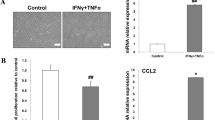
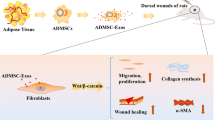
Abstract
Accelerating wound healing is a key consideration for surgeons. The three stages of wound healing include the inflammatory response, cell proliferation and tissue repair, and much research has focused on the migration and proliferation of epidermal cells, since this is one of the most important steps in wound healing. Studies have shown that adipose mesenchymal stem cells (ADSCs) can promote wound healing by releasing exosomes, although the specific mechanism remains unclear. To clarify the role of adipose mesenchymal stem cell exosomes (ADSCs-exo), we constructed a HaCaT cells model and a mouse wound healing model to examine the effects of ADSCs-exo on wound healing. CCK8 assays and the scratch test showed that ADSCs-exo could promote the proliferation and migration of HaCaT cells. Western blotting and real-time PCR showed that ADSCs-exo upregulated the phosphorylation of AKT and the expression of HIF-1α in HaCaT cells. HIF-1α expression was reduced by inhibiting AKT phosphorylation,and the migration of HaCaT cells simultaneously slowed. These results were also confirmed in vivo. In conclusion, we confirmed that ADSCs-exo promote the proliferation and migration of HaCaT cells by regulating the activation of the AKT/HIF-1α signaling pathway, thus promoting wound healing.





Similar content being viewed by others
Change history
02 July 2020
In the original publication of the article, the name of one of the corresponding authors was published incorrectly. The name should be 'Ke Tao' instead of 'Kao Tao'. The corrected author group is given in this Correction.
References
Aarabi S, Longaker MT, Gurtner GC (2007) Hypertrophic scar formation following burns and trauma: new approaches to treatment. PLoS Med 4:e234. https://doi.org/10.1371/journal.pmed.0040234
Bjorge IM, Kim SY, Mano JF, Kalionis B, Chrzanowski W (2017) Extracellular vesicles, exosomes and shedding vesicles in regenerative medicine—a new paradigm for tissue repair. Biomater Sci 6:60–78. https://doi.org/10.1039/c7bm00479f
Borges GA, Elias ST, da Silva SM, Magalhaes PO, Macedo SB, Ribeiro AP, Guerra EN (2017) In vitro evaluation of wound healing and antimicrobial potential of ozone therapy. J Cranio-Maxillo-Facial Surg 45:364–370. https://doi.org/10.1016/j.jcms.2017.01.005
Courtnay R, Ngo DC, Malik N, Ververis K, Tortorella SM, Karagiannis TC (2015) Cancer metabolism and the Warburg effect: the role of HIF-1 and PI3K. Mol Biol Rep 42:841–851. https://doi.org/10.1007/s11033-015-3858-x
Fu X, Li H (2009) Mesenchymal stem cells and skin wound repair and regeneration: possibilities and questions. Cell Tissue Res 335:317–321. https://doi.org/10.1007/s00441-008-0724-3
Goodarzi P et al (2018) Mesenchymal stem cells-derived exosomes for wound regeneration. Adv Exp Med Biol 1119:119–131. https://doi.org/10.1007/5584_2018_251
Goodarzi G, Maniati M, Qujeq D (2019) The role of microRNAs in the healing of diabetic ulcers. Int Wound J 16:621–633. https://doi.org/10.1111/iwj.13070
Gurtner GC, Werner S, Barrandon Y, Longaker MT (2008) Wound repair and regeneration. Nature 453:314–321. https://doi.org/10.1038/nature07039
Hao ZC, Lu J, Wang SZ, Wu H, Zhang YT, Xu SG (2017) Stem cell-derived exosomes: a promising strategy for fracture healing. Cell Prolif. https://doi.org/10.1111/cpr.12359
Hinz N, Jucker M (2019) Distinct functions of AKT isoforms in breast cancer: a comprehensive review. Cell Commun Signal 17:154. https://doi.org/10.1186/s12964-019-0450-3
Hoke GD, Ramos C, Hoke NN, Crossland MC, Shawler LG, Boykin JV (2016) Atypical diabetic foot ulcer keratinocyte protein signaling correlates with impaired wound healing. J Diabetes Res 2016:1586927. https://doi.org/10.1155/2016/1586927
Jin Y et al (2013) MicroRNA-99 family targets AKT/mTOR signaling pathway in dermal wound healing. PLoS ONE 8:e64434. https://doi.org/10.1371/journal.pone.0064434
