Abstract
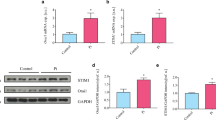
Diabetes and chronic kidney disease (CKD) both trigger vascular osteogenic signaling and calcification leading to early death by cardiovascular events. Osteogenic signaling involves upregulation of the transcription factors CBFA1, MSX2, and SOX9, as well as alkaline phosphatase (ALP), an enzyme fostering calcification by degrading the calcification inhibitor pyrophosphate. In CKD, osteogenic signaling is triggered by hyperphosphatemia, which upregulates the serum and glucocorticoid-inducible kinase SGK1, a strong stimulator of the Ca2+-channel ORAI1. The channel is activated by STIM1 and accomplishes store-operated Ca2+-entry (SOCE). The present study explored whether exposure of human aortic smooth muscle cells (HAoSMCs) to high extracellular glucose concentrations similarly upregulates ORAI1 and/or STIM1 expression, SOCE, and osteogenic signaling. To this end, HAoSMCs were exposed to high extracellular glucose concentrations (15 mM, 24 h) without or with additional exposure to the phosphate donor ß-glycerophosphate. Transcript levels were estimated using qRT-PCR, protein abundance using Western blotting, ALP activity using a colorimetric assay kit, calcium deposits utilizing Alizarin red staining, cytosolic Ca2+-concentration ([Ca2+]i) by Fura-2-fluorescence, and SOCE from increase of [Ca2+]i following re-addition of extracellular Ca2+ after store depletion with thapsigargin (1 μM). As a result, glucose enhanced the transcript levels of SGK1 and ORAI1, ORAI2, and STIM2, protein abundance of ORAI1, SOCE, the transcript levels of CBFA1, MSX2, SOX9, and ALPL, as well as calcium deposits. Moreover, glucose significantly augmented the stimulating effect of ß-glycerophosphate on transcript levels of SGK1 and ORAI1, SOCE, the transcript levels of osteogenic markers, as well as calcium deposits. ORAI1 inhibitor MRS1845 (10 μM) significantly blunted the glucose-induced upregulation of the CBFA1 and MSX2 transcript levels. In conclusion, the hyperglycemia of diabetes stimulates expression of SGK1 and ORAI1, thus, augmenting store-operated Ca2+-entry and osteogenic signaling in HAoSMCs.








Similar content being viewed by others
References
Becchetti A, Arcangeli A (2010) Integrins and ion channels in cell migration: implications for neuronal development, wound healing and metastatic spread. Adv Exp Med Biol 674:107–123
Bergmeier W, Weidinger C, Zee I, Feske S (2013) Emerging roles of store-operated Ca(2)(+) entry through STIM and ORAI proteins in immunity, hemostasis and cancer. Channels (Austin) 7:379–391. https://doi.org/10.4161/chan.24302
Betjes MG (2013) Immune cell dysfunction and inflammation in end-stage renal disease. Nat Rev Nephrol 9:255–265. https://doi.org/10.1038/nrneph.2013.44
Blacher J, Guerin AP, Pannier B, Marchais SJ, London GM (2001) Arterial calcifications, arterial stiffness, and cardiovascular risk in end-stage renal disease. Hypertension 38:938–942
Burgoyne RD (2007) Neuronal calcium sensor proteins: generating diversity in neuronal Ca2+ signalling. Nat Rev Neurosci 8:182–193. https://doi.org/10.1038/nrn2093
Capiod T (2013) The need for calcium channels in cell proliferation. Recent Pat Anticancer Drug Discov 8:4–17
Chaudhari S, Wu P, Wang Y, Ding Y, Yuan J, Begg M, Ma R (2014) High glucose and diabetes enhanced store-operated Ca(2+) entry and increased expression of its signaling proteins in mesangial cells. Am J Phys Renal Phys 306:F1069–F1080. https://doi.org/10.1152/ajprenal.00463.2013
