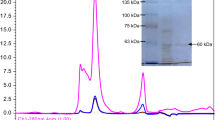
Abstract
Photolyases are flavoproteins that repair ultraviolet-induced DNA lesions (cyclobutane pyrimidine dimer or CPD, and pyrimidine (6-4) pyrimidone photoproducts or (6-4)-PPs), using blue light as an energy source. These enzymes are substrate specific, meaning that a specific photolyase repairs either a CPD or a (6-4)-PP. In this work, we produced a class II CPD-photolyase (called as PhrSph98) from the Antarctic bacterium Sphingomonas sp. UV9 by recombinant DNA technology and we purified the enzyme using immobilized metal affinity chromatography. By using an immunochemistry assay, with monoclonal antibodies against CPD and (6-4)-PP, we found that PhrSph98 repairs both DNA lesions. The result was confirmed by immunocytochemistry using immortalized non-tumorigenic human keratinocytes. Results from structure prediction, pocket computation, and molecular docking analyses showed that PhrSph98 has the two expected protein domains (light-harvesting antenna and a catalytic domain), a larger catalytic site as compared with photolyases produced by mesophilic organisms, and that both substrates fit the catalytic domain. The results obtained from predicted homology modeling suggest that the electron transfer pathway may occur following this pathway: Y389-W369-W390-F376-W381/FAD. The evolutionary reconstruction of PhrSph98 suggests that this is a missing link that reflects the transition of (6-4)-PP repair into the CPD repair ability for the class II CPD-photolyases. To the best of our knowledge, this is the first report of a naturally occurring bifunctional, CPD and (6-4)-PP, repairing enzyme.
Key points
• We report the first described bifunctional CPD/(6-4)-photoproducts repairing enzyme. The bifunctional enzyme reaches the nuclei of keratinocyte and repairs the UV-induced DNA damage. The enzyme should be a missing link from an evolutionary point of view. The enzyme may have potential uses in the pharmaceutical and cosmetic industries.








Similar content being viewed by others
References
Almagro Armenteros JJ, Tsirigos KD, Sønderby CK, Petersen TN, Winther O, Brunak S, von Heijne G, Nielsen H (2019) SignalP 5.0 improves signal peptide predictions using deep neural networks. Nat Biotechnol. https://doi.org/10.1038/s41587-019-0036-z
Boukamp P, Petrussevska RT, Breitkreutz D, Hornung J, Markham A, Fusenig NE (1988) Normal keratinization in a spontaneously immortalized aneuploid human keratinocyte cell line. J Cell Biol 106:761–771. https://doi.org/10.1083/jcb.106.3.761
Brameier M, Krings A, MacCallum RM (2007) NucPred—predicting nuclear localization of proteins. Bioinformatics. 23:1159–1160. https://doi.org/10.1093/bioinformatics/btm066
Cadet J, Richard Wagner J (2013) DNA base damage by reactive oxygen species, oxidizing agents, and UV radiation. Cold Spring Harb Perspect Biol 5. https://doi.org/10.1101/cshperspect.a012559
Coesel S, Mangogna M, Ishikawa T, Heijde M, Rogato A, Finazzi G, Todo T, Bowler C, Falciatore A (2009) Diatom PtCPF1 is a new cryptochrome/photolyase family member with DNA repair and transcription regulation activity. EMBO Rep 10:655–661. https://doi.org/10.1038/embor.2009.59
D’Amico S, Claverie P, Collins T, Georlette D, Gratia E, Hoyoux A, Meuwis MA, Feller G, Gerday C, Marahiel MA, Russell R, Warren G, Davies PL (2002) Molecular basis of cold adaptation. In: Philosophical Transactions of the Royal Society B: Biological Sciences
Dikbas UM, Tardu M, Canturk A, Gul S, Ozcelik G, Baris I, Ozturk N, Kavakli IH (2019) Identification and characterization of a new class of (6-4) photolyase from Vibrio cholerae. Biochemistry. 58:4352–4360. https://doi.org/10.1021/acs.biochem.9b00766
Essen LO, Klar T (2006) Light-driven DNA repair by photolyases. Cell Mol Life Sci 63:1266–1277
