regulatory networks involved in Arabidopsis response to Botrytis infection. Plant Journal, 48, 28–44.
Baccelli, I., Lombardi, L., Luti, S., Bernardi, R., Picciarelli, P., Scala, A., & Pazzagli, L. (2014). Cerato-platanin induces resistance in Arabidopsis leaves through stomatal perception, overexpression of salicylic acid- and ethylene-signalling genes and camalexin biosynthesis. PLoS One, 9(6), e100959.
Breen, S., Williams, S. J., Outram, M., Kobe, B., & Solomon, P. S. (2017). Emerging insights into the functions of pathogenesis-related protein 1. Trends in Plant Science, 22, 871–879.
Caro, M. d. P., Holton, N., Conti, G., Venturuzzi, A. L., Martínez-Zamora, M. G., Zipfel, C., Asurmendi, S., & Díaz-Ricci, J. C. (2020). The fungal subtilase AsES elicits a PTI-like defence response in Arabidopsis thaliana plants independently of its enzymatic activity. Molecular Plant Pathology, 21, 147–159. https://doi.org/10.1111/mpp.12881.
Chalfoun, N. R., Grellet, C. F., Martinez-Zamora, M. G., Diaz-Perales, A., Castagnaro, A. P., & Díaz-Ricci, J. C. (2013). Purification and characterization of AsES protein: a subtilisin secreted by Acremonium strictum is a novel plant defense elicitor. Journal of Biological Chemistry, 288, 14098–14113.
Chalfoun, N. R., Durman, S. B., Budeguer, F., Caro, M. P., Bertani, R. P., Di Peto, P., Stenglein, S. A., Filippone, M. P., Moretti, E. R., Díaz-Ricci, J. C., Welin, B., & Castagnaro, A. P. (2018a). Development of PSP1, a biostimulant based on the elicitor AsES for disease management in monocot and dicot crops. Frontiers in Plant Science, 9, 844.
Chalfoun, N. R., Durman, S. B., González-Montaner, J., Reznikov, S., De Lisi, V., González, V., Moretti, E. R., Devani, M. R., Ploper, L. D., Castagnaro, A. P., & Welin, B. (2018b). Elicitor-based biostimulant PSP1 protects soybean against late season diseases in field trials. Frontiers in Plant Science, 9, 763.
Chen, Z., Wang, J., Li, Y., Zhong, Y., Liao, J., Lu, S., Wang, L., Wang, X., & Chen, S. (2018). Dry mycelium of Penicillium chrysogenum activates defense via gene regulation of salicylic acid and jasmonic acid signaling in Arabidopsis. Physiological and Molecular Plant Pathology., 103, 54–61.
Claverie, J., Balacey, S., Lemaître-Guillier, C., Daphnée Brulé, D., Chiltz, A., Granet, L., Noirot, E., Daire, X., Darblade, B., Héloir, M. C., & Poinssot, B. (2018). The cell wall-derived Xyloglucan is a new DAMP triggering plant immunity in Vitis vinifera and Arabidopsis thaliana. Frontiers in Plant Science., 9, 1725.
Dean, R., Van Kan, J. A., Pretorius, Z. A., Hammond-Kosack, K. E., Di Pietro, A., Spanu, P. D., Rudd, J. J., Dickman, M., Kahmann, R., Ellis, J., & Foster, G. D. (2012). The top 10 fungal pathogens in molecular plant pathology. Molecular Plant Pathology, 13(4), 414–430.
Di Rienzo, J. A., Casanoves, F., Balzarini, M. G., Gonzalez, L., Tablada, M., & Robledo, C. W. (2013). InfoStat versión 2013. Resource document. Grupo InfoStat, FCA, Universidad Nacional de Córdoba, Argentina. http://www.infostat.com.ar
Feng, H., Xia, W., Shan, C., Zhou, T., Cai, W., & Zhang, W. (2015). Quaternized chitosan oligomers as novel elicitors inducing protection against B. cinerea in Arabidopsis. International Journal of Biological Macromolecules., 72, 364–369.
fgStatistics (2009). Resource document. In: Di Rienzo J. A., editor. pp. Facultad de Ciencias Agropecuarias, Universidad Nacional de Córdoba, Argentina. https://sites.google.com/site/fgstatistics/
Frías, M., González, C., & Brito, N. (2011). BcSpl1, a cerato-platanin family protein, contributes to Botrytis cinerea virulence and elicits the hypersensitive response in the host. The New Phytologist, 192(2), 483–495.
