Abstract
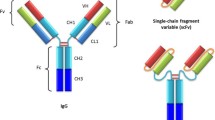
Producing a functional anti-IL-2Rα antibody in Leishmania tarentolae, a trypanosomatid protozoan non-pathogenic to human, is a cost-effective and safe alternative for the production of therapeutic recombinant proteins. Expression cassettes encoding heavy and light chains of the anti-IL-2Rα antibody were cloned into two separate pLEXSY vectors. The plasmids were then used to transfect L. tarentolae laboratory strain p10 and stable expression of a recombinant humanized anti-IL-2Rα antibody was obtained. The heavy and light chains successfully assembled to produce a tetrameric 150 kDa antibody. The antibody was glycosylated and functional as assessed by enzymatic deglycosylation and cell proliferation assays respectively. L. tarentolae can be used as an efficient, cost and labor-effective expression system for the production of therapeutic recombinant proteins.







Similar content being viewed by others
References
Basile G, Peticca M (2009) Recombinant protein expression in Leishmania tarentolae. Mol Biotechnol 43:273–278. https://doi.org/10.1007/s12033-009-9213-5
Ben-Ari ET (2004) Dual purpose: some cancer therapies used to treat autoimmune diseases. J Natl Cancer Inst 96:577–579. https://doi.org/10.1093/jnci/96.8.577
Berting A, Farcet MR, Kreil TR (2010) Virus susceptibility of Chinese hamster ovary (CHO) cells and detection of viral contaminations by adventitious agent testing. Biotechnol Bioeng 106:598–607. https://doi.org/10.1002/bit.22723
Breitling R et al (2002) Non-pathogenic trypanosomatid protozoa as a platform for protein research and production. Protein Expr Purif 25:209–218. https://doi.org/10.1016/S1046-5928(02)00001-3
Church A (2003) Clinical advances in therapies targeting the interleukin-2 receptor. QJM 96:91–102. https://doi.org/10.1093/qjmed/hcg014
Chon JH, Zarbis-Papastoitsis G (2011) Advances in the production and downstream processing of antibodies. New Biotechnol 28:458–463. https://doi.org/10.1016/j.nbt.2011.03.015
Del Val IJ, Polizzi KM, Kontoravdi C (2016) A theoretical estimate for nucleotide sugar demand towards Chinese Hamster Ovary cellular glycosylation. Sci Rep 6:28547. https://doi.org/10.1038/srep28547
De Marco A (2009) Strategies for successful recombinant expression of disulfide bond-dependent proteins in Escherichia coli. Microb Cell Fact 8:26. https://doi.org/10.1186/1475-2859-8-26
Dhara VG, Naik HM, Majewska NI, Betenbaugh MJ (2018) Recombinant antibody production in CHO and NS0 cells: differences and similarities. Biodrugs 32:571–584. https://doi.org/10.1007/s40259-018-0319-9
Fritsche C, Sitz M, Weiland N, Breitling R, Pohl HD (2007) Characterization of the growth behavior of Leishmania tarentolae–a new expression system for recombinant proteins. J Basic Microbiol 47:384–393. https://doi.org/10.1002/jobm.200710111
Hossler P, Khattak SF, Li ZJ (2009) Optimal and consistent protein glycosylation in mammalian cell culture. Glycobiology 19:936–949. https://doi.org/10.1093/glycob/cwp079
Hunter M, Yuan P, Vavilala D, Fox M (2018) Optimization of protein expression in mammalian cells. Curr Protoc Protein Sci 95(1):e77. https://doi.org/10.1002/cpps.77
Iberg CA, Hawiger D (2019) Advancing immunomodulation by in vivo antigen delivery to DEC-205 and other cell surface molecules using recombinant chimeric antibodies. Int Immunopharmacol 73:575–580. https://doi.org/10.1016/j.intimp.2019.05.037
Jefferis R (2005) Glycosylation of recombinant antibody therapeutics. Biotechnol Prog 21:11–16. https://doi.org/10.1021/bp040016jCCC