Franz S, Ignatz E, Wenzel S, Zielosko H, Ngurah Putu EPG, Maestre-Reyna M, Tsai MD, Yamamoto J, Mittag M, Essen LO (2018) Structure of the bifunctional cryptochrome aCRY from Chlamydomonas reinhardtii. Nucleic Acids Res 46:8010–8022. https://doi.org/10.1093/nar/gky621
Fullana N, Braña V, José Marizcurrena J, Morales D, Betton J-M, Marín M, Castro-Sowinski S (2017) Identification, recombinant production and partial biochemical characterization of an extracellular cold-active serine-metalloprotease from an Antarctic Pseudomonas isolate. AIMS Bioeng 4:386–401. https://doi.org/10.3934/bioeng.2017.3.386
Hancock JM, Zvelebil MJ, Zvelebil MJ (2004) UniProt. In: Dictionary of Bioinformatics and Computational Biology
Hitomi K, Arvai AS, Yamamoto J, Hitomi C, Teranishi M, Hirouchi T, Yamamoto K, Iwai S, Tainer JA, Hidema J, Getzoff ED (2012) Eukaryotic class II cyclobutane pyrimidine dimer photolyase structure reveals basis for improved ultraviolet tolerance in plants. J Biol Chem 287:12060–12069. https://doi.org/10.1074/jbc.M111.244020
Holub D, Lamparter T, Elstner M, Gillet N (2019) Biological relevance of charge transfer branching pathways in photolyases. Phys Chem Chem Phys 21:17072–17081. https://doi.org/10.1039/c9cp01609k
Johnson JL, Lowell BC, Ryabinina OP, Lloyd RS, McCullough AK (2011) TAT-mediated delivery of a DNA repair enzyme to skin cells rapidly initiates repair of UV-induced DNA damage. J Invest Dermatol 131:753–761. https://doi.org/10.1038/jid.2010.300
Kiontke S, Geisselbrecht Y, Pokorny R, Carell T, Batschauer A, Essen LO (2011) Crystal structures of an archaeal class II DNA photolyase and its complex with UV-damaged duplex DNA. EMBO J 30:4437–4449. https://doi.org/10.1038/emboj.2011.313
Kiontke S, Gnau P, Haselsberger R, Batschauer A, Essen LO (2014) Structural and evolutionary aspects of antenna chromophore usage by class II photolyases. J Biol Chem 289:19659–19669. https://doi.org/10.1074/jbc.M113.542431
Kleiner O, Butenandt J, Carell T, Batschauer A (1999) Class II DNA photolyase from Arabidopsis thaliana contains FAD as a cofactor. Eur J Biochem 264:161–167. https://doi.org/10.1046/j.1432-1327.1999.00590.x
Kosugi S, Hasebe M, Entani T, Takayama S, Tomita M, Yanagawa H (2008) Design of peptide inhibitors for the importin α/β nuclear import pathway by activity-based profiling. Chem Biol 15:940–949. https://doi.org/10.1016/j.chembiol.2008.07.019
Kosugi S, Hasebe M, Matsumura N, Takashima H, Miyamoto-Sato E, Tomita M, Yanagawa H (2009a) Six classes of nuclear localization signals specific to different binding grooves of importin α. J Biol Chem 284:478–485. https://doi.org/10.1074/jbc.M807017200
Kosugi S, Hasebe M, Tomita M, Yanagawa H (2009b) Systematic identification of cell cycle-dependent yeast nucleocytoplasmic shuttling proteins by prediction of composite motifs. Proc Natl Acad Sci U S A 106:10171–10176. https://doi.org/10.1073/pnas.0900604106
Kulms D, Zeise E, Pöppelmann B, Schwarz T (2002) DNA damage, death receptor activation and reactive oxygen species contribute to ultraviolet radiation-induced apoptosis in an essential and independent way. Oncogene. 21:5844–5851. https://doi.org/10.1038/sj.onc.1205743
Kumar S, Stecher G, Tamura K (2016) MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol Biol Evol. https://doi.org/10.1093/molbev/msw054
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 227:680–685. https://doi.org/10.1038/227680a0
Larkin MA, Blackshields G, Brown NP, Chenna R, Mcgettigan PA, McWilliam H, Valentin F, Wallace IM, Wilm A, Lopez R, Thompson JD, Gibson TJ, Higgins DG (2007) Clustal W and Clustal X version 2.0. Bioinformatics. https://doi.org/10.1093/bioinformatics/btm404
