Abstract
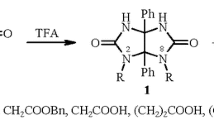
New derivatives of aminouracilindolone have been prepared and their structure has been studied. The reaction of 2-chloro-1H-indole-3-carbaldehyde and its N-substituted analogs with 6-aminouracil has not led to the derivatives of uracilpyrimidinindole but has been accompanied by the chlorine atom substitution with a hydroxyl group, followed by the formation of an earlier unknown aminouracilindolone. Structure of one of the obtained compounds has been established by single-crystal X-ray diffraction analysis.




Similar content being viewed by others
REFERENCES
Corey, E.J., Czako, B., and Kurti, L., Molecules and Medicine, Hoboken: Wiley, 2007. https://doi.org/10.1002/bmb.20179
Pozharskii, A.F., Soldatenkov, A.T., and Katritzky, A.R., Heterocycles in Life and Society: an Introduction to Heterocyclic Chemistry, Biochemistry and Applications, Chichester: Wiley, 2011. https://doi.org/10.1002/9781119998372
Foote, K.M., Blades, K., Cronin, A., Fillery, S., Guichard, S.S., Hassall, L., Hickson, I., Jacq, X., Jewsbury, P.J., and Mc-Guire, T.M., J. Med. Chem., 2013, vol. 56, p. 2125. https://doi.org/10.1021/jm301859s
Kondreddi, R.R., Jiricek, J., Rao, S.P., Lakshminarayana, S.B., Camacho, L.R., Rao, R., Herve, M., Bifani, P., Ma, N.L., Kuhen, K., Goh, A., Chatterjee, A.K., Dick, T., Diagana, T.T., Manjunatha, U.H., and Smith, P.W., J. Med. Chem., 2013, vol. 56, p. 8849. https://doi.org/10.1021/jm4012774
Yeung, K.S., Qiu, Z., Xue, Q., Fang, H., Yang, Z., Zadjura, L., D’Arienzo, C., Eggers, B.J., Riccardi, K., Shi, P.Y., Gong, Y.F., Browning, M.R., Gao, Q., Hansel, S., Santone, K., Lin, P.F, Meanwell, N.A., and Kadow, J.F., Bioorg. Med. Chem. Lett., 2013, vol. 23, p. 198. https://doi.org/10.1016/j.bmcl.2012.10.115
Fatahala, S., Khedr, M.A., and Mohamed, M.S., Acta. Chim. Slov., 2017, vol. 64, p. 865. https://doi.org/10.17344/acsi.2017.3481
Silveira, C.C., Mendes, S.R., Soares, J.R., Martinez, D.M., and Savegnago, L., Tetrahedron Lett., 2013, vol. 54, p. 4926. doi.org/10.1016/j.tetlet.2013.07.004
Santillan, A.Jr., McClure, K.J., Allison, B.D., Lord, B., Boggs, J.D., Morton, K.L., Everson, A.M., Nepomuceno, D., Letavic, M.A., Lee-Dutra, A., Lovenberg, T.W., Carruthers, N.I., and and Grice, C.A., Bioorg. Med. Chem. Lett., 2010, vol. 20, p. 6226. https://doi.org/10.1016/j.bmcl.2010.08.103
Murinov, Yu.I., Grabovskii, S.A., and Kabal’nova, N.N., Russ. Chem. Bull., 2019, vol. 68, no. 5, p. 946. https://doi.org/10.1007/s11172-019-2505-4
Kundu, N.G., Das, P., Balzarini, J., and De Clercq, E., Bioorg. Med. Chem., 1997, vol. 5, p. 2011. https://doi.org/10.1016/S0968-0896(97)00114-4
Gazivoda, T., Raić-Malić, S., Marjanović, M., Kralj, M., Pavelić, K., Balzarini, J., De Clercq, E., and Mintas, M., Bioorg. Med. Chem., 2007, vol. 15, p. 749. https://doi.org/10.1016/j.bmc.2006.10.046
Design of Hybrid Molecules for Drug Development, Decker, M., Ed., Amsterdam: Elsevier, 2017.
Suzdalev, K.F., Babakova, M.N., Kartsev, V.G., and Krasnov, K.A., Heterocycles, 2015, vol. 91, no. 1, p. 64. https://doi.org/10.3987/COM-14-13135
Suzdalev, K.F., Vikrischuk, N.I., Prikhodko, K.A., Shasheva, E.Yu., Kurbatov, S.V., Bogus, S.K., and GalenkoYaroshevsky, P.A., Сhem. Heterocycl. Compd., 2016, vol. 52, p. 303. https://doi.org/10.1007/s10593-016-1882-y
Sheldrick, G.M., SHELXTL, Bruker AXS Inc., Madison, Wisconsin, USA, 2000.
Funding
This study was performed in the scope of the State Task (no. 0089-2019-0013, V.V. Tkachev, G.V. Shilov). The experiments were performed using the equipment of the Center for Collective Usage “Molecular Spectroscopy” of Southern Federal University and the Educational-Research Laboratory of Resonance Spectroscopy, Department of Chemistry of Natural and High-Molecular Compounds, Southern Federal University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
No conflict of interest was declared by the authors.
Rights and permissions
About this article
Cite this article
Vikrishchuk, N.I., Tkachev, V.V., Popov, L.D. et al. Synthesis and Structure of New Aminouracilindolones. Russ J Gen Chem 90, 799–803 (2020). https://doi.org/10.1134/S1070363220050060
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363220050060