Frías, M., Brito, N., & González, C. (2013). The Botrytis cinerea cerato-platanin BcSpl1 is a potent inducer of systemic acquired resistance (SAR) in tobacco and generates a wave of salicylic acid expanding from the site of application. Molecular Plant Pathology, 14(2), 191–196.
Glazebrook, J. (2005). Contrasting mechanisms of defense against biotrophic and necrotrophic pathogens. Annual Review of Phytopathology, 43, 205–227.
Granado, J., Felix, G., & Boller, T. (1995). Perception of fungal sterols in plants (Subnanomolar concentrations of Ergosterol elicit extracellular Alkalinization in tomato cells). Plant Physiology, 107, 485–490.
Grellet-Bournonville, C. F., Martinez-Zamora, M. G., Castagnaro, A. P., & Diaz-Ricci, J. C. (2012). Temporal accumulation of salicylic acid activates the defense response against Colletotrichum in strawberry. Plant Physiology and Biochemistry, 54, 10–16.
Hael Conrad, V., Abou-Mansour, E., Diaz-Ricci, J. C., Métraux, J. P., & Serrano, M. (2015). The novel elicitor AsES triggers a defense response against Botrytis cinerea in Arabidopsis thaliana. Plant Science, 241, 120–127.
Hael Conrad, V., Perato, S. M., Arias, M. E., Martinez-Zamora, M. G., Di Peto, P. L. A., Martos, G. G., Castagnaro, A. P., Díaz-Ricci, J. C., & Chalfoun, N. R. (2017). The elicitor protein AsES induces a systemic acquired resistance response accompanied by systemic microbursts and micro-HRs in Fragaria ananassa. Molecular Plant-Microbe Interactions, 31(1), 46–60.
Henry, G., Thonart, P., & Ongena, M. (2012). PAMPs, MAMPs, DAMPs and others: an update on the diversity of plant immunity elicitors. Biotechnology, Agronomy, Society and Environment, 16(2), 257–268.
Jacobs, A. K., Volker, L., Burton, R. A., Panstruga, R., Strizhov, N., Schulze-Lefert, P., & Fincher, G. B. (2003). An Arabidopsis callose synthase, GSL5, is required for wound and papillary callose formation. The Plant Cell., 15(11), 2503–2513.
Klemptner, R. L., Sherwood, J. S., Tugizimana, F., Dubery, I. A., & Piater, L. A. (2014). Ergosterol as a MAMP. Molecular Plant Pathology, 15(7), 747–761.
Koornneef, A., & Pieterse, C. M. J. (2009). Cross talk in defense signaling. Plant Physiology, 146, 839–844.
Laquitaine, L., Gomes, E., François, J., Marchive, C., Pascal, S., Hamdi, S., Rossitza, A., Serge, D., & Pierre, C. T. (2006). Molecular basis of ergosterol-induced protection of grape against Botrytis cinerea: induction of type I LTP promoter activity, WRKY, and stilbene synthase gene expression. Molecular Plant-Microbe Interactions., 19(10), 1103–1112.
Li, J., Brader, G., & Palva, E. T. (2004). The WRKY70 transcription factor: a node of convergence for jasmonate-mediated and salicylate-mediated signals in plant defense. The Plant Cell, 16, 319–331.
Maffei, M. E., Arimura, G., & Mithofer, A. (2012). Natural elicitors, effectors and modulators of plant responses. Natural Products Reports., 29(11), 1288–1303.
Meng, X., Xu, J., He, Y., Yang, K. Y., Mordorski, B., Liu, Y., & Zhang, S. (2013). Phosphorylation of an ERF transcription factor by Arabidopsis MPK3/MPK6 regulates plant defense gene induction and fungal resistance. The Plant Cell, 25(3), 1126–1142.