Ma H, Holub D, Gillet N, Kaeser G, Thoulass K, Elstner M, Krauß N, Lamparter T (2019) Two aspartate residues close to the lesion binding site of Agrobacterium (6-4) photolyase are required for Mg2+ stimulation of DNA repair. FEBS J 286:1765–1779. https://doi.org/10.1111/febs.14770
Marizcurrena JJ, Morel MA, Braña V, Morales D, Martinez-López W, Castro-Sowinski S (2017) Searching for novel photolyases in UVC-resistant Antarctic bacteria. Extremophiles 21:409–418. https://doi.org/10.1007/s00792-016-0914-y
Marizcurrena JJ, Morales D, Smircich P, Castro-Sowinski S (2019) Draft genome sequence of the UV-resistant Antarctic bacterium Sphingomonas sp. strain UV9. Microbiol Resour Announc. https://doi.org/10.1128/mra.01651-18
Masson F, Laino T, Rothlisberger U, Hutter J (2009) A QM/MM investigation of thymine dimer radical anion splitting catalyzed by DNA photolyase. ChemPhysChem 10:400–410. https://doi.org/10.1002/cphc.200800624
Müller P, Yamamoto J, Martin R, Iwai S, Brettel K (2015) Discovery and functional analysis of a 4th electron-transferring tryptophan conserved exclusively in animal cryptochromes and (6-4) photolyases. Chem Commun 51:15502–15505. https://doi.org/10.1039/c5cc06276d
Oberpichler I, Pierik AJ, Wesslowski J, Pokorny R, Rosen R, Vugman M, Zhang F, Neubauer O, Ron EZ, Batschauer A, Lamparter T (2011) A photolyase-like protein from Agrobacterium tumefaciens with an iron-sulfur cluster. PLoS One 6:e26775. https://doi.org/10.1371/journal.pone.0026775
Oliveri P, Fortunato AE, Petrone L, Ishikawa-Fujiwara T, Kobayashi Y, Todo T, Antonova O, Arboleda E, Zantke J, Tessmar-Raible K, Falciatore A (2014) The cryptochrome/photolyase family in aquatic organisms. Mar Genomics 14:23–37. https://doi.org/10.1016/j.margen.2014.02.001
Ozturk N (2017) Phylogenetic and functional classification of the photolyase/cryptochrome family. Photochem Photobiol 93:104–111
Pérez F, Varela M, Canclini L, Acosta S, Martínez-López W, López GV, Hernández P (2019) Furoxans and tocopherol analogs—furoxan hybrids as anticancer agents. Anti-Cancer Drugs 30:330–338. https://doi.org/10.1097/CAD.0000000000000721
Petersen JL (2001) A gene required for the novel activation of a class II DNA photolyase in Chlamydomonas. Nucleic Acids Res 29:4472–4481. https://doi.org/10.1093/nar/29.21.4472
Robert X, Gouet P (2014) Deciphering key features in protein structures with the new ENDscript server. Nucleic Acids Res 42:W320–W324. https://doi.org/10.1093/nar/gku316
Sambrook J, Russell DW (2001) Molecular cloning: a laboratory manual. Cold Spring Harb Lab Press Cold Spring Harb NY 61:17–18. https://doi.org/10.1016/0092-8674(90)90210-6
Sancar GB, Smith FW, Heelis PF (1987) Purification of the yeast PHR1 photolyase from an Escherichia coli overproducing strain and characterization of the intrinsic chromophores of the enzyme. J Biol Chem
Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T, Preibisch S, Rueden C, Saalfeld S, Schmid B, Tinevez JY, White DJ, Hartenstein V, Eliceiri K, Tomancak P, Cardona A (2012) Fiji: an open-source platform for biological-image analysis. Nat Methods 9:676–682
Schrödinger L (2015) The PyMol Molecular Graphics System, Versión 1.8. Thomas Hold
Selby CP, Sancar A (2006) A cryptochrome/photolyase class of enzymes with single-stranded DNA-specific photolyase activity. Proc Natl Acad Sci U S A 103:17696–17700. https://doi.org/10.1073/pnas.0607993103
Slamovits CH, Keeling PJ (2004) Class II photolyase in a microsporidian intracellular parasite. J Mol Biol 341:713–721. https://doi.org/10.1016/j.jmb.2004.06.032
Steffen C, Thomas K, Huniar U, Hellweg A, Rubner O, Schroer A (2010) AutoDock4 and AutoDockTools4: automated docking with selective receptor flexibility. J Comput Chem. https://doi.org/10.1002/jcc
