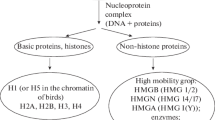
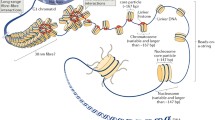
Abstract—In the first part of this review (Biophysics, 63, 858 (2018)), the structure of H1 family linker histones, their posttranslational modifications, as well as the role of H1 histone in the formation of compact transcriptionally inactive chromatin, were considered. The second part is devoted to the role of H1 family linker histones in the structural organization of chromatin at different levels: from nucleosomes to metaphase chromosomes. The mechanisms of interaction of H1 histone with other elements of chromatin, including with DNA and nuclear proteins, are discussed.



Similar content being viewed by others
REFERENCES
A. E. White, A. R. Hieb, and K. Luger, Sci. Rep. 6, 19122 (2016).
C. L. Woodcock and R. P. Ghosh, Cold Spring Harb. Perspect. Biol. 2 (5), a000296 (2010).
K. Luger, M. L. Dechassa, and D. J. Tremethick, Nat. Rev. Mol. Cell Biol. 13, 436 (2012).
T. L. Caterino and J. J. Hayes, Biochem. Cell Biol. 89, 35 (2011).
E. Chikhirzhina, G. Chikhirzhina, and A. Polyanichko, Biomed. Spectr. Imag. 3, 345 (2014).
E. V. Chikhirzhina and V. I. Vorob’ev, Tsitologiya 44, 721 (2002).
S. A. Grigoryev, G. Arya, S. Correll, et al., Proc. Natl. Acad. Sci. U. S. A. 106, 13317 (2009).
A. Kalashnikova, M. Porter-Goff, U. Muthurajan, et al., J. Roy. Soc. Interface 10, 20121022 (2013).
J. Ausio, Bioessays 37, 46 (2015).
A. Sadakierska-Chudy and M. Filip, Neurotox. Res. 27, 172 (2015).
A. V. Ilatovskii, D. V. Lebedev, M. V. Filatov, et al., Tsitologiya 54 (4), 298 (2012)
A. L. Olins and D. E. Olins, Science 184, 868 (1974).
H. Schiessel, J. Widom, R. F. Bruinsma, et al., Phys. Rev. Lett. 86, 4414 (2001).
A. M. Polyanichko and E. V. Chikhirzhina, J. Mol. Struct. 1044, 167 (2013).
K. Luger, A. W. Mader, R. K. Richmond, et al., Nature 389, 251 (1997).
R. K. Suto, R. S. Edayathumangalam, C. L. White, et al., J. Mol. Biol. 326, 371 (2003).
T. Schalch, S. Duda, D. F. Sargent, et al., Nature 436, 138 (2005).
A. Klug, Annu. Rev. Biochem. 79, 1 (2010).
A. Polyanichko and H. Wieser, Biopolymers 78, 329 (2005).
A. Polyanichko, E. Chikhirzhina, V. Andruschchenko, et al., Biopolymers 83, 182 (2006).
A. Polyanichko and H. Wieser, in Methods in Protein Structure and Stability Analysis: Vibrational Spectroscopy, ed. by E. Permyakov and V. Uversky (Nova Science Publ., New York, 2007), pp. 267–302.
A. Polyanichko and E. Chikhirzhina, Spectroscopy 27, 393 (2012).
A. M. Polyanichko, V. V. Andrushchenko, P. Bour, et al., Circular Dichroism: Theory and Spectroscopy, ed. by D. S. Rodgers (Nova Science Publ., New York, 2012), pp. 67–126.