Jefferis R, Lund J, Pound JD (1998) IgG-Fc-mediated effector functions: molecular definition of interaction sites for effector ligands and the role of glycosylation. Immunol Rev 163:59–76. https://doi.org/10.1111/j.1600-065X.1998.tb01188
Jones SD, Castillo FJ, Levine HL (2007) Advances in the development of therapeutic monoclonal antibodies. BioPharm Int 20:96–114
Jørgensen ML, Friis NA, Just J, Madsen P, Petersen SV, Kristensen P (2014) Expression of single-chain variable fragments fused with the Fc-region of rabbit IgG in Leishmania tarentolae. Microb Cell Fact 13:9. https://doi.org/10.1186/1475-2859-13-9
Kandus A, Arnol M, Omahen K, Oblak M, Vidan-Jeras B, Kmetec A, Bren AF (2010) Basiliximab versus daclizumab combined with triple immunosuppression in deceased donor renal transplantation: a prospective, randomized study. Transplantation 89:1022–1027. https://doi.org/10.1097/TP.0b013e3181d02496
Ke N, Berkmen M (2014) Production of disulfide-bonded proteins in Escherichia coli. Curr Protoc Mol Biol. https://doi.org/10.1002/0471142727.mb1601bs108
Khan KH (2013) Gene expression in mammalian cells and its applications. Adv Pharm Bull 3:257–263. https://doi.org/10.5681/apb.2013.042
Lai JY, Klatt S, Lim TS (2019) Potential application of Leishmania tarentolae as an alternative platform for antibody expression. Crit Rev Biotechnol 39:380–394. https://doi.org/10.1080/07388551.2019.1566206
Majidi J, Barar J, Baradaran B, Abdolalizadeh J, Omidi Y (2009) Target therapy of cancer: implementation of monoclonal antibodies and nanobodies. Hum Antib 18:81–100. https://doi.org/10.3233/HAB-2009-0204
Nazari R, Davoudi N (2011) Cloning and expression of truncated form of tissue plasminogen activator in Leishmania tarentolae. Biotechnol Lett 33:503–508. https://doi.org/10.1007/s10529-010-0470-y
Niimi T (2012) Recombinant protein production in the eukaryotic protozoan parasite Leishmania tarentolae: a review. Methods Mol Biol 824:307–315. https://doi.org/10.1007/978-1-61779-433-9_15
Stockwin L, Holmes S (2003) The role of therapeutic antibodies in drug discovery. Biochem Soc Trans 31:433–436. https://doi.org/10.1042/bst0310433
Sugino M, Niimi T (2012) Expression of multisubunit proteins in Leishmania tarentolae. Methods Mol Biol (Clifton, NJ) 824:317–325. https://doi.org/10.1007/978-1-61779-433-9_16
Young R, Rance J (2012) Mammalian expression vector with a highly efficient secretory signal sequence. Google Patents, https://patents.google.com/patent/US20100240097A1/en
Acknowledgements
This article was extracted from Akram Jalali`s PhD thesis and financially supported by the ‘School of Advanced Technologies in Medicine, Shahid Beheshti University of Medical Sciences (though Grant No. 5086), and was carried out at the Cellular and Molecular Biology Research Center, the directors of which we gratefully.
Funding
The study was funded by ‘School of Advanced Technologies in Medicine, Shahid Beheshti University of Medical Sciences, Tehran, Iran (though Grant No. 5086).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There are no conflict of interests for authors.
Ethics Approval
The ethics process was approved by Shahid Beheshti University of Medical Sciences Ethical Committee with IR.SBMU.REC.1394.99 number.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jalali, A., Bandehpour, M., Chegeni, R. et al. Expression, Purification, and Evaluation of Anti-IL-2Rα Antibody Secreted by Leishmania tarentolae. Int J Pept Res Ther 27, 301–307 (2021). https://doi.org/10.1007/s10989-020-10088-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10989-020-10088-6