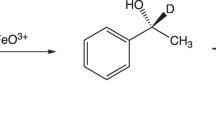
Abstract
AR-A014418, 1-(4-methoxybenzyl)-3-(5-nitrothiazol-2-yl)urea is a potential drug with potent activities against cancer cell, nociceptive pain and neurodegenerative disorders. In this paper, the stable deuterium-labeled [2H3]AR-A014418 was synthesized in seven steps with 21% overall yield starting from the commercially available [2H4]methanol, p-hydroxybenzaldehyde and 2-amino-5-nitrothiazole. The chemical and isotope purity of the title compound were over 99% and 98%, respectively. This labeled [2H3]AR-A014418 can be used as an internal standard for LC–MS analysis in drug absorption, distribution and other pharmacokinetics studies.
Graphic abstract




Similar content being viewed by others
References
Bhat R, Xue Y, Berg S, Hellberg S, Ormö M, Nilsson Y, Radesäter AC, Jerning E, Markgren PO, Borgegård T, Nylöf M, Giménez-Cassina A, Hernández F, Lucas JJ, Díaz-Nido J, Avila J (2003) Structural insights and biological effects of glycogen synthase kinase 3-specific inhibitor AR-A014418. J Biol Chem 278:45937–45945
Carter YM, Kunnimalaiyaan S, Chen H, Gamblin TC, Kunnimalaiyaan M (2014) Specific glycogen synthase kinase-3 inhibition reduces neuroendocrine markers and suppresses neuroblastoma cell growth. Cancer Biol Ther 15:510–515
Koh SH, Kim Y, Kim HY, Hwang S, Lee CH, Kim SH (2007) Inhibition of glycogen synthase kinase-3 suppresses the onset of symptoms and disease progression of G93A-SOD1 mouse model of ALS. Exp Neurol 205:336–346
Martins DF, Rosa AO, Gadotti VM, Mazzardo-Martins L, Nascimento FP, Egea J, López MG, Santos ARS (2011) The antinociceptive effects of AR-A014418, a selective inhibitor of glycogen synthase kinase-3 beta, in mice. J Pain 12:315–322
Kunnimalaiyaan S, Gamblin TC, Kunnimalaiyaan M (2015) Glycogen synthase kinase-3 inhibitor AR-A014418 suppresses pancreatic cancer cell growth via inhibition of GSK-3-mediated Notch1 expression. HPB (Oxford) 17:770–776
Yadav AK, Vashishta V, Joshi N, Taneja P (2014) AR-A 014418 used against GSK3beta downregulates expression of hnRNPA1 and SF2/ASF splicing factors. J Oncol 2014:695325
Bliss-Moreau M, Coarfa C, Xiao W, Gunaratne P, Krett NL, Rosen ST (2013) Synergy of small-molecule inhibitors in cutaneous T-cell lymphoma cells: a discovery tool to define new therapeutic targets in T-cell receptor (TCR) signaling pathways. Blood 122:4327
Eichelbaum M, von Unruh GE, Somogyi A (1982) Application of stable labelled drugs in clinical pharmacokinetic investigations. Clin Pharmacokinet 7:490–507
Sturkenboom MGG, van der Lijke H, Jongedijk EM, Kok WT, Greijdanus B, Uges DRA, Alffenaar JWC (2015) Quantification of isoniazid, pyrazinamide and ethambutol in serum using liquid chromatography-tandem mass spectrometry. J Appl Bioanal 1:89–98
Barrish A, Olah TV, Gatto GJ, Michel KB, Dobrinska MR, Gilbert JD (2015) The use of stable isotope labelling and liquid chromatography/tandem mass spectrometry techniques to study the pharmacokinetics and bioavailability of the antimigraine drug, MK-0462 (rizatriptan) in dogs. Rapid Commun Mass Spectrom 10:1033–1037
Liang DW, Zhang M, Tan L, Wang YQ (2019) Synthesis of piroxicam-d3. Food Drug 21:120–123 (in Chinese)
Monte FL, Kramer T, Boländer A, Plotkin B, Eldar-Finkelman H, Fuertes A, Dominguez J, Schmidt B (2011) Synthesis and biological evaluation of glycogen synthase kinase 3 (GSK-3) inhibitors: an fast and atom efficient access to 1-aryl-3-benzylureas. Bioorg Med Chem Lett 21:5610–5615
Vasdev N, Garcia A, Stableford WT, Young AB, Meyer JH, Houle S, Wilson AA (2005) Synthesis and ex vivo evaluation of carbon-11 labeled N-(4-methoxybenzyl)-N′-(5-nitro-1,3-thiazol-2-yl)urea ([11C]AR-A014418): a radiolabelled glycogen synthase kinase-3b specific inhibitor for PET studies. Bioorg Med Chem Lett 15:5270–5273
Hicks JW, Wilson AA, Rubie EA, Woodgett JR, Houle S (2012) Towards the preparation of radiolabeled 1-aryl-3-benzyl ureas: radiosynthesis of [11C-carbonyl] AR-A014418 by [11C]CO2 fixation. Bioorg Med Chem Lett 22:2099–2101
Bhatt MV, Kulkarni SU (1983) Cleavage of ethers. Synthesis 1983:249–282
Learmonth DA, Alves PC (2002) Improved method for demethylation of nitro-catechol methyl ethers. Synth Commun 32:641–649
Szostak M, Sautier B, Spain M, Procter DJ (2014) Electron transfer reduction of nitriles using SmI2-Et3N-H2O: synthetic utility and mechanism. Org Lett 16:1092–1095
Thurow S, Lenardão EJ, Just-Baringo X, Procter DJ (2017) Reduction of selenoamides to amines using SmI2-H2O. Org Lett 19:50–53
Duspara PA, Islam MS, Lough AJ, Batey RA (2012) Synthesis and reactivity of N-alkyl carbamoylimidazoles: development of N-methyl carbamoylimidazole as a methyl isocyanate equivalent. J Org Chem 77:10362–10368
Acknowledgements
We are grateful for financial support from the Applied Basic Research and Development Programs of the Science and Technology Foundation of Ya’an (2017YYJSKF15).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liang, D., Wang, Y., Yan, T. et al. Synthesis of deuterium-labeled 1-(4-methoxybenzyl)-3-(5-nitrothiazol-2-yl)urea (AR-A014418) as an internal standard for LC–MS analysis. J Radioanal Nucl Chem 325, 217–222 (2020). https://doi.org/10.1007/s10967-020-07227-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-020-07227-0