Kim D, Ku B, Choi EM (2019) Se-methylselenocysteine stimulates migration and antioxidant response in HaCaT keratinocytes: implications for wound healing. J Trace Elem Med Biol 58:126426. https://doi.org/10.1016/j.jtemb.2019.126426
Kosaka N, Yoshioka Y, Hagiwara K, Tominaga N, Katsuda T, Ochiya T (2013) Trash or treasure: extracellular microRNAs and cell-to-cell communication. Front Genet 4:173. https://doi.org/10.3389/fgene.2013.00173
Kucharzewski M, Rojczyk E, Wilemska-Kucharzewska K, Wilk R, Hudecki J, Los MJ (2019) Novel trends in application of stem cells in skin wound healing. Eur J Pharmacol 843:307–315. https://doi.org/10.1016/j.ejphar.2018.12.012
Lai P et al (2018) A potent immunomodulatory role of exosomes derived from mesenchymal stromal cells in preventing cGVHD. J Hematol Oncol 11:135. https://doi.org/10.1186/s13045-018-0680-7
Li H, Fu X (2012) Mechanisms of action of mesenchymal stem cells in cutaneous wound repair and regeneration. Cell Tissue Res 348:371–377. https://doi.org/10.1007/s00441-012-1393-9
Li W, Fan J, Chen M, Woodley DT (2004) Mechanisms of human skin cell motility. Histol Histopathol 19:1311–1324. https://doi.org/10.14670/HH-19.1311
Li X et al (2018) Exosomes from adipose-derived stem cells overexpressing Nrf2 accelerate cutaneous wound healing by promoting vascularization in a diabetic foot ulcer rat model. Exp Mol Med 50:29. https://doi.org/10.1038/s12276-018-0058-5
Maroulakou IG, Oemler W, Naber SP, Klebba I, Kuperwasser C, Tsichlis PN (2008) Distinct roles of the three Akt isoforms in lactogenic differentiation and involution. J Cell Physiol 217:468–477. https://doi.org/10.1002/jcp.21518
Moon KM et al (2012) The effect of secretory factors of adipose-derived stem cells on human keratinocytes. Int J Mol Sci 13:1239–1257. https://doi.org/10.3390/ijms13011239
Nong K et al (2016) Hepatoprotective effect of exosomes from human-induced pluripotent stem cell-derived mesenchymal stromal cells against hepatic ischemia-reperfusion injury in rats. Cytotherapy 18:1548–1559. https://doi.org/10.1016/j.jcyt.2016.08.002
Pratheesh MD et al (2017a) Comparative study on characterization and wound healing potential of goat (Capra hircus) mesenchymal stem cells derived from fetal origin amniotic fluid and adult bone marrow. Res Vet Sci 112:81–88. https://doi.org/10.1016/j.rvsc.2016.12.009
Pratheesh MD et al (2017b) Evaluation of persistence and distribution of intra-dermally administered PKH26 labelled goat bone marrow derived mesenchymal stem cells in cutaneous wound healing model. Cytotechnology 69:841–849 https://doi.org/10.1007/s10616-017-0097-0
Rahmani F et al (2019) Role of regulatory miRNAs of the PI3K/AKT/mTOR signaling in the pathogenesis of hepatocellular carcinoma. J Cell Physiol. https://doi.org/10.1002/jcp.29333
Rani S, Ritter T (2016) The exosome—a naturally secreted nanoparticle and its application to wound healing. Adv Mater 28:5542–5552. https://doi.org/10.1002/adma.201504009
Rani S, Ryan AE, Griffin MD, Ritter T (2015) Mesenchymal stem cell-derived extracellular vesicles: toward cell-free therapeutic applications. Mol Ther 23:812–823. https://doi.org/10.1038/mt.2015.44
Riazifar M, Pone EJ, Lotvall J, Zhao W (2017) Stem cell extracellular vesicles: extended messages of regeneration. Annu Rev Pharmacol Toxicol 57:125–154. https://doi.org/10.1146/annurev-pharmtox-061616-030146
Ruthenborg RJ, Ban JJ, Wazir A, Takeda N, Kim JW (2014) Regulation of wound healing and fibrosis by hypoxia and hypoxia-inducible factor-1. Mol Cells 37:637–643. https://doi.org/10.14348/molcells.2014.0150
Schreml S, Szeimies RM, Prantl L, Landthaler M, Babilas P (2010) Wound healing in the 21st century. J Am Acad Dermatol 63:866–881. https://doi.org/10.1016/j.jaad.2009.10.048
Ti D, Hao H, Fu X, Han W (2016) Mesenchymal stem cells-derived exosomal microRNAs contribute to wound inflammation Science China. Life Sci 59:1305–1312. https://doi.org/10.1007/s11427-016-0240-4