Steurer B, Turkyilmaz Y, van Toorn M, van Leeuwen W, Escudero-Ferruz P, Marteijn JA (2019) Fluorescently-labelled CPD and 6-4PP photolyases: new tools for live-cell DNA damage quantification and laser-assisted repair. Nucleic Acids Res 47:3536–3549. https://doi.org/10.1093/nar/gkz035
Studier FW (2005) Protein production by auto-induction in high-density shaking cultures. Protein Expr Purif 41:207–234. https://doi.org/10.1016/j.pep.2005.01.016
Tian W, Chen C, Lei X, Zhao J, Liang J (2018) CASTp 3.0: computed atlas of surface topography of proteins. Nucleic Acids Res. https://doi.org/10.1093/nar/gky473
van Oers MM, Lampen MH, Bajek MI, Vlak JM, Eker APM (2008) Active DNA photolyase encoded by a baculovirus from the insect Chrysodeixis chalcites. DNA Repair (Amst) 7:1309–1318. https://doi.org/10.1016/j.dnarep.2008.04.013
Veluchamy S, Rollins JA (2008) A CRY-DASH-type photolyase/cryptochrome from Sclerotinia sclerotiorum mediates minor UV-A-specific effects on development. Fungal Genet Biol 45:1265–1276. https://doi.org/10.1016/j.fgb.2008.06.004
von Zadow A, Ignatz E, Pokorny R, Essen LO, Klug G (2016) Rhodobacter sphaeroides CryB is a bacterial cryptochrome with (6–4) photolyase activity. FEBS J 283:4291–4309. https://doi.org/10.1111/febs.13924
Yamada D, Dokainish HM, Iwata T, Yamamoto J, Ishikawa T, Todo T, Iwai S, Getzoff ED, Kitao A, Kandori H (2016) Functional conversion of CPD and (6-4) photolyases by mutation. Biochemistry. 55:4173–4183. https://doi.org/10.1021/acs.biochem.6b00361
Zhang F, Scheerer P, Oberpichler I, Lamparter T, Krauß N (2013) Crystal structure of a prokaryotic (6-4) photolyase with an Fe-S cluster and a 6,7-dimethyl-8-ribityllumazine antenna chromophore. Proc Natl Acad Sci U S A 110:7217–7222. https://doi.org/10.1073/pnas.1302377110
Zhang M, Wang L, Zhong D (2017) Photolyase: dynamics and mechanisms of repair of sun-induced DNA damage. Photochem Photobiol 93:78–92
Zimmermann L, Stephens A, Nam SZ, Rau D, Kübler J, Lozajic M, Gabler F, Söding J, Lupas AN, Alva V (2018) A completely reimplemented MPI bioinformatics toolkit with a new HHpred server at its core. J Mol Biol 430:2237–2243. https://doi.org/10.1016/j.jmb.2017.12.007
Acknowledgments
The authors thank the Uruguayan Antarctic Institute for the logistic support during the stay in the Antarctic Base Artigas. S. Castro-Sowinski, L. Canclini, and J. J. Marizcurrena are members of the National Research System (SNI, Sistema Nacional de Investigadores).
Funding
This work was partially supported by PEDECIBA (Programa de Desarrollo de las Ciencias Básicas), CSIC (Project C667), ANII (Project FMV_3_2016_1_1226654), Comisón Honoraria de Lucha Contra el Cáncer, and donations by Celsius Laboratory (http://www.celsius.uy/). The work of JJM was supported by ANII and CAP (Comisión Académica de Posgrado, UdelaR). The provisional patent was funded by ANII (Agencia Nacional de Investigación e Innovación, PAT_X_2017_1_140739) & CSIC (Comisión Sectorial de Investigación Científica).
Author information
Authors and Affiliations
Contributions
JJM produced and purified the recombinant photolyase and performed the in vitro DNA photorepair, the structure prediction, pocket computation, and molecular docking experiments. AC, LC, and PH conducted the immunocytochemistry assays. DV conducted the HPLC experiments. TL conducted the evolutionary reconstruction. SCS guided most experiments and wrote the manuscript.
Corresponding author
Ethics declarations
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 362 kb)
Rights and permissions
About this article
Cite this article
Marizcurrena, J.J., Acosta, S., Canclini, L. et al. A natural occurring bifunctional CPD/(6-4)-photolyase from the Antarctic bacterium Sphingomonas sp. UV9. Appl Microbiol Biotechnol 104, 7037–7050 (2020). https://doi.org/10.1007/s00253-020-10734-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-020-10734-5