A. M. Polyanichko, V. I. Vorob’ev, and E. V. Chikhirzhina, Mol. Biol. (Moscow) 47 (2), 412 (2013).
J. Zlatanova and J. Yaneva, DNA Cell Biol. 10, 239 (1991).
E. V. Chikhirzhina, T. Yu. Starkova, E. I. Kostyleva, et al., Tsitologiya 53 (10), 826 (2011).
E. I. Ramm, E. V. Chikhirzhina, E. I. Kostyleva, et al., Biokhimiya 60 (1), 150 (1995).
E. V. CHikhirzhina, E. I. Kostyleva, E. I. Ramm et al., Tsitologiya 40 (10), 883 (1998).
A. M. Polyanichko, S. G. Davydenko, E. V. Chikhirzhina, et al., Tsitologiya 42 (8), 787 (2000).
E. V. Chikhirzhina, A. M. Polyanichko, A. N. Skvortsov, et al., Mol. Biol. (Moscow) 36 (3), 525 (2002).
A. M. Polyanichko, E. V. Chikhirzhina, A. N. Skvortsov, et al., J. Biomol. Struct. Dyn. 19, 1053 (2002).
E. Chikhirzhina, T. Starkova, E. Kostyleva, et al., Spectroscopy: Int. J. 27, 433 (2012).
E. V. Chikhirzhina, A. M. Polyanichko, E. I. Kostyleva, et al., Mol. Biol. (Moscow) 45 (2), 318 (2011).
A. V. Fonin, O. V. Stepanenko, K. K. Turoverov, et al., Tsitologiya 52 (11), 946 (2010).
A. Fonin, O. V. Stepanenko, I. M. Kuznetsova, et al., Spectroscopy 24, 165 (2010).
E. Chikhirzhina, T. Starkova, and A. Polyanichko, Biophysics (Moscow) 63 (6), 858 (2018).
J. B. Baldwin, P. G. Boseley, E. M. Bradbury, et al., Nature 253, 245 (1975).
R. D. Kornberg, Science 184, 868 (1974).
C. L. Peterson and M. A. Laniel, Curr. Biol. 14, R546 (2004).
C. A. Davey, D. F. Sargent, K. Luger, et al., J. Mol. Bio-l. 319, 1097 (2002).
S. Bilokapic, M. Strause, and M. Halic, Nat. Struct. Mol. Biol. 25, 101 (2018).
S. Bilokapic, M. Strause, and M. Halic, Nat. Commun. 9, 1330 (2018).
S. Bilokapic, M. Strauss, and M. Halic, Sci. Rep. 8, 7046 (2018).
G. Arents, R. W. Burlingame, B. C. Wang, et al., Proc. Natl. Acad. Sci. U. S. A. 88, 10148 (1991).
J. M. Harp, B. L. Hanson, D. E. Timm, et al., Acta Cryst. D: Biol. Cryst. 56, 1513 (2000).
C. M. Wood, J. M. Nicholson, S. J. Lambert, et al., Acta Cryst. F: Struct. Biol. Cryst. Comm. 61, 541 (2005).
A. Jerzmanowski, in Chromatin Struture and Dynamics: State-of-the-Art, Ed. by J. Zlatanova and S. H. Leuba (Elsevier, London, 2004), pp. 75–102.