Moffat, C. S., Ingle, R. A., Wathugala, D. L., Saunders, N. J., Knight, H., & Knight, M. R. (2012). ERF5 and ERF6 play redundant roles as positive regulators of JA/Et-mediated defense against Botrytis cinerea in Arabidopsis. PLoS One, 7(4), e35995.
Monjil, M. S., Nozawa, T., Shibata, Y., Takemoto, D., Ojika, M., & Kawakita, K. (2015). Methanol extract of mycelia from Phytophthora infestans-induced resistance in potato. Comptes Rendus Biologies., 338(3), 185–196.
Nie, P., Li, X., Wang, S., Guo, J., Zhao, H., & Niu, D. (2017). Induced systemic resistance against Botrytis cinerea by Bacillus cereus AR156 through a JA/ET- and NPR1-dependent signaling pathway and activates PAMP-triggered immunity in Arabidopsis. Frontiers in Plant Science., 8, 238.
Olmedo, M. G., González, M. M., Cabrerizo, F. M., Rapisarda, V. A., & Volentini, S. I. (2017). Antifungal activity of β-carbolines on Penicillium digitatum and Botrytis cinerea. Food Microbiology, 62, 9–14.
Perato, S. M., Martínez-Zamora, M. G., Salazar, S. M., & Díaz-Ricci, J. C. (2018). The elicitor AsES stimulates ethylene synthesis, induce ripening and enhance protection against disease naturally produced in avocado fruit. Scientia Horticulturae., 240, 288–292.
Pfaffl, M. W. (2001). A new mathematical model for relative quantification in real- time RT-PCR. Nucleic Acid Research., 29, e45.
Phukan, U. J., Jeena, G. S., & Shukla, R. K. (2016). WRKY transcription factors: molecular regulation and stress responses in plants. Frontiers in Plant Science., 7, 760.
Pieterse, C. M. J., Leon-Reyes, A., Van der Ent, S., & Van Wees, S. C. M. (2009). Networking by small-molecule hormones in plant immunity. Nature Chemistry Biology., 5, 308–316.
Riechmann, J. L., & Meyerowitz, E. M. (1998). The AP2/EREBP family of plant transcription factors. Biological Chemistry, 379, 633–646.
Ruijter, J. M., Ramakers, C., Hoogaars, W. M., Karlen, Y., Bakker, O., van den Hoff, M. J., & Moorman, A. F. (2009). Amplification efficiency: linking baseline and bias in the analysis of quantitative PCR data. Nucleic Acids Research., 37(6), e45.
Ryals, J. A., Neuenschwander, U. H., Willits, M. G., Molina, A., Steiner, H. Y., & Hunt, M. D. (1996). Systemic acquired resistance. The Plant Cell, 8(10), 1809–1819.
Salazar, S., Castagnaro, A., Arias, M., Chalfoun, N. R., Tonello, U., & Diaz-Ricci, J. C. (2007). Induction of a defense response in strawberry mediated by an avirulent strain of Colletotrichum. European Journal of Plant Pathology., 117(2), 109–122.
Schneider, C. A., Rasband, W. S., & Eliceiri, K. W. (2012). NIH image to ImageJ: 25 years of image analysis. Nature Methods., 671, 671–675. https://doi.org/10.1038/nmeth.2089.
Sewelam, N., Kazan, K., & Schenk, P. M. (2016). Global plant stress signaling: reactive oxygen species at the cross-road. Frontiers in Plant Science., 7, 187.
Stegmann, M., Monaghan, J., Smakowska-Luzan, E., Rovenich, H., Lehner, A., Holton, N., Belkhadir, Y., & Zipfel, C. (2017). The receptor kinase FER is a RALF-regulated scaffold controlling plant immune signaling. Science., 355(6322), 287–289.
Thomma, B. P., Penninckx, I. A., Broekaert, W. F., & Cammue, B. P. (2001). The complexity of disease signaling in Arabidopsis. Current Opinion in Immunology., 13(1), 63–68.
Thuerig, B., Felix, G., Binder, A., Boller, T., & Tamm, L. (2005). An extract of Penicillium chrysogenum elicits early defense-related responses and induces resistance in Arabidopsis thaliana independently of known signalling pathways. Physiological and Molecular Plant Pathology., 67(3–5), 180–193.