Xiao W, Tang H, Wu M, Liao Y, Li K, Li L, Xu X (2017) Ozone oil promotes wound healing by increasing the migration of fibroblasts via PI3K/Akt/mTOR signaling pathway. Biosci Rep. https://doi.org/10.1042/bsr20170658
Xie Y et al (2019) PI3K/Akt signaling transduction pathway, erythropoiesis and glycolysis in hypoxia (review). Mol Med Rep 19:783–791. https://doi.org/10.3892/mmr.2018.9713
Xuan NT, Toan NL, Mao CV, Vuong NB, Giang NT, Hoang NH (2019) Regulation of dendritic cell function by A20 through high glucose-induced Akt2 signaling Journal of receptor and signal. Transduct Res 39:434–441. https://doi.org/10.1080/10799893.2019.1690511
Yang HL, Tsai YC, Korivi M, Chang CT, Hseu YC (2017) Lucidone promotes the cutaneous wound healing process via activation of the PI3K/AKT Wnt/beta-catenin and NF-kappaB signaling pathways. Biochim Biophysica Acta Mol Cell Res 1864:151–168. https://doi.org/10.1016/j.bbamcr.2016.10.021
Yang C, Luo L, Bai X, Shen K, Liu K, Wang J, Hu D (2020) Highly-expressed micoRNA-21 in adipose derived stem cell exosomes can enhance the migration and proliferation of the HaCaT cells by increasing the MMP-9 expression through the PI3K/AKT pathway. Arch Biochem Biophys 681:108259. https://doi.org/10.1016/j.abb.2020.108259
Zhang J et al (2016) Exosomes derived from human endothelial progenitor cells accelerate cutaneous wound healing by promoting angiogenesis through Erk1/2 signaling. Int J Biol Sci 12:1472–1487. https://doi.org/10.7150/ijbs.15514
Zhang T, Suo C, Zheng C, Zhang H (2019a) Hypoxia and metabolism in metastasis. Adv Exp Med Biol 1136:87–95. https://doi.org/10.1007/978-3-030-12734-3_6
Zhang Y, Yan H, Xu Z, Yang B, Luo P, He Q (2019b) Molecular basis for class side effects associated with PI3K/AKT/mTOR pathway inhibitors. Expert Opin Drug Metab Toxicol 15:767–774. https://doi.org/10.1080/17425255.2019.1663169
Zhao B et al (2018) Exosomal microRNAs derived from human amniotic epithelial cells accelerate wound healing by promoting the proliferation and migration of fibroblasts. Stem cells Int 2018:5420463. https://doi.org/10.1155/2018/5420463
Funding
This study was supported by National Natural Science Foundation of China (No. 81871561).
Author information
Authors and Affiliations
Contributions
YZ, methodology data curation, writing original draft, review and editing. FH, methodology, validation, investigation, writing review & editing, Visualization. LG, writing original draft, formal analysis. PJ, resources, investigation, validation. XKY, resources investigation and validation. ML, investigation and validation. KT, review and editing, funding acquisition. DH. Conceptualization, writing review & editing, Supervision, Project administration.
Corresponding authors
Ethics declarations
Conflict of interest
The author declare that they have no competing interest.
Ethical approval
All mice were conducted in accordance with the National Institutes of Health Guide for the care and use of laboratory animals and approved by the Biological Research Ethics Committee of the Chinese Academy of Sciences.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10735_2020_9887_MOESM1_ESM.tif
Electronic supplementary material 1 Supplementary Figure. Scratch test in HIF-1α silenced HaCaT cells. HaCaT cells were divided into four groups: the control group, HIF-1α siRNA group, exosomes group and exosomes + HIF-1α siRNA group. A. The level of siHIF-1α in HIF-1α silenced HaCaT cells. B-C. The distance from the scratch was measured at 0, 24, and 48h in each group after stimulation and scratch. Bar = 50μm, **p<0.01, n = 6. (TIF 1480 kb)
Rights and permissions
About this article
Cite this article
Zhang, Y., Han, F., Gu, L. et al. Adipose mesenchymal stem cell exosomes promote wound healing through accelerated keratinocyte migration and proliferation by activating the AKT/HIF-1α axis. J Mol Hist 51, 375–383 (2020). https://doi.org/10.1007/s10735-020-09887-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-020-09887-4