Cohen RA (1993) Dysfunction of vascular endothelium in diabetes mellitus. Circulation 87
Courjaret R, Machaca K (2012) STIM and Orai in cellular proliferation and division. Front Biosci (Elite Ed) 4:331–341
Fadini GP, Pauletto P, Avogaro A, Rattazzi M (2007) The good and the bad in the link between insulin resistance and vascular calcification. Atherosclerosis 193:241–244. https://doi.org/10.1016/j.atherosclerosis.2007.05.015
Fahrner M, Muik M, Derler I, Schindl R, Fritsch R, Frischauf I, Romanin C (2009) Mechanistic view on domains mediating STIM1-Orai coupling. Immunol Rev 231:99–112. https://doi.org/10.1111/j.1600-065X.2009.00815.x
Fleischhacker E, Esenabhalu VE, Spitaler M, Holzmann S, Skrabal F, Koidl B, Kostner GM, Graier WF (1999) Human diabetes is associated with hyperreactivity of vascular smooth muscle cells due to altered subcellular Ca2+ distribution. Diabetes 48:1323–1330. https://doi.org/10.2337/diabetes.48.6.1323
Foley RN, Parfrey PS, Sarnak MJ (1998) Clinical epidemiology of cardiovascular disease in chronic renal disease. Am J Kidney Dis 32:S112–S119
Graier WF, Posch K, Fleischhacker E, Wascher TC, Kostner GM (1999) Increased superoxide anion formation in endothelial cells during hyperglycemia: an adaptive response or initial step of vascular dysfunction? Diabetes Res Clin Pract 45:153–160. https://doi.org/10.1016/s0168-8227(99)00045-5
Graier WF, Posch K, Wascher TC, Kostner GM (1997) Role of superoxide anions in changes of endothelial vasoactive response during acute hyperglycemia. Horm Metab Res 29:622–626. https://doi.org/10.1055/s-2007-979113
Graier WF, Simecek S, Kukovetz WR, Kostner GM (1996) High D-glucose-induced changes in endothelial Ca2+/EDRF signaling are due to generation of superoxide anions. Diabetes 45:1386–1395. https://doi.org/10.2337/diab.45.10.1386
Graier WF, Wascher TC, Lackner L, Toplak H, Krejs GJ, Kukovetz WR (1993) Exposure to elevated D-glucose concentrations modulates vascular endothelial cell vasodilatory response. Diabetes 42:1497–1505. https://doi.org/10.2337/diab.42.10.1497
Haarhaus M, Brandenburg V, Kalantar-Zadeh K, Stenvinkel P, Magnusson P (2017) Alkaline phosphatase: a novel treatment target for cardiovascular disease in CKD. Nat Rev Nephrol 13:429–442. https://doi.org/10.1038/nrneph.2017.60
Hwang DY, Chien SC, Hsu YW, Kao CC, Cheng SY, Lu HC, Wu MS, Chang JM (2014) Genetic polymorphisms of ORAI1 and chronic kidney disease in Taiwanese population. Biomed Res Int 2014:290863–290866. https://doi.org/10.1155/2014/290863
Jha JC, Ho F, Dan C, Jandeleit-Dahm K (2018) A causal link between oxidative stress and inflammation in cardiovascular and renal complications of diabetes. Clin Sci (Lond) 132:1811–1836. https://doi.org/10.1042/CS20171459
Kappel S, Borgstrom A, Stoklosa P, Dorr K, Peinelt C (2019) Store-operated calcium entry in disease: beyond STIM/Orai expression levels. Semin Cell Dev Biol 94:66–73. https://doi.org/10.1016/j.semcdb.2019.01.003
Kapustin AN, Chatrou ML, Drozdov I, Zheng Y, Davidson SM, Soong D, Furmanik M, Sanchis P, De Rosales RT, Alvarez-Hernandez D, Shroff R, Yin X, Muller K, Skepper JN, Mayr M, Reutelingsperger CP, Chester A, Bertazzo S, Schurgers LJ, Shanahan CM (2015) Vascular smooth muscle cell calcification is mediated by regulated exosome secretion. Circ Res 116:1312–1323. https://doi.org/10.1161/CIRCRESAHA.116.305012
Lang F, Bohmer C, Palmada M, Seebohm G, Strutz-Seebohm N, Vallon V (2006) (Patho)physiological significance of the serum- and glucocorticoid-inducible kinase isoforms. Physiol Rev 86:1151–1178. https://doi.org/10.1152/physrev.00050.2005