Trotel-Aziz, P., Couderchet, M., Vernet, G., & Aziz, A. (2006). Chitosan stimulates defense reactions in grapevine leaves and inhibits development of Botrytis cinerea. European Journal of Plant Pathology., 114(4), 405–413.
Trouvelot, S., Héloir, M. C., Poinssot, B., Gauthier, A., Paris, F., Guillier, C., Combier, M., Trdá, L., Daire, X., & Adrian, M. (2014). Carbohydrates in plant immunity and plant protection: roles and potential application as foliar sprays. Frontiers in Plant Science., 5, 592.
UNDP. (2015). Institutional arrangements. United Nations Development Programme. http://www.undp.org/content/undp/en/home/ourwork/capacitybuilding/drivers_of_change/institut_arrangemt
Vatsa, P., Chiltz, A., Luini, E., Vandelle, E., Pugin, A., & Roblin, G. (2011). Cytosolic calcium rises and related events in ergosterol-treated Nicotiana cells. Plant Physiology Biochemistry., 49(7), 764–773.
Veronese, P., Nakagami, H., Bluhm, B., AbuQamar, S., Chen, X., Salmeron, J., Dietrich, R. A., Hirt, H., & Mengiste, T. (2006). The membrane-anchored BOTRYTIS-INDUCEDKINASE1 plays distinct roles in Arabidopsis resistance to necrotrophic and biotrophic pathogens. The Plant Cell, 18, 257–273.
Wiesel, L., Newton, A. C., Elliott, I., Booty, D., Gilroy, E. M., Birch, P. R. J., & Hein, I. (2014). Molecular effects of resistance elicitors from biological origin and their potential for crop protection. Frontiers in Plant Science., 5, 655.
Yang, Y., Zhang, H., Li, G., Li, W., Wang, X., & Song, F. (2009). Ectopic expression of MgSM1, a Cerato-platanin family protein from Magnaporthe grisea, confers broad-spectrum disease resistance in Arabidopsis. Plant Biotechnology Journal., 7(8), 763–777.
Zhang, Y., Yang, X., Zeng, H., Guo, L., Yuan, J., & Qiu, D. (2014). Fungal elicitor protein PebC1 from Botrytis cinerea improves disease resistance in Arabidopsis thaliana. Biotechnology Letters, 36(5), 1069–1078.
Zhang, Y., Gao, Y., Liang, Y., Dong, Y., Yang, X., Yuan, J., & Qiu, D. (2017). The Verticillium dahliae SnodProt1-like protein VdCP1 contributes to virulence and triggers the plant immune system. Frontiers in Plant Science., 8, 1880.
Zhong, Y., Peng, J., Chen, Z., Xie, H., Luo, D., Dai, J. R., Yan, F., Wang, J. G., Dong, H. Z., & Chen, S. Y. (2015). Dry mycelium of Penicillium chrysogenum activates defense responses and restricts the spread of tobacco mosaic virus in tobacco. Physiological and Molecular Plant Pathology., 103, 54–61.
Acknowledgements
This paper was partially supported with grants of the Universidad Nacional de Tucumán (PIUNT 26/D642), Agencia Nacional de Promoción Científica y Tecnológica (PICT 2017-0653), CONICET (PUE-2016-0104) and INTA (PNHFA 1106073 and TUSGO 1231101). VHC is postdoctoral CONICET fellow, RHTG and FJRS are Ph.D. CONICET fellows, SM and JCDR are CONICET researchers, and SMS is INTA researcher.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The manuscript has not been published and is not under consideration for publication elsewhere. All authors have approved the manuscript and agreed to submit the paper to the European Journal of Plant Pathology. The research was conducted in the absence of any commercial relationships that could be considered as a potential conflict of interest.
Rights and permissions
About this article
Cite this article
Hael Conrad, V., Tomas Grau, R.H., Moschen, S.N. et al. Fungal-derived extracts induce resistance against Botrytis cinerea in Arabidopsis thaliana. Eur J Plant Pathol 158, 45–58 (2020). https://doi.org/10.1007/s10658-020-02054-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-020-02054-1