B. Sarg, W. Helliger, H. Talasz, et al., J. Biol. Chem. 281, 6573 (2006).
A. Kowalski and J. Palyga, Cell Biol. Int. 36, 981 (2012).
J. Ocampo, F. Cui, V. B. Zhurkin, and D. J. Clark, Nucleus 7, 382 (2016).
X. Lu and J. C. Hansen, J. Biol. Chem. 279, 8701 (2004).
C. Crane-Robinson, Biochim. Biophys. Acta 1859, 431 (2016).
D. J. Tremethick, Cell 128, 651 (2007).
G. Li and D. Reinberg, Curr. Opin. Genet. Dev. 21, 175 (2011).
D. Angelov, J. Vitolo, V. Mutskov, et al., Proc. Natl. Acad. Sci. U. S. A. 98, 6599 (2001).
B. R. Zhou, J. Jiang, H. Feng, et al., Mol. Cell 59, 628 (2015).
B. R. Zhou, H. Feng, R. Ghirlando, et al., J. Mol. Biol. 428, 3948 (2016).
J. Bednar, I. Garcia-Saez, R. Boopathi, et al., Mol. Cell, 66, 384 (2017).
K. Maeshima, R. Imai, T. Hikima, et al., Methods 70, 154 (2014).
A. A. Kalashnikova, D. D. Winkler, S. J. McBryant, et al., Nucleic Acids Res. 41, 4026 (2013).
P. Zhu and G. Li, IUBMB Life 68, 873 (2016).
D. V. Fyodorov, B. R. Zhou, A. I. Skoultchi, et al., Nat. Rev. Mol. Cell Biol. 19, 192 (2018).
P. J. Robinson and D. Rhodes, Curr. Opin. Struct. Biol. 16, 336 (2006).
P. J. Robinson, L. Fairall, A. T. Van Huynh, et al., Proc. Natl. Acad. Sci. U. S. A. 103, 6506 (2006).
V. Andrushchenko, J. H. van de Sande, and H. Wieser, Biopolymers 72, 374 (2003).
A. Polyanichko and E. Chikhirzhina, Adv. Biomed. Spectrosc. 7, 185 (2013).
E. Chikhirzhina, T. Starkova, E. Kostyleva et al., Adv. Biomed. Spectrosc. 7, 177 (2013).
T. Maniatis, J. H. Venable, Jr., and L. S. Lerman, J. Mol. Biol. 84, 37 (1974).
A. L. Turner, M. Watson, O. G. Wilkins, et al., Proc. Natl. Acad. Sci. U. S. A., 115 (47), 11964 (2018).
F. Totsingan and A. J. Bell, Jr., Prot. Sci. 22, 1552 (2013).
J. Zlatanova and K. van Holde, BioEssays 20, 584 (1998).
J. N. Yaneva, E. G. Paneva, S. I. Zacharieva et al., Z. Naturforsch. C 61, 879 (2006).
J. N. Yaneva, E. G. Paneva, S. I. Zacharieva et al., Z. Naturforsch. C 62, 905 (2007).
K. P. Nightingale, D. Pruss, and A. P. Wolffe, J. Biol. Chem. 271, 7090 (1996).
X. Lu and J. C. Hansen, Biochem. Cell Biol. 81, 173 (2003).
X. Lu, B. Hamkalo, M. H. Parseghian, et al., Biochemistry 48, 164 (2009).
A. A. Kalashnikova, R. A. Rogge, and J. C. Hansen, Biochim. Biophys. Acta 1859, 455 (2016).
A. Roque, I. Ponte, and P. Suau, Chromosoma 126, 83 (2017).
A. Roque, I. Ponte, and P. Suau, Biochim. Biophys. Acta 1859, 444 (2016).
A. Roque, I. Ponte, J. L. Arrondo, et al., Nucleic Acids Res. 36, 4719 (2008).
H. E. Kasinsky, J. D. Lewis, J. B. Dacks, et al., FASEB J. 15, 34 (2001).
J. C. Hansen, X. Lu, E. D. Ross, et al., J. Biol. Chem. 281, 1853 (2006).
J. Allan, D. Ram, N. Harborne, et al., J. Cell. Biol. 98, 1320 (1984).
B. R. Zhou, H. Feng, H. Kato, et al., Proc. Natl. Acad. Sci. U. S. A. 110, 19390 (2013).
M. J. Hendzel, M. A. Lever, E. Crawford, et al., J. Biol. Chem. 279, 20028 (2004).
N. Raghuram, G. Carrero, J. Th’ng, et al., Biochem. Cell Biol. 87, 189 (2009).
J. P. Th’ng, R. Sung, M. Ye, et al., J. Biol. Chem. 280, 27809 (2005).
H. Fang, D. J. Clark, and J. J. Hayes, Nucleic Acids Res. 40, 1475 (2012).
J. Allan, P. G. Hartman, C. Crane-Robinson, et al., Nature 288, 675 (1980).
Y. Zhou, S. Gershman, V. Ramakrishnan, et al., Nature 395, 402 (1998).
S. Lambert, S. Muyldermans, J. Baldwin, et al., Biochem. Biophys. Res. Commun. 179, 810 (1991).
J. Hayes, Biochemistry 35, 11931 (1996).
D. Pruss, B. Bartholomew, J. Persinger, et al., Science 274, 614 (1996).
D. T. Brown, T. Izard, and T. Misteli, Nat. Struct. Mol. Biol. 13, 250 (2006).
L. Fan and V. A. Roberts, Proc. Natl. Acad. Sci. U. S. A. 103, 8384 (2006).
S. J. McBryant, X. Lu, and J. C. Hansen, Cell Res. 20, 519 (2010).
J.-Q. Ni, L.-P. Liu, D. Hess, et al., Genes Dev. 20, 1959 (2006).