Lang F, Eylenstein A, Shumilina E (2012) Regulation of Orai1/STIM1 by the kinases SGK1 and AMPK. Cell Calcium 52:347–354. https://doi.org/10.1016/j.ceca.2012.05.005
Lang F, Guelinckx I, Lemetais G, Melander O (2017) Two liters a day keep the doctor away? Considerations on the pathophysiology of suboptimal fluid intake in the common population. Kidney Blood Press Res 42:483–494. https://doi.org/10.1159/000479640
Lang F, Ritz E, Alesutan I, Voelkl J (2014) Impact of aldosterone on osteoinductive signaling and vascular calcification. Nephron Physiol 128:40–45. https://doi.org/10.1159/000368268
Lang F, Ritz E, Voelkl J, Alesutan I (2013) Vascular calcification--is aldosterone a culprit? Nephrol Dial Transplant 28:1080–1084. https://doi.org/10.1093/ndt/gft041
Lang F, Shumilina E (2013) Regulation of ion channels by the serum- and glucocorticoid-inducible kinase SGK1. FASEB J 27:3–12. https://doi.org/10.1096/fj.12-218230
London GM, Guerin AP, Marchais SJ, Metivier F, Pannier B, Adda H (2003) Arterial media calcification in end-stage renal disease: impact on all-cause and cardiovascular mortality. Nephrol Dial Transplant 18:1731–1740
Ma K, Liu P, Al-Maghout T, Sukkar B, Cao H, Voelkl J, Alesutan I, Pieske B, Lang F (2019) Phosphate-induced ORAI1 expression and store-operated Ca(2+) entry in aortic smooth muscle cells. J Mol Med 97:1465–1475. https://doi.org/10.1007/s00109-019-01824-7
Ma X, Chen Z, Wang L, Wang G, Wang Z, Dong X, Wen B, Zhang Z (2018) The pathogenesis of diabetes mellitus by oxidative stress and inflammation: its inhibition by berberine. Front Pharmacol 9:782. https://doi.org/10.3389/fphar.2018.00782
Mizobuchi M, Towler D, Slatopolsky E (2009) Vascular calcification: the killer of patients with chronic kidney disease. J Am Soc Nephrol 20:1453–1464. https://doi.org/10.1681/ASN.2008070692
Moccia F, Dragoni S, Lodola F, Bonetti E, Bottino C, Guerra G, Laforenza U, Rosti V, Tanzi F (2012) Store-dependent Ca(2+) entry in endothelial progenitor cells as a perspective tool to enhance cell-based therapy and adverse tumour vascularization. Curr Med Chem 19:5802–5818
Orrenius S, Zhivotovsky B, Nicotera P (2003) Regulation of cell death: the calcium-apoptosis link. Nat Rev Mol Cell Biol 4:552–565. https://doi.org/10.1038/nrm1150
Peinelt C, Vig M, Koomoa DL, Beck A, Nadler MJ, Koblan-Huberson M, Lis A, Fleig A, Penner R, Kinet JP (2006) Amplification of CRAC current by STIM1 and CRACM1 (Orai1). Nat Cell Biol 8:771–773. https://doi.org/10.1038/ncb1435
Penna A, Demuro A, Yeromin AV, Zhang SL, Safrina O, Parker I, Cahalan MD (2008) The CRAC channel consists of a tetramer formed by Stim-induced dimerization of Orai dimers. Nature 456:116–120. https://doi.org/10.1038/nature07338
Prakriya M, Feske S, Gwack Y, Srikanth S, Rao A, Hogan PG (2006) Orai1 is an essential pore subunit of the CRAC channel. Nature 443:230–233. https://doi.org/10.1038/nature05122
Prevarskaya N, Skryma R, Shuba Y (2011) Calcium in tumour metastasis: new roles for known actors. Nat Rev Cancer 11:609–618. https://doi.org/10.1038/nrc3105
Putney JW Jr (2007) New molecular players in capacitative Ca2+ entry. J Cell Sci 120:1959–1965. https://doi.org/10.1242/jcs.03462
Roderick HL, Cook SJ (2008) Ca2+ signalling checkpoints in cancer: remodelling Ca2+ for cancer cell proliferation and survival. Nat Rev Cancer 8:361–375. https://doi.org/10.1038/nrc2374
Sahu I, Pelzl L, Sukkar B, Fakhri H, Al-Maghout T, Cao H, Hauser S, Gutti R, Gawaz M, Lang F (2017) NFAT5-sensitive Orai1 expression and store-operated Ca(2+) entry in megakaryocytes. FASEB J 31:3439–3448. https://doi.org/10.1096/fj.201601211R
Salter RD, Watkins SC (2009) Dendritic cell altered states: what role for calcium? Immunol Rev 231:278–288. https://doi.org/10.1111/j.1600-065X.2009.00806.x
Samakai E, Hooper R, Soboloff J (2013) The critical role of STIM1-dependent Ca2+ signalling during T-cell development and activation. Int J Biochem Cell Biol 45:2491–2495. https://doi.org/10.1016/j.biocel.2013.07.014
Schmid E, Bhandaru M, Nurbaeva MK, Yang W, Szteyn K, Russo A, Leibrock C, Tyan L, Pearce D, Shumilina E, Lang F (2012) SGK3 regulates Ca(2+) entry and migration of dendritic cells. Cell Physiol Biochem 30:1423–1435. https://doi.org/10.1159/000343330
Schmidt S, Liu G, Liu G, Yang W, Honisch S, Pantelakos S, Stournaras C, Honig A, Lang F (2014) Enhanced Orai1 and STIM1 expression as well as store operated Ca2+ entry in therapy resistant ovary carcinoma cells. Oncotarget 5:4799–4810. https://doi.org/10.18632/oncotarget.2035
Shaw PJ, Feske S (2012) Regulation of lymphocyte function by ORAI and STIM proteins in infection and autoimmunity. J Physiol 590:4157–4167. https://doi.org/10.1113/jphysiol.2012.233221
Smyth JT, Hwang SY, Tomita T, DeHaven WI, Mercer JC, Putney JW (2010) Activation and regulation of store-operated calcium entry. J Cell Mol Med 14:2337–2349. https://doi.org/10.1111/j.1582-4934.2010.01168.x
Steitz SA, Speer MY, Curinga G, Yang HY, Haynes P, Aebersold R, Schinke T, Karsenty G, Giachelli CM (2001) Smooth muscle cell phenotypic transition associated with calcification: upregulation of Cbfa1 and downregulation of smooth muscle lineage markers. Circ Res 89:1147–1154
Stengel B (2010) Chronic kidney disease and cancer: a troubling connection. J Nephrol 23:253–262
Tesfamariam B, Cohen RA (1992) Free radicals mediate endothelial cell dysfunction caused by elevated glucose. Am J Phys 263:H321–H326. https://doi.org/10.1152/ajpheart.1992.263.2.H321
Tong X, Ying J, Pimentel DR, Trucillo M, Adachi T, Cohen RA (2008) High glucose oxidizes SERCA cysteine-674 and prevents inhibition by nitric oxide of smooth muscle cell migration. J Mol Cell Cardiol 44:361–369. https://doi.org/10.1016/j.yjmcc.2007.10.022
Towler DA, Shao JS, Cheng SL, Pingsterhaus JM, Loewy AP (2006) Osteogenic regulation of vascular calcification. Ann N Y Acad Sci 1068:327–333. https://doi.org/10.1196/annals.1346.036
Vig M, Peinelt C, Beck A, Koomoa DL, Rabah D, Koblan-Huberson M, Kraft S, Turner H, Fleig A, Penner R, Kinet JP (2006) CRACM1 is a plasma membrane protein essential for store-operated Ca2+ entry. Science 312:1220–1223. https://doi.org/10.1126/science.1127883
Voelkl J, Luong TT, Tuffaha R, Musculus K, Auer T, Lian X, Daniel C, Zickler D, Boehme B, Sacherer M, Metzler B, Kuhl D, Gollasch M, Amann K, Muller DN, Pieske B, Lang F, Alesutan I (2018) SGK1 induces vascular smooth muscle cell calcification through NF-kappaB signaling. J Clin Invest 128:3024–3040. https://doi.org/10.1172/JCI96477
Waldegger S, Barth P, Raber G, Lang F (1997) Cloning and characterization of a putative human serine/threonine protein kinase transcriptionally modified during anisotonic and isotonic alterations of cell volume. Proc Natl Acad Sci U S A 94:4440–4445. https://doi.org/10.1073/pnas.94.9.4440
Wang Y, Chaudhari S, Ren Y, Ma R (2015) Impairment of hepatic nuclear factor-4alpha binding to the Stim1 promoter contributes to high glucose-induced upregulation of STIM1 expression in glomerular mesangial cells. Am J Phys Renal Phys 308:F1135–F1145. https://doi.org/10.1152/ajprenal.00563.2014