H. J. Szerlong, J. A. Herman, C. M. Krause et al., J. Mol. Biol. 427, 2056 (2015).
J. Zlatanova, Trends Biochem. Sci. 15, 273 (1990).
H. Lee, R. Habas, and C. Abate-Shen, Science 304, 1675 (2004).
P. J. Laybourn and J. T. Kadonaga, Science 254, 238 (1991).
Q. Lin, A. Inselman, X. Han, et al., J. Biol. Chem. 279, 23525 (2004).
Y. Zheng, S. John, J. J. Pesavento, et al., J. Cell Biol. 189, 407 (2010).
S. Yang, B. J. Kim, L. N. Toro, et al., Proc. Natl. Acad. Sci. U. S. A. 110, 1708 (2013).
Y. V. Postnikov, L. Trieschmann, A. Rickers, et al., J. Mol. Biol. 252, 423 (1995).
Y. Postnikov and M. Bustin, Biochim. Biophys. Acta 1799, 62 (2010).
H. Kato, H. van Ingen, B. R. Zhou, et al., Proc. Natl. Acad. Sci. U. S. A. 108, 12283 (2011).
R. D. Phair, P. Scaffidi, C. Elbi, et al., Mol. Cell. Biol. 24, 6393 (2004).
Y. V. Postnikov, V. V. Shick, A. V. Belyavsky, et al., Nucleic Acids Res. 19, 717 (1991).
T. Deng, Z. I. Zhu, S. Zhang, et al., Mol. Cell. Biol. 33, 3377 (2013).
H. F. Ding, M. Bustin, and U. Hansen, Mol. Cell. Biol. 17, 5843(1997).
K. J. Murphy, A. R. Cutter, H. Fang, et al., Nucleic Acids Res. 45, 9917 (2017).
M. Stros, Biochim. Biophys. Acta 1799, 101 (2010).
M. Stros, E. Polanska, M. Kucirek, et al., PLoS One 10, e0138774 (2015).
R. Reeves, DNA Repair 36, 122 (2015).
F. A. Atcha, A. Syed, B. Wu, et al., Mol Cell Biol. 27, 8352 (2007).
L. H. Pevny and S. K. Nicolis, Int. J. Biochem. Cell Biol. 42, 421 (2010).
P. Bernard and V. R. Harley, Int. J. Biochem. Cell Biol. 42, 400 (2010).
F. Oppel, N. Muller, G. Schackert, et al., Mol Cancer. 10, 137(2011).
O. Leis, A. Eguiara, E. Lopez-Arribillaga, et al., Oncogene 31, 1354 (2012).
T. Chi, Nat. Rev. Immunol. 4, 965 (2004).
M. Stros, D. Launholt, and K. D. Grasser, Cell. Mol. Life Sci. 64, 2590 (2007).
D. Lai, M. Wan, J. Wu, et al., Proc. Natl. Acad. Sci. U. S. A. 106, 1169 (2009).
R. Catena, E. Escoffier, C. Caron, et al., Biol. Reprod. 80, 358 (2009).
S. Park and S. J. Lippard, Biochemistry 51, 6728 (2012).
G. J. Sullivan and B. McStay, Nucleic Acids Res. 26, 3555 (1998).
V. Ramakrishnan, Annu. Rev. Biophys. Biomol. Struct. 26, 83 (1997).
Y. V. Postnikov and M. Bustin, Biochim. Biophys. Acta 1859, 462 (2016).
A. M. Polyanichko, Z. V. Leonenko, D. Kramb, et al., Biophysics (Moscow) 53 (3), 202 (2008).
L. Cato, K. Stott, M. Watson, et al., Mol. Biol. 384, 1262 (2008).
A. Polyanichko and H. Wieser, Spectroscopy 24, 239 (2010).
L. A. Kohlstaedt, E. C. Sung, A. Fujishige, et al., J. Biol. Chem. 262, 524 (1987).
L. A. Kohlstaedt and R. D. Cole, Biochemistry 33, 570 (1994).
A. M. Polyanichko, B. A. Dribinskii, I. B. Kipenko, et al., Strukt. Dinam. Mol. Sistem 8A, 3 (2010).
E. Polanska, S. Pospisilova, and M. Stros, PLoS One 9, e89070 (2014).
P. Widlak, M. Kalinowska, M. H. Parseghian, et al., Biochemistry 44, 7871 (2005).
S. W. Harshman, N. L.Young, M. R. Parthun, et al., Nucleic Acids Res. 41, 9593 (2013).
E. Cheung, A. S. Zarifyan, and W. L. Kraus, Mol. Cell. Biol. 22, 2463 (2002).
R. D. Phair and T. Misteli, Nature 404, 604 (2000).
M. Harrer, H. Luhrs, M. Bustin, et al., J. Cell Sci. 117, 3459 (2004).
F. Catez, H. Yang, K. J. Tracey, et al., Mol. Cell. Biol. 24, 4321 (2004).
A. Allahverdi, R. Yang, N. Korolev, et al., Nucleic Acids Res. 39, 1680 (2011).