Warr C, Shaw K, Azim A, Piper M, Parsons L (2018) Using Mouse and Drosophila Models to Investigate the Mechanistic Links between Diet, Obesity, Type II Diabetes, and Cancer. 19:4110
Wascher TC, Toplak H, Krejs GJ, Simecek S, Kukovetz WR, Graier WF (1994) Intracellular mechanisms involved in D-glucose-mediated amplification of agonist-induced Ca2+ response and EDRF formation in vascular endothelial cells. Diabetes 43:984–991. https://doi.org/10.2337/diab.43.8.984
Wu P, Ren Y, Ma Y, Wang Y, Jiang H, Chaudhari S, Davis ME, Zuckerman JE, Ma R (2017) Negative regulation of Smad1 pathway and collagen IV expression by store-operated Ca(2+) entry in glomerular mesangial cells. Am J Phys Renal Phys 312:F1090–F1100. https://doi.org/10.1152/ajprenal.00642.2016
Wu P, Wang Y, Davis ME, Zuckerman JE, Chaudhari S, Begg M, Ma R (2015) Store-operated Ca2+ channels in mesangial cells inhibit matrix protein expression. J Am Soc Nephrol 26:2691–2702. https://doi.org/10.1681/ASN.2014090853
Yeromin AV, Zhang SL, Jiang W, Yu Y, Safrina O, Cahalan MD (2006) Molecular identification of the CRAC channel by altered ion selectivity in a mutant of Orai. Nature 443:226–229. https://doi.org/10.1038/nature05108
Yuan T, Yang T, Chen H, Fu D, Hu Y, Wang J, Yuan Q, Yu H, Xu W, Xie X (2019) New insights into oxidative stress and inflammation during diabetes mellitus-accelerated atherosclerosis. Redox Biol 20:247–260. https://doi.org/10.1016/j.redox.2018.09.025
Zhang SL, Kozak JA, Jiang W, Yeromin AV, Chen J, Yu Y, Penna A, Shen W, Chi V, Cahalan MD (2008) Store-dependent and -independent modes regulating Ca2+ release-activated Ca2+ channel activity of human Orai1 and Orai3. J Biol Chem 283:17662–17671. https://doi.org/10.1074/jbc.M801536200
Zhang SL, Yu Y, Roos J, Kozak JA, Deerinck TJ, Ellisman MH, Stauderman KA, Cahalan MD (2005) STIM1 is a Ca2+ sensor that activates CRAC channels and migrates from the Ca2+ store to the plasma membrane. Nature 437:902–905. https://doi.org/10.1038/nature04147
Zhu X, Ma K, Zhou K, Voelkl J, Alesutan I, Leibrock C, Nurnberg B, Lang F (2020) Reversal of phosphate-induced ORAI1 expression, store-operated Ca(2+) entry and osteogenic signaling by MgCl2 in human aortic smooth muscle cells. Biochem Biophys Res Commun 523:18–24. https://doi.org/10.1016/j.bbrc.2019.11.005
Acknowledgments
The authors gratefully acknowledge the meticulous preparation of the manuscript by Lejla Subasic.
Funding
Xuexue Zhu, Kuo Zhou, and Hang Cao are supported by the Chinese Scholarship Council. Bernd Nürnberg is supported by the Deutsche Forschungsgemeinschaft (DFG; NU 53/12–2). The sponsor(s) had no role in study design, the collection, analysis and interpretation of data, in the writing of the report, and in the decision to submit the article for publication.
Author information
Authors and Affiliations
Contributions
KM, BS, XZ, KZ, and HC performed experiments and analyzed data; FL, JV, IA, and BN designed research; FL drafted and wrote the manuscript. All authors corrected and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ma, K., Sukkar, B., Zhu, X. et al. Stimulation of ORAI1 expression, store-operated Ca2+ entry, and osteogenic signaling by high glucose exposure of human aortic smooth muscle cells. Pflugers Arch - Eur J Physiol 472, 1093–1102 (2020). https://doi.org/10.1007/s00424-020-02405-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-020-02405-1