P. Trojer, J. Zhang, M. Yonezawa, et al., J. Biol. Chem. 284, 8395 (2009).
T. K. Hale, A. Contreras, A. J. Morrison, et al., Mol. Cell 22, 693 (2006).
M. Brehove, T. Wang, J. North, et al., J. Biol. Chem. 290, 22612 (2015).
R. Morra, T. Fessl, Y. Wang, et al., Methods Mol. Biol. 1431, 175 (2016).
I. H. McColl, E. W. Blanch, A. C. Gill, et al., J. Am. Chem. Soc. 125, 10019 (2003).
F. Zhu, N. W. Isaacs, L. Hecht, et al., Structure 13, 1409 (2005).
L. D. Barron, Biomed. Spectr. Imag. 4, 223 (2015).
M. L. Mello and B. C. Vidal, PLoS One 7, e43169 (2012).
H. B. Stuhrmann, Acta Crystallogr. A 64, 181 (2008).
Y. Joti, T. Hikima, Y. Nishino, et al., Nucleus 3, 404 (2012).
I. Garcia-Saez, H. Menoni, R. Boopathi, et al., Mol. Cell 72, 1 (2018).
M. A. Ozturk, V. Cojocaru, and R. C. Wade, Structure 26, 1 (2018).
M. A. Ozturk, V. Cojocaru, and R. C. Wade, Biophys. J. 114, 2363 (2018).
B. R. Zhou, J. Jiang, R. Ghirlando, et al., J. Mol. Biol. 430, 3093 (2018).
M. A. Ozturk, G. V. Pachov, R. C. Wade, et al., Nucleic Acids Res. 44, 6599 (2016).
J. C. Hansen, Ann. Rev. Biophys. Biomol. Struct. 31, 361 (2002).
F. Song, P. Chen, D. Sun, et al., Science 344, 376 (2014).
C. L. Woodcock, J. Cell Biol. 125, 11 (1994).
C. Kizilyaprak, D. Spehner, D. Devys, et al., PLoS One 5, e11039 (2010).
M. P. Scheffer, M. Eltsov, and A. S. Frangakis, Proc. Natl. Acad. Sci. U. S. A. 108, 16992 (2011).
M. Eltsov, K. M. Maclellan, K. Maeshima et al., Proc. Natl. Acad. Sci. U. S. A. 105, 19732 (2008).
E. Fussner, M. Strauss, U. Djuric, et al., EMBO Rep. 13, 992 (2012).
H. D. Ou, S. Phan, T. J. Deerinck, et al., Science 357, pii: eaag0025 (2017).
T. Nozaki, R. Imai, M. Tanbo, et al., Mol. Cell 67, 282 (2017).
M. A. Ricci, C. Manzo, M. F. Garcı’a-Parajo, et al., Cell 160, 1145 (2015).
K. Maeshima, R. Rogge, S. Tamura, et al., EMBO J. 35, 1115 (2016).
K. Maeshima, R. Imai, S. Tamura, et al., Chromosoma 123, 225 (2014).
S. A. Grigoryev, G. Bascom, J. M. Buckwalter, et al., Proc. Natl. Acad. Sci. U. S. A. 113, 1238 (2016).
W. Li, P. Chen, J. Yu, et al., Mol Cell 64, 120 (2016).
A. Bancaud, S. Huet, N. Daigle, et al., EMBO J. 28, 3785 (2009).
A. Routh, S. Sandin, and D. Rhodes, Proc. Natl. Acad. Sci. U. S. A. 105, 8872 (2008).
Funding
This work was supported by the Russian Foundation for Basic Research (grant no. 18-08-01500 (structural studies of DNA–protein interactions) and grant no. 18-04-01199 (studying the role of nuclear proteins HMGB1/2, H1 in the structural organization of chromatin).
Author information
Authors and Affiliations
Contributions
All of the authors made equal contributions to preparing this manuscript.
Corresponding authors
Ethics declarations
The authors declare that they have no conflict of interest.
This article does not contain any studies involving animals or human participants performed by any of the authors.
Additional information
Translated by A. Barkhash
Rights and permissions
About this article
Cite this article
Chikhirzhina, E.V., Starkova, T.Y. & Polyanichko, A.M. The Role of Linker Histones in Chromatin Structural Organization. 2. Interaction with DNA and Nuclear Proteins. BIOPHYSICS 65, 202–212 (2020). https://doi.org/10.1134/S0006350920020049
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006350920020049